Zenker’s Diverticulum with Charcot-Marie-Tooth Disease: A Case Report of Rare Occurrence in a Middle-Aged Male Treated with Esophageal Cricopharyngeal Myotomy
by Sagar K Kamaraju*, Saketh K Kamaraju, Praveen K Kamaraju
McLaren Bay Region, Bay City, Michigan, USA
*Corresponding Author: Sagar K Kamaraju, McLaren Bay Region, Bay City, Michigan, USA
Received Date: 02 September 2025
Accepted Date: 08 September 2025
Published Date: 10 September 2025
Citation: Kamaraju SK, Kamaraju SK, Kamaraju PK. (2025). Zenker’s Diverticulum with Charcot-Marie-Tooth Disease: A Case Report of Rare Occurrence in a Middle-Aged Male Treated with Esophageal Cricopharyngeal Myotomy. Ann Case Report. 10: 2400. DOI:10.29011/2574-7754.102400
Abstract
Charcot-Marie-Tooth disease (CMT) is a hereditary degenerative condition characterized by slow progression, and it ranks among the most prevalent neuromuscular disorders [1]. Zenker's diverticulum (ZD) is an acquired sac-like outpouching of tissue located dorsally at the pharyngoesophageal junction through Killian's dehiscence and believed to be caused by dysfunction of the cricopharyngeal muscle [2, 3]. We detail the case of a 48-year-old male, who presented to the emergency room for difficulty swallowing with intractable vomiting for one week. Endoscopic evaluation revealed a recurrent Zenker’s diverticulum, while esophageal motility distal to the diverticulum appeared normal on esophageal manometry. Following confirmation of Zenker’s diverticulum via EGD, an open cricopharyngeal myotomy and Zenker’s diverticulectomy was performed. The patient’s anatomy allowed for easy identification of the diverticulum, with minimal scar tissue, which helped protect the recurrent laryngeal nerves and made the open surgery easier to perform. This case demonstrates the importance of the effects of Charcot-Marie-Tooth disease on striated muscle within the gastrointestinal tract.
Keywords: Esophageal Manometry; Cricopharyngeal Myotomy; Zenker’s Diverticulectomy; Neuromuscular Disorders; Hyperlipidemia
Introduction
Charcot-Marie-Tooth disease (CMT) is a hereditary degenerative condition characterized by slow progression, and it ranks among the most prevalent neuromuscular disorders [4]. The predominant inheritance pattern in Charcot-Marie-Tooth disease is autosomal dominant, however there are also subtypes linked to X chromosomal and autosomal recessive inheritance. Besides various inheritance patterns, numerous genes are linked to CMT, highlighting the disorder's heterogeneity [5].
CMT is characterized by progressive weakness and degeneration of peripheral nerves, which can lead to impaired function of muscles, including the cricopharyngeal muscle [6]. Dysfunction of this muscle is significant because it contributes to the formation of Zenker’s diverticulum (ZD), which is an acquired sac-like outpouching of tissue located dorsally at the pharyngoesophageal junction through Killian's dehiscence [1,3]. ZD is an uncommon disorder found within elderly patients who show signs of dysphagia and pulmonary aspiration. ZD is often classified by size, typically measured in the craniocaudal direction. The three size classifications are small (up to 2 cm), intermediate (2-4 cm), and large (greater than 4 cm) [7]. Open surgery with cricopharyngeal myotomy has long been the conventional treatment with satisfactory results, but is associated with high complication rates [2]. Therefore, since Zenker’s diverticulum typically affects elderly patients, less invasive treatments are generally preferred.
Case Presentation
We report a case of a 48-year-old male, who presented to the emergency room with difficulty swallowing with intractable vomiting for one week. The patient reported that shortly after swallowing liquids, food, or medications, he would experience episodes of coughing or vomiting, raising suspicion of Zenker’s diverticulum and potentially oropharyngeal dysphagia. The patient has a past medical history of Charcot-Marie-Tooth disease type 1, hyperlipidemia, pulmonary embolism, and transposition of the great vessels, which required surgical correction during childhood. Furthermore, the patient reported losing weight and denied any history of diabetes or prior malignancies. Additionally, the patient’s family history includes hypotension in both parents, as well as maternal history significant for cancer and increased stroke risk.
An esophagogastroduodenoscopy (EGD) was performed, and initial examination revealed a dilated esophagus with Zenker’s diverticulum measuring 4.2 cm arising from the proximal esophagus at the pharyngoesophageal junction (Figure 1). Mild proximal esophageal dilation was observed, likely due to outflow obstruction caused by the diverticulum. No strictures or stenosis were identified in the mid or distal esophagus, and the gastroesophageal junction appeared normal. While a video swallow study was not performed, it may have provided additional information into the extent of this patient’s oropharyngeal dysphagia, particularly relevant in light of his symptoms and underlying history of CMT.
