Vitreous Metastasis from a Known Bladder Squamous Cell Carcinoma - A Case Report
by Charlotte Wortmann1, Clemens Lange1,3, Albrecht Lommatzsch1,2, Heinz-Wolfram Bernd4, Katrin Schlack5, Arnd Heiligenhaus1,2*
1Department of Ophthalmology at St. Franziskus Hospital Muenster, Germany
2Department of Ophthalmology, University of Duisburg-Essen, Germany
3Eye Center, University Hospital of Freiburg, Freiburg, Germany
4Hematopathology Luebeck, Luebeck, Germany
5Clinic for Urology, University of Muenster, Muenster Germany
*Corresponding author: Arnd Heiligenhaus, Department of Ophthalmology at St. Franziskus Hospital Muenster, Hohenzollernring 74, 48145 Muenster, Germany
Received Date: 03 March 2026
Accepted Date: 09 March 2026
Published Date: 11 March 2026
Citation: Wortmann C, Lange C, Lommatzsch A, Bernd HW, Schlack K, et al. (2026) Vitreous Metastasis from a Known Bladder Squamous Cell Carcinoma - A Case Report. Ann Case Report. 11: 2556. DOI: https://doi.org/10.29011/2574-7754.102556
Abstract
Background: Intraocular, particularly vitreous, metastases are extremely rare and this case highlights the diagnostic challenge of distinguishing such metastases from immune-mediated or lymphoproliferative processes in patients receiving immune checkpoint inhibitor therapy. Case Report: A 70-year-old woman with a history of squamous cell carcinoma of the urinary bladder presented with progressive visual loss in the left eye due to vitreous infiltration. Recently, she was treated with combined chemotherapy (gemcitabine/ cisplatin; GemCis) and nivolumab, a programmed death-1 (PD-1) binding immune checkpoint inhibitor (ICI). Initial vitrectomy revealed a cell-rich vitreous with T-cell clonality of unclear origin. After further recurrence of the vitreous infiltration, a second vitrectomy was performed, and this disclosed atypical epithelial cells positive for AE1/AE3, consistent with non-small cell carcinoma. The findings suggested vitreous metastasis from the known bladder carcinoma. Consequently, ICI / chemo- and eye-radiation therapy was instituted accordingly. Conclusion: This case underscores the diagnostic difficulty of vitreous opacities in patients with prior malignancy and immunotherapy, emphasizing the need for repeated vitrectomy and comprehensive pathological analysis to identify the exceptionally rare occurrence of vitreous metastasis from bladder squamous cell carcinoma.
Keywords: bladder squamous cell carcinoma, polymorphic vitreous cells, uveitis masquerade syndrome, vitreous cells, vitreous metastasis
Introduction
Intraocular metastases represent rare manifestations of systemic malignancies, most frequently originating from breast and lung carcinomas. [1] Metastatic involvement of the vitreous body is exceedingly uncommon and has been described only in rare cases.
Bladder carcinoma, particularly squamous cell type, very rarely metastasizes to ocular structures. The case illustrates the diagnostic challenges in distinguishing intraocular metastasis from immunemediated or lymphoproliferative processes, especially in patients undergoing immune checkpoint inhibitor therapy.
Case Presentation
A 70-year-old female patient was referred to our department with a suspected differential diagnosis of vitreous hemorrhage in the left eye. According to the patient’s history, she had noticed increasing floaters in the left eye in the recent two months. Approximately two weeks prior to presentation, her vision had become progressively cloudy.
Her ophthalmological history was otherwise unremarkable. Past medical history included a squamous cell carcinoma (SCC) of the urinary bladder, diagnosed two years earlier, and combined chemotherapy (gemcitabine/cisplatin; GemCis) and nivolumab, a programmed death-1 (PD-1) binding immune checkpoint inhibitor (ICI), in the recent 10 months. There was no known tobacco smoking, alcohol or substance abuse.
On examination, the patient was in stable general condition and fully oriented. The anterior and posterior segments of the right eye were unremarkable. Slit-lamp examination of the left eye revealed moderate pleomorphic keratic precipitates, few anterior chamber cells, and numerous polymorphic cells in the vitreous cavity. By ophthalmoscopy, the posterior segment was unremarkable.
B-scan ultrasonography of the left eye confirmed vitreous opacities without evidence of intraocular, retinal or extraocular mass formation (Figure 1). The optical coherence tomography (OCT) showed isolated pre-retinal adhesions.
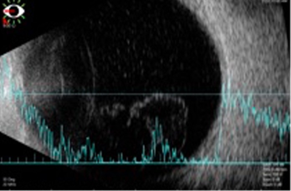
Figure 1: B-scan ultrasonography of the left eye with vitreous opacities
Results
Histopathological analysis demonstrated T-cell clonality; however, despite the presence of certain mutations, the underlying etiology could not be clearly determined. Except for a positive Borrelia IgG with negative IgM, the comprehensive blood and serological tests were inconspicuous. The patient was subsequently referred for hematologic and oncologic evaluation. Further diagnostics including cerebral MRI, PET-CT, and lumbar puncture showed no evidence of cerebral lymphoma. The consulting oncologists raised the differential diagnosis of a T-cell–mediated immune reaction to nivolumab. Whereas no other clinical or laboratory signs typical of an untoward side effect to the immune-checkpoint inhibitor was detected, an intraocular malignancy remained a possible (albeit rare) differential diagnosis.
