Neutropenia Incidence: A Retrospective Analysis of Clinical Data of Cancer Patients Receiving Systemic Anti-Cancer Therapy to Assess the Impact on Treatment Pathways
Authors: Amy Vercell1*, Janelle Yorke2, Dawn Dowding3
*Corresponding Author: Amy Vercell, Christie Patient Centred Research, The Christie NHS Foundation Trust, Wilmslow Road, Manchester, UK.
Received Date: 02 November, 2021
Accepted Date: 25 November, 2021
Published Date: 30 November, 2021
Citation:
Vercell A, Yorke J, Dowding D (2021) Neutropenia Incidence: A Retrospective Analysis of Clinical Data of Cancer Patients Receiving Systemic Anti-Cancer Therapy to Assess the Impact on Treatment Pathways. Int J Nurs Health Care Res 4: 1261. DOI: https://doi.org/10.29011/2688-9501.101261
Abstract
Background: Cancer represents a major health issue and economic burden to healthcare systems worldwide. Almost one third of people who have cancer will receive systemic anti-cancer therapy, which can result in treatment toxicities, such as neutropenia, that patients must navigate. Identifying those at highest risk of neutropenia related complications to refine existing clinical pathways can not only improve the patient experience, but also increase efficiency of hospital resources.
Objective: To explore the incidence of patients with solid tumours who have their systemic anti-cancer therapy deferred due to neutropenia or attend the cancer center unwell post treatment.
Method: A retrospective analysis of clinical data extracted from electronic patient records and electronic prescribing systems for specific time periods.
Findings: The total number of deferrals due to neutropenia were 7% (n = 224) in the last quarter of 2019 and 5% (n = 169) in the last quarter of 2020. The total 2020 data showed the infection care pathway was triggered by 980 patients. The majority (70%, n = 686) had received systemic anti-cancer therapy for a solid tumour within the last six weeks. Of these, 11% (n = 74) were neutropenic.
Conclusion: The highest incidence of neutropenia related complications was seen in patients with breast, gynecological and lower gastrointestinal cancers. However, the numbers of patients with confirmed neutropenia are low.
Keywords: Acute oncology; Clinical pathways; Medical oncology; Neutropenia incidence; Neutropenic sepsis; Systemic anti-cancer therapy; Treatment deferrals.
Introduction
Many people living with cancer who are receiving systemic anti-cancer therapy (SACT) will experience adverse events outside of the hospital setting, in their own homes [1]. Advances in therapies and clinical services have seen an increase in patients receiving treatment in the outpatient setting [2]. In a United Kingdom (UK) based cancer center where this retrospective analysis was carried out, if patients are unwell, they are asked to contact the 24 hrs hospital hotline to enable a clinical assessment to be carried out, to instigate appropriate management. Those patients with signs and symptoms of possible neutropenic sepsis will be advised to attend hospital for review by the Acute Oncology (AO) team in accordance with national guidelines (National Institute for Health and Care Excellence [3]. This could result in a hospital admission and/or a SACT administration deferral. However, not all patients who attend hospital are found to be neutropenic; it is not uncommon for patients to be medically reviewed and discharged, as clinical examination is normal, and full blood count (FBC) is within satisfactory parameters. Conversely, patients can attend for a SACT administration appointment and be told that their neutrophils are too low for treatment to be delivered safely and subsequently have their treatment deferred by at least one week. The aim of this retrospective analysis was to explore which disease groups and SACT regimens incurred the highest incidence of neutropenia related complications to guide future research regarding home blood monitoring in how to improve safe, timely and efficient SACT delivery.
Literature Review
Cancer incidence rates are projected to rise by 42.5% over the next 20 years [4], with 50% of the population expected to receive a diagnosis of cancer at some point in their lifetime [5]. Cancer represents a major health issue and economic burden to healthcare systems worldwide. Around 28% of people who have been diagnosed with cancer receive SACT [6]. They may incur treatment toxicities and side effects that patients and their caregivers must navigate and manage, with the support and education from their clinical team.
The systemic delivery of drugs that are antineoplastic are classed as SACT [7]. These drugs can include traditional cytotoxic chemotherapy, as well as newer, biological agents, such as monoclonal antibodies, targeted therapies, and immune checkpoint inhibitors [8]. These therapies have varying modes of action, with their anti-cancer effects impacting the cell cycle, causing numerous acute toxicity profiles that are specific to the treatment that has been administered [9].