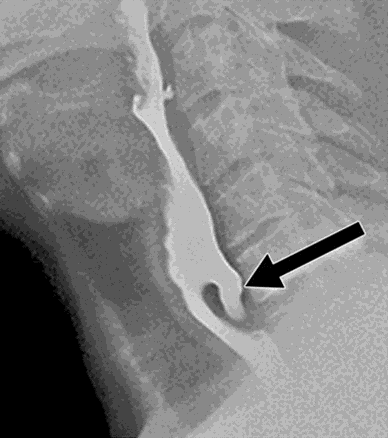
Figure 1: Clinical picture from [15] showing large Zenker’s diverticulum causing esophageal obstruction.
Nonetheless, esophageal manometry was then performed to evaluate for underlying motility disorders and showed normal function distal to the diverticulum (Figure 2). Given the patient’s symptoms, the size of the diverticulum, and his medical history, both endoscopic and open surgical approaches were discussed with the patient and his family at this time, who ultimately chose to proceed with an esophageal cricopharyngeal myotomy. Although less invasive methods such as Peroral Endoscopic Myotomy (POEM) and endoscopic esophageal myotomy with a needle knife are commonly used treatments for Zenker’s diverticulum, these modalities depend on favorable anatomy, which was not the case for this patient.
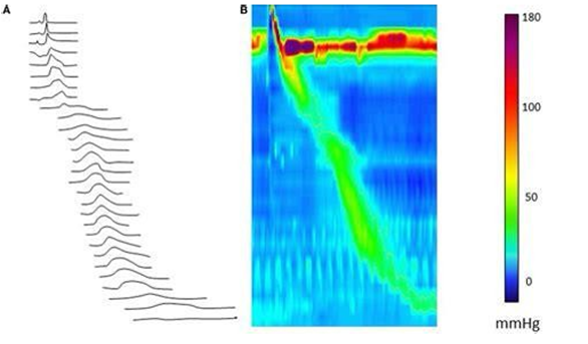
Figure 2: Esophageal manometry showing normal motility below diverticulum reproduced [16].
Therefore, the surgical team initially attempted an endoscopic approach using direct laryngoscopy with a Weerda scope, which advanced posteriorly along the pharyngeal wall until the esophageal diverticulum was seen. However, visualization was limited as the scope could not adequately access the diverticulum for safe endoscopic intervention (Figure 3). Given these limitations and the lack of advanced endoscopic services at our hospital, the decision was made to convert to an open surgical cricopharyngeal myotomy and diverticulectomy. A transverse
Incision was made through the skin and subcutaneous tissue proceeding through the platysma on the ipsilateral side. Dissection continued deeper towards the anterior aspect of the sternocleidomastoid muscle until the omohyoid muscle was identified. The dissection then continued medially until the superior aspect of the thyroid lobe was encountered, which was carefully retracted medially to improve access.

Figure 3: AI-generated intraoperative photograph demonstrating anatomical constraints using Weerda diverticuloscope
An esophageal dilator was then inserted to aid in palpation of the esophagus. This allowed for visualization of the cricopharyngeus muscle, which was identified by its characteristic transverse fibers. Just superior to the cricopharyngeus muscle layer, a large Zenker’s diverticulum that was adherent to surrounding tissue was visualized. Careful dissection was performed to mobilize the diverticulum, which was then elevated and pexied to the prevertebral fascia to provide clear access for the subsequent myotomy (Figure 4). The patient tolerated the procedure well, and there were no complications.
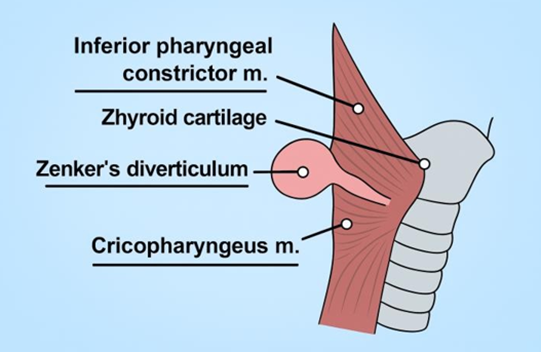
Figure 4: Schematic representation of the anatomical relationship between Zenker’s diverticulum and adjacent pharyngeal structures
Discussion
The presentation of Zenker’s diverticulum is reported to be uncommon in middle-aged men, with the average age of onset being 70-80 years old [8]. Soykan et al. reported that there are no known reports of gastrointestinal involvement with Charcot-Marie-Tooth disease [9]. A broad literature search reveals four cases which have reported laryngeal neuropathy in Charcot-Marie- Tooth disease [10]. There are currently no reported cases of Zenker’s diverticulum in patients with Charcot-Marie-Tooth disease.
CMT affects skeletal muscle by inducing weakness, which may subsequently lead to muscle atrophy. Although CMT manifests muscle symptoms, it primarily stems from nerve dysfunction rather than being a direct muscular disorder [11]. Impaired function of the cricopharyngeal muscle has been linked to the formation of Zenker's diverticulum [12]. Structural irregularities of the cricopharyngeus muscle hinder its ability to dilate properly, potentially contributing to the onset of Zenker's diverticulum.
