Vascular and Nerve-Associated Inflammation in Lipedema Hand and Foot Tissue: A Case Report
by Virginia E. Bailey1, Karen L. Herbst2, Allison Scott3, Rachelle Crescenzi3, Scott T. Hollenbeck1, Sara Al-Ghadban1*
1University of Virginia, Department of Plastic Surgery, Charlottesville, Virginia, USA
2The Roxbury Institute, Tucson, Arizona, USA
3 University of Virginia, Department of Radiology and Medical Imaging, Charlottesville, Virginia, USA
Received Date: 27 March 2026
Accepted Date: 02 April 2026
Published Date: 06 April 2026
Citation: Bailey VE, Herbst KL, Scott A, Crescenzi R, Hollenbeck ST, et al. (2026). Vascular and Nerve-Associated Inflammation in Lipedema Hand and Foot Tissue: A Case Report. Ann Case Report. 11: 2581. DOI: https://doi.org/10.29011/2574-7754.102581
Abstract
Introduction: Lipedema is a chronic, progressive, and painful connective tissue disorder primarily affecting women. Symptoms include pain, limb heaviness, tenderness, easy bruising, and reduced mobility. Histologically, lipedema adipose tissue shows vascular dilation, adipocyte hypertrophy, and immune infiltration by macrophages and mast cells. Pain is thought to arise from heightened dermal sensitivity and neuropathic mechanisms. This study examines nerve-associated inflammation in lipedema, which has not been previously characterized.
Methods: Skin and subcutaneous adipose tissue (5-μm sections) were collected from one patient (hand, foot, back), fixed in formaldehyde, and stained for inflammatory markers (CD68, CD163) and endothelial cells (CD31). H&E and Masson’s trichrome staining were performed. Images were captured and manually quantified.
Results: Cutaneous tissue from the hands and feet demonstrated increased microvascular density (CD31) with thickened walls, perivascular fibrosis, and macrophage infiltration (CD68, CD163). Macrophages were observed along the nerve fibers and outer nerve layers, consistent with localized nerve inflammation alongside vascular remodeling.
Conclusion: This study demonstrates concurrent vascular remodeling and nerve-associated inflammation in lipedema adipose tissue of the hand and feet. These findings highlight that pain in lipedema involves both vascular and neurogenic inflammatory mechanisms, extending the understanding of lipedema pathology beyond the lower extremities.
Keywords: Lipedema; Adipose Tissue; Vascular Remodeling; Inflammation; Nerve Involvement; Fibrosis
Introduction
Lipedema is a chronic, progressive and painful connective tissue disorder characterized by symmetrical, disproportionate accumulation of subcutaneous fat in the extremities, typically sparing the hands and feet [1,2]. Lipedema is often accompanied by pain, easy bruising, heaviness and swelling in the affected areas [3,4]. With an estimated global prevalence of approximately 1 in 72,000 individuals and a marked female predominance, lipedema remains underdiagnosed, largely due to its clinical overlap with obesity, lymphedema, and other adipose tissue disorders [1, 5-8]. Common comorbidities include chronic venous disease [9], obesity, and mental health conditions such as anxiety and depression [10]. Connective tissue abnormalities are also increasingly recognized, with joint hypermobility [11] and connective tissue fragility, as seen in hypermobile Ehlers-Danlos syndrome (hEDS) [12], suggesting shared fascial pathology between connective tissue and adipose disorders.
Currently, there is no curative treatment for lipedema. Conservative management includes compression therapy, manual lymphatic drainage, exercise, and nutritional counseling, while surgical interventions such as tumescent liposuction are typically reserved for refractory cases and may improve function and quality of life [1-2,13]. Emerging evidence highlights the importance of addressing pain and inflammation as central components of disease management.