These toxicities can vary in their severity and required management, ranging from requiring supportive medication in an outpatient setting, to hospitalization and dose interruptions and dose reductions [10]. The National Confidential Enquiry into Patient Outcome and Death [11], (NCEPOD) report (2008) identified concerns regarding the mismanagement of patients receiving SACT who developed neutropenic sepsis, and the subsequent incidence of avoidable deaths.
There are variations in definitions, but for the purpose of this paper neutropenic sepsis is defined as an absolute neutrophil count of 0.5 X 109 per liter or less, combined with a temperature greater than 38o C or any signs or symptoms of sepsis [12]. The National Chemotherapy Advisory Group [13], produced a report that focused on safety and quality, highlighting improvements around the provision of care for cancer patients in terms of elective chemotherapy services and emergency care.
NICE guidelines were created to provide evidencebased recommendations for the prevention, identification, and management of this life-threatening complication of cancer treatment [3]. A key factor in this is prompt recognition of those at risk of neutropenic sepsis.
Neutropenia is a common side effect of conventional cytotoxic chemotherapy, with standard-dose regimens typically causing a nadir around 7-10 days, which can incur around 7 days of neutropenia [14]. There are thought to be around 8 cases per 1000 patients who are receiving chemotherapy who develop neutropenic sepsis [15]. Improved management of neutropenic sepsis has been shown to improve morbidity and mortality rates [16]. Early intervention for suspected neutropenic sepsis has been proven to optimize outcomes, with broad spectrum antibiotic administration time critical [17], the goal being to attenuate clinical deterioration [18].
A facilitator in expediting administration of empirical intravenous antibiotics is the implementation of a patient group direction (PGD), whereby nurses in the UK can administer the first dose of antibiotics to any patient with suspected neutropenic sepsis without a prescription [19]. This, as part of an evidence based, robust clinical pathway, has been pivotal in reducing the door to needle time of first dose antibiotic [20]. The development of ambulatory emergency care pathways for oncology patients in the UK has been recognized as a key tenet in promoting patient safety, whilst reducing pressures on Emergency Departments [21,22].
A literature search, including UK national datasets for SACT activity, did not provide any reliable data on the incidence and impact of neutropenia on SACT deferrals. Variation of definitions used, varying causes of neutropenia included in study populations, as well as including numerous differing types of malignancy, without using standardized statistical models for analysis has resulted in poor quality data [3]. A survey carried out to explore the incidence of unplanned service use by patients who had received their first cycle of chemotherapy identified that treatment toxicities are common and can cause interruptions in their delivery, concluding that proactive symptom management improves toxicity burden and reduces patient reported distress [23]. A systematic review and meta-analysis that explored the association of cancer treatment delay and mortality found that this is a worldwide problem. Whilst this is focused upon the initiation of cancer treatments, rather than deferrals during established SACT delivery, suggest that a delay of as little as four weeks for surgery, SACT and radiotherapy can increase mortality for seven cancers, highlighting the importance of minimizing delays [24]. Fuentes and Frodin analyzed the frequency and indications for late (< 48 hrs) intravenous SACT deferrals and found that 13% (n = 383) of appointments were cancelled late [25]. Hematological toxicity was the deferral reason for 29% (n = 111) of all deferrals.
Objectives
The aims of this retrospective analysis were to
- Identify the number of SACT deferrals made due to neutropenia
- Identify the most prevalent disease groups and SACT regimens that incur a neutropenic deferral
- Identify the number of patients who attend the cancer center unwell post SACT and trigger the infection care pathway; and
- Identify the most prevalent disease groups and SACT regimens that trigger the infection care pathway.
Method
A retrospective analysis using clinical data extracted from electronic patient records (EPR) and iQemo, a SACT electronic prescribing system. Patients with haematological malignancies and lymphoma were excluded from the dataset in accordance with a separate study protocol, which was the impetus for this analysis.
SACT Delivery
The SACT deferral data focusses upon two comparable timeframes: the last quarters of 2019 and 2020. The rationale for focusing on these timeframes was activity in 2020 was skewed by the COVID-19 pandemic, where many cancer services were suspended, thus focusing on this calendar year alone would provide an inaccurate representation of typical SACT activity. However, towards the end of 2020, activity had almost returned to baseline, and thus, a pragmatic decision was made to analyses these two comparable timeframes to indicate overall activity.
Total numbers of intended SACT treatments for these two quarters were identified, with any treatments that were rescheduled for any reason ascertained from iQemo. The rescheduled treatments were reviewed to identify those patients who had a neutrophil count of less than 1.5 x 109 per liter. Patient notes were scrutinized to identify the number of SACT deferrals made due to neutropenia.