This case is significant for its uniqueness in terms of the disease’s anatomical location, the patient’s demographic profile, and his underlying medical history. In a similar case, a 27-year old female presented with Charcot-Marie-Tooth disease and dysphagia; however, no reports of Zenker’s diverticulum were made [13].
Although the endoscopic stapling technique for Zenker’s diverticulum is widely recognized for its shorter operative time, decreased hospital stay, and faster return to resuming oral feedings compared to open surgical repair, it requires favorable anatomy and clear endoscopic visualization to perform the procedure safely. In our case, endoscopic access was limited due to anatomical constraints of the patient and attempts to visualize and expose the diverticulum endoscopically were unsuccessful. The failure to safely expose the diverticulum endoscopically leads to a considerable abandonment rate, which in turn causes significant postoperative complications. Moreover, while rare, endoscopic stapling techniques have led to the occurrence of bilateral vocal fold paresis, particularly when visualization is suboptimal [14]. As a result, the decision to convert to an open surgical approach was made. By proceeding with an open cricopharyngeal myotomy, we lessened traction on the recurrent laryngeal nerves and were able to perform a safe and precise dissection adapted to the patient’s specific anatomy.
Conclusion
It is important for medical providers to be aware of the possible development of Zenker’s diverticulum beyond the typical age of onset. In addition, patients with Charcot-Marie-Tooth disease present unique challenges that require careful management and follow-up. Charcot-Marie-Tooth disease can lead to the degeneration of the cricopharyngeal muscle causing Zenker's diverticulum. In summary, we report an interesting case of Zenker’s Diverticulum with a rare neurological disorder.
References
- Szigeti K, Lupski JR. (2009). Charcot–Marie–Tooth disease. European Journal of Human Genetics. 17: 703-710.
- Bizzotto A, Iacopini F, Landi R, Costamagna G. (2013). Zenker’s diverticulum: Exploring treatment options. Acta Otorhinolaryngologica Italica. 33: 219-229.
- Law R, Katzka DA, Baron TH. (2014). Zenker’s diverticulum. Clinical Gastroenterology and Hepatology. 12: 1773-1782.
- Sman AD, Hackett D, Fiatarone Singh M, Fornusek C, Menezes MP, et al. (2015). Systematic review of exercise for Charcot-Marie-Tooth disease. Journal of the Peripheral Nervous System. 20: 347-362.
- Morena J, Gupta A, Hoyle JC. (2019). Charcot-Marie-Tooth: From molecules to therapy. International Journal of Molecular Sciences. 20: 3419.
- Ganassi M, Zammit PS. (2022). Involvement of muscle satellite cell dysfunction in neuromuscular disorders: Expanding the portfolio of satellite cell-opathies. European Journal of Translational Myology. 32: 10064.
- Zaw ST, Zaw T, Haque M. (2023). Large Zenker’s diverticulum: A case report. Cureus. 15: e36783.
- Cleveland Clinic. (2024). Zenker’s diverticulum: Symptoms, causes & treatment. Cleveland Clinic. Accessed May 27, 2024.
- Soykan I, McCallum RW. (1997). Gastrointestinal involvement in neurologic disorders: Stiff-man and Charcot-Marie-Tooth syndromes. American Journal of the Medical Sciences. 313: 70-73.
- Benson B, Sulica L, Guss J, Blitzer A. (2010) Laryngeal neuropathy of Charcot-Marie-Tooth disease: Further observations and novel mutations associated with vocal fold paresis. The Laryngoscope. 120: 291-296.
- Charcot–Marie–Tooth Association. (2024). The differences between Charcot-Marie-Tooth disease and muscular dystrophy (MD): An overview. Charcot–Marie–Tooth Association. Accessed June 1, 2024.
- Schulze SL, Rhee JS, Kulpa JI, Danielson SK, Toohill RJ, et al. (2002) Morphology of the cricopharyngeal muscle in Zenker and control specimens. Annals of Otology, Rhinology & Laryngology. 111: 573-578.
- Naniwadekar A, Mishra V, Sanjeevi A. (2009). Dysphagia in a patient with Charcot-Marie-Tooth disease: 544. American Journal of Gastroenterology. 104: S204.
- Thorne M, Harris P, Marcus K, Teknos TN. (2004). Bilateral vocal fold paresis after endoscopic stapling diverticulotomy for Zenker’s diverticulum. Head & Neck. 26: 294-297.
- Jaffer NM, Ng E, Au WF, Steele CM. (2015). Fluoroscopic evaluation of oropharyngeal dysphagia: Anatomic, technical, and common etiologic factors. American Journal of Roentgenology. 204: 49-58.
- Rommel N, Rayyan M, Scheerens C, Omari T. (2017). The potential benefits of applying recent advances in esophageal motility testing in patients with esophageal atresia. Frontiers in Pediatrics. 5: 137.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.