Pain in lipedema is a hallmark feature and may present as tenderness to pressure, spontaneous pain, and mechanical hypersensitivity [3,14-16]. While the mechanisms remain incompletely understood, both vascular and neurogenic components are thought to contribute. Histologically, lipedema adipose tissue demonstrates vascular remodeling and perivascular inflammation [17-19], including dilated blood and lymphatic vessels [17, 20-21], endothelial hyperplasia [22], perivascular fibrosis, and macrophage infiltration [23-25], which may contribute to interstitial fluid accumulation and tissue pain [17,26]. In addition, peripheral nerves may be affected by localized inflammation, leading to neurogenic pain and hypersensitivity. Despite increasing recognition of vascular and inflammatory alterations in lipedema adipose tissue, the extent to which these changes occur outside the lower extremities remains poorly understood. In this case report, we examine vascular remodeling and nerve-associated inflammation in lipedema tissue obtained from the hand and foot sites traditionally considered spared to better characterize the systemic nature of the disease.
Methods
Written informed consent was obtained from the patient prior to tissue collection.
Quality of Life Assessment (WHOQOL-100): Patient-reported quality of life was assessed using the validated World Health Organization Quality of Life-100 (WHOQOL-100) questionnaire. Surveys were completed and scored according to standard guidelines, with higher scores indicating better quality of life on a scale from 4 to 20.
Tissue Collection: Subcutaneous tissue samples were collected from the dorsal hands and feet, and back during tumescent liposuction procedures (Figure 1). Specimens were immediately fixed in 10% neutral buffered formalin, processed, and embedded in paraffin. Serial sections of 5 μm thickness were prepared for hematoxylin and eosin (H&E) staining and immunohistochemical analysis.
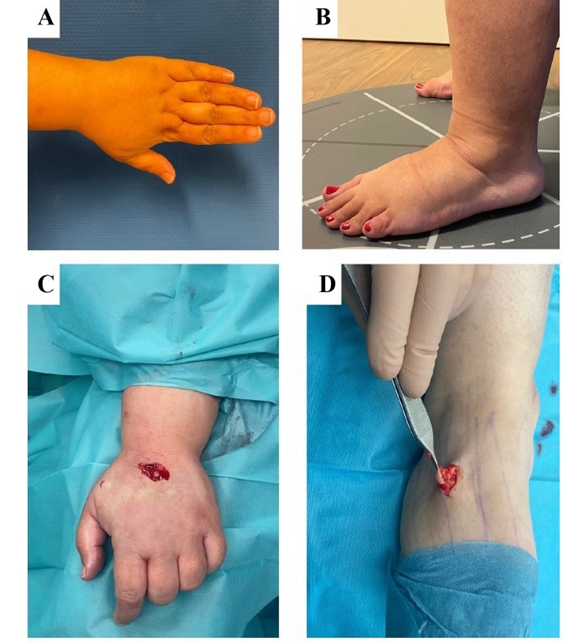
Figure 1: Clinical presentation and biopsy site localization in the hands and feet.
(A, B) Clinical images showing increased adipose tissue distribution in the hand and foot. (C, D) Skin biopsies obtained from the dorsal aspect of the hand and foot, respectively, showing the biopsy sites during tissue collection.
Immunohistochemistry and Histologic Analysis: All staining was performed by San Diego Pathologists Medical Group (California, USA). Paraffin-embedded tissue sections (skin and adipose tissue) were stained using antibodies against macrophages
(CD68, DAKO/Agilent M0814, CA, USA; CD163, NCL-LCD163, Leica Biosystems, IL, USA), endothelial cells (CD31,
DAKO/Agilent M0823, CA, USA) and Neurofilament (DAKO/ Agilent M0762, CA, USA). Sections were counterstained with hematoxylin. Masson’s trichrome staining (Sigma kit HT102A1KT, MO, USA) was used to evaluate collagen deposition and fibrosis. All slides were coded and evaluated in a blinded manner by two independent investigators. Positive control tissues known to express each marker were used to verify staining specificity, while negative controls were prepared by omitting the primary antibody. Images were acquired using an EVOS 5000 imaging system.