Patients who attend the cancer center unwell post SACT
The incidence of patients for the full calendar year of 2020 who attended the cancer center’s main site unwell post SACT with potential neutropenic sepsis was identified. Most common disease groups and SACT regimens were explored and actual numbers of patients who were found to be neutropenic were identified by reviewing the EPR.
Results
SACT Delivery
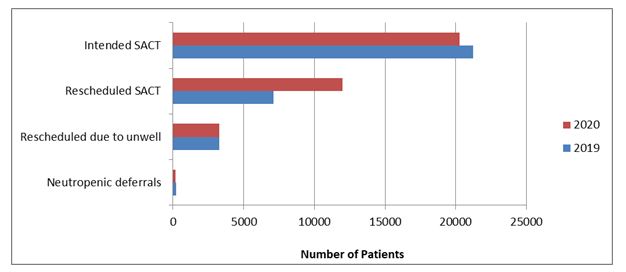
In the last quarters of 2019 and 2020 respectively, the total number of intended SACT treatments was 21,218 and 20,266, of which 7128 (33.6%) and 11,973 (59.1%) were rescheduled, respectively. Deferrals made due to neutropenia totally 224 patients (7%) in 2019, compared with 169 (5%) in 2020. These data are illustrated in (Figure 1).
Patients with breast, gynecology and lower gastrointestinal (GI) cancers experienced the highest incidence of neutropenic deferrals. This is illustrated in (Figure 2).
Patients receiving Oxaliplatin and Modified de Gramont for lower GI cancer had the highest deferral rates in 2019, totally 21 (9 %), followed by patients with metastatic breast cancer receiving Palbociclib (n = 18, 8 %). Twelve patients (5 %) with ovarian cancer receiving Carboplatin and Paclitaxel had their treatment deferred in 2019. In contrast, in 2020 individuals receiving Carboplatin and Paclitaxel had the highest deferral rates with 23 patients (14 %) having their treatment deferred. Two regimens given to people with lower GI cancers; Oxaliplatin and Modified de Gramont, and Irinotecan and Modified de Gramont, totally 18 (11 %) and 15 (9 %) neutropenic deferrals in 2020 respectively. These data are illustrated in (Figures 3, 4).
Patients who attend the cancer centre unwell post SACT
In 2020, 980 individuals were referred to the cancer center unwell via an outpatient route and triggered the infection care pathway. Of these, 70% (n = 686) had received SACT within the last six weeks for a solid tumour. As with the SACT activity data, patients with breast, lower GI and gynecological cancers were the largest cohorts who attended the hospital unwell post treatment, which accounted for 45% of all patients who came in unwell, as illustrated in (Figure 5).
There were 74 (11%) patients who attended the hospital unwell post SACT who were found to have a neutrophil count <1.0 x 109 per liter. Patients with ovarian cancer were the largest cohort to be admitted unwell and found to be neutropenic, accounting for 16% (n = 12) of the patients, with people with osteosarcoma accounting for 15% (n = 11), as illustrated in (Figure 6).
The SACT regimens support this, with Doxorubicin and Cisplatin having the highest incidence of neutropenic admissions; 9% (n = 7), followed by Vincristine, Doxorubicin, Cyclophosphamide, Etoposide, Ifosfamide (VDC IE) (7%, n = 5), which are regimens given to patients with sarcoma. There were smaller numbers of multiple SACT regimens given to patients with ovarian cancer that totally 12 patients (16 %), as illustrated in (Figure 7).
There were 612 patients (89%) who attended the cancer center unwell post SACT who were found to have a neutrophil count ≥ 1.0 x 109 per liter. Patients with colon cancer were the largest patient cohort (n = 78, 13%), and this was reflected in the top two regimens, which were Oxaliplatin and Modified de Gramont (n = 27) and Irinotecan and Modified de Gramont (n = 24), as reflected in (Figures 8 and 9).
Discussion
The results of this retrospective analysis highlight that the incidence of neutropenia related SACT deferrals was relatively low, and that the numbers of patients admitted to hospital with confirmed neutropenia is also low. These data illustrated slightly fewer neutropenia related deferrals in 2020 compared to 2019, which could be due to a known increased use of Granulocyte Colony-Stimulating Factor (GCSF) during the pandemic, which was nationally led, the goal of which was to minimize neutropenic episodes, and thus, reduce the risk of a hospital admission. However, the process of diagnosing neutropenia represents a burden to the patient and healthcare system, with potentially deleterious consequences to the patient if mismanaged. SACT induced toxicities and their impact are considered key indicators of quality for patient centered cancer care [23], and therefore, prompt detection of neutropenia can not only improve clinical outcomes, but also patient experience.