Due to progressive pleomorphic keratic precipitates, anterior chamber cells, vitreous cell infiltration and now agglomeration of hyperreflective, pre-retinal deposits as detected by OCT (Figure 2), a second diagnostic vitrectomy with repeated pathological examination and Borrelia PCR was performed. Cytopathological examination of cytospin preparations revealed cell-rich material with anisometric and polymorphic cells. These corresponded mostly to atypical large epithelial cells with positive immunocytological staining for cytokeratin AE1/AE3 antibody, and a small population of interspersed small T lymphocytes positive for CD3. Accordingly, these findings were interpreted as carcinoma cells in the vitreous, consistent with metastatic spread of the known SCC of the bladder. (Figures 3, 4) Further evaluation by a second reference pathology laboratory confirmed the presence of numerous atypical epithelial cells consistent with a non–small cell carcinoma. No evidence of malignant lymphoma was found.
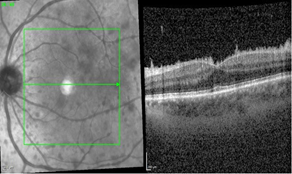
Figure 2: OCT with agglomeration of pre-retinal precipitates.
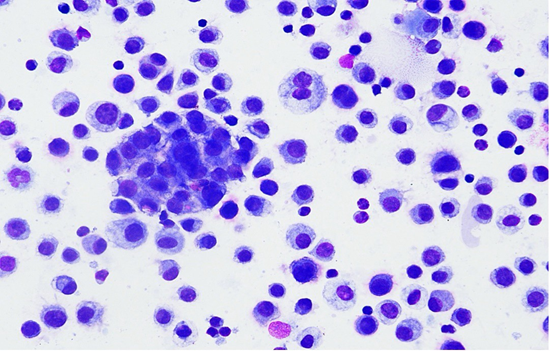
Figure 3: Atypical large epithelial cells with positive immuncytological staining for cytokeratin AE1/AE3 antibody.
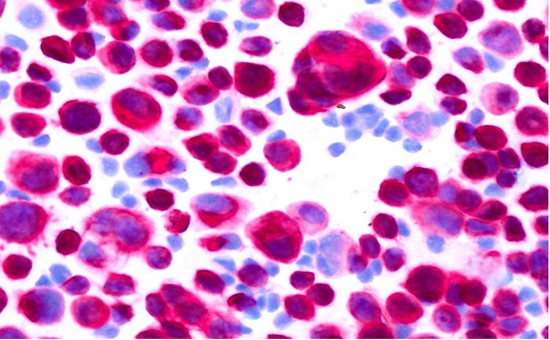
Figure 4: Atypical large epithelial cells, Pappenheim stain.
Taken together, these findings strongly supported the suspicion of a vitreous metastasis from the patient’s known bladder squamous cell carcinoma. Thus, GemCis and nivolumab were continued, and ocular radiation of the left orbita were initiated. Radiotherapy was delivered to a total dose of 50.4 Gy using intensitymodulated radiotherapy (IMRT) with a sliding window technique, administered in daily fractions of 1.8 Gy, five fractions per week.
Discussion
Intraocular metastases are uncommon and most often involve the uveal tract, particularly the choroid, due to its rich vascular supply. [2] Vitreous metastases, in contrast, are extremely rare and may occur secondary to hematogenous spread or direct extension from adjacent intraocular structures. Only a few cases of vitreous involvement from bladder carcinoma have been described previously in the literature [3-5].
In the present case, the patient’s known history of squamous cell carcinoma of the urinary bladder and the identification of atypical epithelial cells in the vitreous strongly suggest metastatic dissemination. The positive immunohistochemical staining for AE1/AE3 supported an epithelial origin of infiltrating cells, consistent with carcinoma. [6, 7] The lack of independent epithelial tumors at other sites of the body, and of intraocular or extraocular tumor masses on imaging and the lack of lymphoma markers in pathology helped exclude other potential diagnoses, such as primary intraocular lymphoma or inflammatory pseudotumor.
The differential diagnosis was further complicated by the patient’s prior immunotherapy with nivolumab, a PD-1 inhibitor known to induce various immune-mediated adverse effects, including ocular inflammation. [8, 9] A T-cell–mediated immune reaction was initially considered based on the first pathological findings of T-cell clonality. However, the subsequent demonstration of atypical epithelial cells obtained by a second vitrectomy confirmed the neoplastic rather than inflammatory nature of the process.