Case Presentation
A 45-year-old Arab female with a clinical diagnosis of Stage 3 lipedema presented with chronic, symmetric pain and swelling affecting the lower and upper extremities, abdomen, back, hands, and feet. Symptoms had been present for over 35 years and had significantly worsened during the past decade. The pain was described as aching and hypersensitive, most pronounced in the calves, upper arms, and feet, and was accompanied by heaviness, tenderness, and easy bruising. She reported that the pain worsened with prolonged standing and warm temperatures and improved partially with elevation, rest, and compression therapy, though relief was temporary. The patient denied numbness but reported progressive limitations in mobility, difficulty with clothing fit, and discomfort during routine daily activities. Symptom onset occurred around puberty (approximately age 10). Family history was notable for similar fat distribution and pain symptoms in female relatives on both the maternal and paternal sides. She also reported a family history of thyroid disease. Physical examination demonstrated bilateral, symmetric enlargement of tender, nodular subcutaneous adipose tissue on the feet, calves, thighs, abdomen, back, arms, breasts, and hands, with tender, nodular subcutaneous adipose tissue and non-pitting edema. Skin changes included uneven texture, visible telangiectasias, and frequent bruising. Her past medical history was notable for hypermobile Ehlers-Danlos syndrome (hEDS), mast cell activation syndrome (MCAS), and polycystic ovary syndrome (PCOS). Past surgical history included repair of a fractured humerus, cholecystectomy, and umbilical hernia repair. She had also undergone five prior lipedema reduction surgeries, including two skin excision procedures.
Results
Patient-Reported Quality of Life and Pain Profile The lowest-scoring domains were physical health, level of independence, and psychological health. Within these domains, the lowest-rated facets included body image and appearance (7/20), pain and discomfort (8/20), and sexual activity (9/20). The patient’s pain profile demonstrated a substantial impact on daily function, as reflected in both domain and facet scores and by the presence of pain even in the absence of external stimulation. The qualitative pain phenotype included sensations described as “tingling, throbbing, pressing, gnawing, cramping, tender, and taut” which may be consistent with an inflammatory pain pattern. Descriptors such as “pinching, burning, and splitting” were not reported.
Increased Blood Vessel Density, Fibrosis & Inflammation in Lipedema Foot Skin & Hypodermis
Histopathologic evaluation of lipedema skin from the foot revealed marked dermal vascular and stromal remodeling. H&E staining showed numerous small, irregular blood vessels in the dermis, some extending toward the epidermis (Figure 2A). Masson’s trichrome staining demonstrated dense collagen deposition around dermal vessels, consistent with fibrosis (Figure 2B). CD31 staining confirmed endothelial cells lining these vessels, several of which extended toward the epidermis (Figure 2C). CD68 and CD163 immunostaining identified perivascular macrophages, including M2-polarized cells in the dermis, suggesting a link between vascular remodeling and inflammatory or reparative responses (Figures 2D-E).
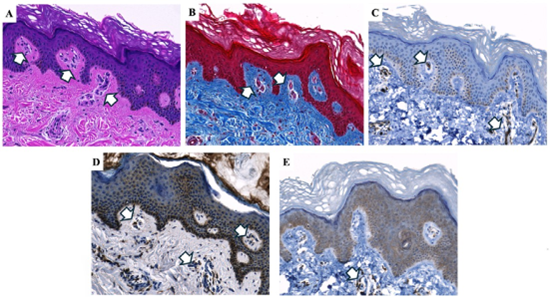
Figure 2: Histopathological and immunohistochemical analysis of lipedema skin from the foot. H&E staining (A) demonstrates blood vessels extending into the epidermis. Masson’s trichrome staining (B) demonstrates dense collagen deposition in the dermis (blue), indicative of fibrosis. CD31 immunohistochemistry (C) highlights endothelial cells, confirming the presence of blood vessels in both the epidermis and dermis. CD68 and CD163 staining (D, E) identifies perivascular macrophages surrounding dermal blood vessels. White arrows indicate blood vessels in panels A-C and macrophages in panels D-E. Images were acquired using a 20×objective.
Further analysis of the hypodermal tissue from the foot demonstrated prominent vascular enlargement and fibrosis. H&E staining revealed dilated blood vessels with increased vascular wall thickness, consistent with vascular hypertrophy and angiogenesis (Figure 3A). Masson’s trichrome staining highlighted marked collagen deposition surrounding enlarged vessels (Figure 3B). CD31 immunohistochemistry confirmed the vascular structures and demonstrated numerous enlarged blood vessels within the hypodermis (Figure 3C). The pronounced thickness of the CD31-positive vessel walls relative to the luminal space further emphasizes structural vascular remodeling and increased vascular density within lipedema-affected hypodermal tissue.