The cancer center where this service evaluation was conducted treats patients across a very large geographical area, with some patients travelling a significant distance for their appointment to subsequently find that their treatment is not going ahead as planned. From a patient experience perspective, this is challenging in terms of the physical and mental effort required to attend hospital, as well as the financial burden of travelling. There is a growing awareness of cancer poverty, with financial hardship being a significant aspect of receiving a cancer diagnosis.
A report written by Macmillan identified that 83% of people with cancer will be worse off financially, as they experience a loss of income and/or increased costs as a direct consequence of a cancer diagnosis [26]. Capacity on the cancer unit was a frequent reason for rescheduling, so reducing last minute deferrals for neutropenia would free up more appointment slots, as supported by Fuentes and Frodin [25].
Outpatient ambulatory care for emergency patients with a low-risk febrile neutropenia has already been proven as safe and effective [27]. However, there are challenges regarding the delivery of outpatient care that can mean it is not feasible for all patients, with transport issues being a key constraint [15].
It was apparent that patients with breast, gynecological or lower GI cancers had the highest incidence of neutropenic complications. This is unsurprising, as these are the largest patient cohorts that are treated and are also predominantly treated with conventional cytotoxic chemotherapy. It is important to note that novel treatments, such as immunotherapy, do not tend to cause profound neutropenia as is typically seen in conventional cytotoxic chemotherapy [28-30] explored the incidence of isolated neutropenia in patients who are receiving immune checkpoint inhibitors and claim that this is a rare but serious adverse event, although underreported. The data in this service evaluation supports this, as there were no reported incidences of neutropenic episodes related to immunotherapy.
There was a cohort of patients in 2019 with small cell lung cancer who were found to be neutropenic but did not incur a treatment deferral. This was discussed with the lung team, who explained that these patients were having their bloods checked two weeks after the first cycle of chemotherapy. This was a historic protocol that arose from findings from the NCEPOD (2008) report to assess nadir and treatment toxicities to maximize proactive management should dose interruptions or reductions be required. Due to COVID-19, this face to face appointment was cancelled, with a remote consultation taking place instead, without blood tests being obtained.
Limitations
Whilst every effort has been made to ensure all data have been captured for this analysis, there is a possibility that a small number may have been missed. Those patients who have their bloods obtained locally to them will not have the results recorded on the cancer center’s system. If they are found to be neutropenic and thus, have their treatment deferred, to capture this activity a note needs to be documented onto the EPR. If this note is not made, data analysts will not have captured them within our data. However, a discussion with the lead nurse of the outreach service suggests that this occurrence is rare, so it can be assumed that our results are generally reflective of the reality.
Conclusion
This retrospective analysis identified that there are a small but significant patient group who experience neutropenia related complications. The highest incidence was seen in patients with breast, gynecological and lower GI cancers. However, the numbers of patients with confirmed neutropenia are low. The creation of a home blood monitoring device as part of a separate study was the impetus for this retrospective analysis of clinical data. The development of minimally disruptive healthcare strategies for people living with cancer who are receiving SACT are essential to optimize their quality of life, whilst simultaneously improving the efficiency of hospital resources. These data could be used to guide where to commence research into exploring where within existing clinical pathways home blood monitoring could sit.
Acknowledgements
This paper could not have been written without the assistance of two research assistants, Jake Coomber-Moore and Claire Barnes, who helped enormously with the data collection.
Amy Vercell is partially funded by the National Institute for Health Research Applied Research Collaboration Greater Manchester and supported by the NIHR Manchester Biomedical Research Centre. The views expressed in this publication are those of the author(s) and not necessarily those of the National Institute for Health Research or the Department of Health and Social Care.
Figures
References
- Marshall E, Young A, Clark P, Selby P (2014) Problem Solving in Acute Oncology. Clinical Publishing, Oxford.
- Cooksley T (2021) Emergency Oncology in the United Kingdom. In: Todd K.H., Thomas, Jr. C.R., Alagappan K. (eds) Oncologic Emergency Medicine. Springer, Cham.
- National Institute for Health and Care Excellence (NICE) (2020) Neutropenic sepsis: prevention and management in people with cancer.
- Smittenaar C, Petersen K, Stewart K, Moitt N (2016) Cancer incidence and mortality projections in the UK until 2035. Br J Cancer 115: 1147
- Cancer Research UK (2020) cancer incidence statistics. Cancer Research UK.
- National Cancer Registration and Analysis Service and Cancer Research UK (2018) Chemotherapy, Radiotherapy and Surgical Tumour Resections in England. London: Public Health England,
- NHS England (2013) 2013/14 NHS standard contract for cancer: chemotherapy (adult). B15/S/a.