In line with the classic observations the diagnostic challenges encountered in our case are not unexpected. [10] Vitreous aspirates often contain scant, degenerated, or morphologically ambiguous cells, making reliable cytologic interpretation inherently difficult. Their work emphasized that epithelial cells are not normally present in the vitreous, and when identified, especially in cohesive or atypical clusters, they should raise strong suspicion for a malignant epithelial process rather than an inflammatory reaction. Moreover, it was highlighted that benign reactive cells may mimic malignancy, while small numbers of tumor cells may initially be overlooked, supporting our experience that the first diagnostic vitrectomy produced inconclusive results. Consistent with their recommendation that diagnostic yield can be improved through repeat vitreous sampling and adjunctive techniques, the second vitrectomy combined with immunohistochemistry ultimately enabled a clearer distinction between immune-mediated inflammation and metastatic epithelial infiltration in our patient.
In contrast to previously reported cases of ocular involvement by squamous cell carcinoma of the urinary bladder, in which retinal lesions and orbital infiltration were described, malignant cells in our case were detected exclusively in the vitreous body. The underlying mechanism responsible for this isolated vitreous infiltration remains unclear and warrants further investigation in future studies.
Conclusion
This case highlights the diagnostic challenges in patients with prior malignancy and immunotherapy presenting with vitreous opacities. Repeated diagnostic vitrectomy and comprehensive pathological evaluation, e.g. with immunohistochemistry and genetics are essential for distinguishing metastatic infiltration from immune-mediated or lymphoproliferative lesions. [11] The overall findings are most consistent with vitreous metastasis from bladder squamous cell carcinoma an exceedingly rare manifestation of this tumor entity.
Acknowledgements
We thank Thorwald Hakenes (Department of Ophthalmology at St. Franziskus Hospital) for his support during this study.
Patient consent
Written informed consent was obtained from patient.
Disclosure
The authors have no financial interest in any of the materials used in this study/case.
Funding
No funding or grant support
Conflicts of Interest
C.W. none; C.L. has received honorary by Amgen, AbbVie, Bayer, Novartis and Roche.; A.L. has received research grants from BMBF: 16SV8638 advisory: Bayer, Roche and Sandoz, lecture fees: Zeiss, Sandoz, Novartis, Roche, Bayer; H.W.B. none; K.S. has received honorary by AAA, Acromion, Amgen, Apogepha, Astellas, AstraZeneca, Bayer, BMS, DGU-Akademie, Eisai, EUSA-Pharma, Fosanis, Hexal, Ipsen, Janssen Cilag, Merck Healthcare, MSD, Novartis, Pfizer, Roche, Sanofi, streamedup!; A.H. has received research grants from BMBF (FKZ, 01ER1504C), DFG (He 1877/24-1), and honorary by Pfizer, MSD Sharp and Dohme, and Santen.
References
- Shields CL, Kalafatis NE, Gad M. (2023). Metastatic tumours to the eye. Review of metastasis to the iris, ciliary body, choroid, retina, optic disc, vitreous, and/or lens capsule. Eye (Lond). 37: 809-814.
- Shields CL, Shields JA, Gross NE, Schwartz GP, Lally SE. (1997). Survey of 520 eyes with uveal metastases. Oph-thalmology. 104 1265-1276.
- Whalen KE, Eagle Jr RC, Vrabec TR. (2018). CASE OF METASTATIC UROTHELIAL CARCINOMA OF THE RETINA AND VITREOUS. Retin Cases Brief Rep. 12: 177-180.
- Giordano G, Olivieri N, D’Andrea MR, Di Raimo T, Manfrin E, et al. (2019). Urothe-lial bladder carcinoma metastasizing to the eye: A systematic review and case report. Oncology Letters, 17: 462-467.
- Wang L-U, Hsieh T-H, Chen F-T, Chen Y-J, Wang J-K. (2024). Insights into Retinal Metastasis from Sys-temic Carcinoma: A Systematic Review of Clinical and Multimodal Imaging Characteristics. JCM. 2024; 13: 6037.
- Selves J, Long-Mira E, Mathieu MC, Rochaix P, Ilié M. (2018). Immunohistochemistry for Diagnosis of Metastatic Carcinomas of Unknown Primary Site. Cancers, 10: 108.
- Sinha GM, Hegde V. (2023). Detection of micro-metastasis using cytokeratins (AE1/AE3) in haematoxy-lin & eosin-stained N0 lymph nodes of oral squamous cell carcinoma. The Indian journal of medical research, 157: 316-321.
- Theillac C, Straub M, Breton AL, Thomas L, Dalle S. (2017). Bilateral uveitis and macular edema induced by Nivolumab: a case report. BMC ophthalmology. 17: 227.
- Martens A, Schauwvlieghe PP, Madoe A, Casteels I, Aspeslagh S. (2023). Ocular adverse events asso-ciated with immune checkpoint inhibitors, a scoping review. Journal of ophthalmic inflammation and infection, 13: 5.
- Midena E, Segato T, Piermarocchi S, Boccato P. (1985). Fine needle aspiration biopsy in ophthalmology. Surv Oph-thalmol. 29: 410-422.
- Gonzales JA, Chan CC. (2007). Biopsy techniques and yields in diagnosing primary intraocular lym-phoma. International ophthalmology. 27: 241-250.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.