- Leach C (2019) Complications of systemic anti-cancer treatment. Medicine 48: 48-51.
- Hanahan D, Weinberg RA (2011) Hallmarks of Cancer: The Next Cell 144: 646-674.
- Djebbari F, Stoner N, Lavender VT (2018) A systematic review of nonstandard dosing of oral anticancer therapies. BMC Cancer 18: 1154.
- The National Confidential Enquiry into Patient Outcome and Death (NCEPOD) (2008) Systemic Anti-Cancer Therapy: For better, for worse?
- Forde C, McMullan R, Clarke M, Wilson RH, Plummer R, et al. (2020) Early switch from intravenous to oral antibiotic therapy in patients with cancer who have low-risk neutropenic sepsis (the EASI-SWITCH trial): study protocol for a randomised controlled trial. Trials 21: 431.
- National Chemotherapy Advisory Group (NCAG) (2009) Chemotherapy Services in England: Ensuring quality and safety.
- Klastersky J, de Naurois J, Rolston K, Rapoport B, Maschmeyer G, et al. (2016) ‘Management of febrile neutropaenia: ESMO Clinical Practice Guidelines. Ann Oncol 27: v111-v118.
- Cooksley T, Holland M, Klastersky J (2015) Ambulatory Outpatient Management of patients with low risk febrile neutropaenia. Acute Med 14: 178-181.
- Clarke RT, Jenyon T, van Hamel Parsons V, King AJ (2013) Neutropenic sepsis: management and complications. Clin Med (Lond) 13: 185-187.
- Mattison G, Bilney M, Haji-Michael P, Cooksley T (2016) A nurse-led protocol improves the time to first dose intravenous antibiotics in septic patients post chemotherapy. Support Care Cancer 24: 5001-5005.
- White L, Ybarra M (2017) Neutropenic Fever, Hematology/Oncology Clinics of North America 31: 981-993.
- National Institute for Health and Care Excellence (NICE) (2017) Patient group directions. Medicines practice guidelines. MPG2.
- Forde C, Scullin P (2017) Chasing the Golden Hour – Lessons learned from improving initial neutropenic sepsis management. BMJ Qual Improv Rep 6: u204420.w6531.
- Cooksley T, Marshall W, Ahn S, Lasserson DS, Marshall E, et al. (2020) Ambulatory emergency oncology: A key tenet of future emergency oncology care. Int J Clin Pract 74: e13436.
- Marshall E (2019) Ambulatory management in low risk neutropenic sepsis - A plea for integrated acute cancer care. Acute Med 18: 6-7.
- Harrison JM, Stella PJ, LaVasseur B, Adams PT, Swafford L, et al. (2016) Toxicity-Related Factors Associated with Use of Services Among Community Oncology Patients. J Oncol Pract 12: e818-e827.
- Hanna T, King W, Thibodeau S, Jalink M, Paulin GA, et al. (2020) Mortality due to cancer treatment delay: systematic review and meta BMJ 371: m4087.
- Fuentes S, Frödin JE (2015) Why is intravenous chemotherapy cancelled and how often. Could it be prevented? A prospective analysis of all planned and given intravenous anti-tumor treatments at the Department of Oncology, Karolinska University Hospital, Stockholm during one month. Acta Oncol 54: 1056-1062.
- Macmillan Cancer Support (2013) Cancer’s hidden price tag: Revealing the costs behind the illness.
- Marshall W, Campbell G, Knight T, Al-Sayad T, Cooksley T, et al. (2020) Emergency Ambulatory Management of Low-Risk Febrile Neutropenia: Multinational Association for Supportive Care in Cancer Fits—Real-World Experience From a UK Cancer Center. J Emerg Med 58: 444-448.
- Boegeholz J, Brueggen CS, Pauli C, Dimitriou F, Haralambieva E, et (2020) Challenges in diagnosis and management of neutropenia upon exposure to immune-checkpoint inhibitors: meta-analysis of a rare immune-related adverse side effect. BMC Cancer 20: 300.
- Naqash A, Appah E, Yang L, Muzaffar M, Marie MA, et al. (2019) ‘Isolated neutropenia as a rare but serious adverse event secondary to immune checkpoint inhibition. J Immunother Cancer 7: 169.
- Williams MD, Braun LA, Cooper LM, Johnston J, Weiss RV, et al. (2004) Hospitalized cancer patients with severe sepsis: analysis of incidence, mortality, and associated costs of care. Crit Care 8: R291-R298.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.