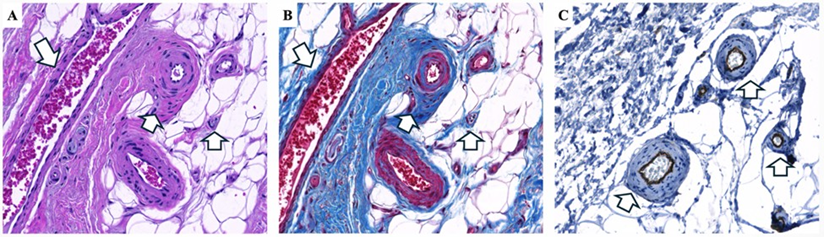
Figure 3: Histopathological and immunohistochemical analysis of hypodermal lipedema tissue from the foot. H&E staining (A) reveals enlarged blood vessels with increased vascular wall thickness; white arrows indicating multiple blood vessels (angiogenesis) and vascular wall hypertrophy. Masson’s trichrome staining (B) demonstrates marked fibrosis with dense collagen deposition (blue) surrounding enlarged vessels. CD31 immunohistochemistry (C) highlights endothelial cells, confirming the vascular structures and demonstrating enlarged blood vessels and angiogenesis. White arrows indicate blood vessels in all panels. Images were acquired using a 20× objective.
Fibrosis, Inflammation & Thick Capillary Wall in Lipedema Hand Skin & Hypodermis
Next, we examined skin and hypodermal tissue from the hand. Histopathologic analysis revealed vascular, fibrotic, and inflammatory changes similar to those observed in the lower extremity. H&E staining demonstrated increased dermal angiogenesis with distortion of the epidermal dermal junction and focal elongation of rete ridges (Figure 4A). Masson’s trichrome staining showed increased collagen deposition in the dermis and around blood vessels, extending toward the epidermis (Figure 4B). CD31 immunohistochemistry confirmed numerous dermal blood vessels of varying caliber (Figure 4C). CD68 staining identified macrophages distributed throughout the dermis and hypodermis, with prominent perivascular localization surrounding blood vessels (Figure 4D).
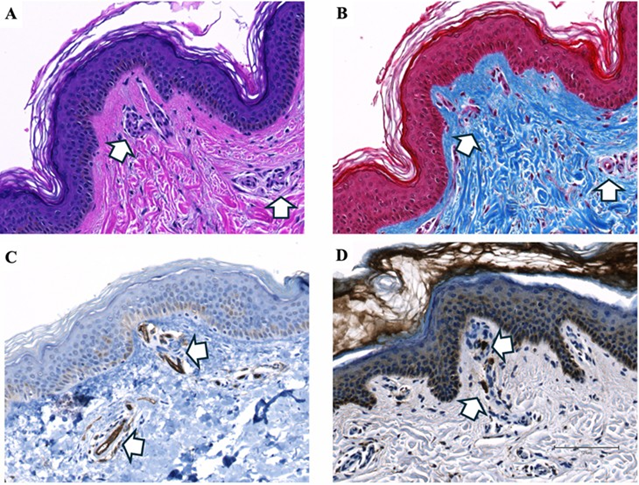
Figure 4: Histopathological and immunohistochemical analysis of lipedema skin from the hand. H&E staining (A) shows increased angiogenesis with numerous blood vessels in the epidermis and dermis, some extending toward the epidermal-dermal junction. Masson’s trichrome staining (B) demonstrates extensive dense collagen deposition (blue), consistent with fibrosis surrounding dermal vessels. CD31 immunohistochemistry (C) highlights endothelial cells, confirming the presence of blood vessels in the epidermis and dermis. CD68 staining (D) identifies macrophages surrounding the invading blood vessels. White arrows indicate blood vessels (A-C) or macrophages (D). Images were acquired using a 20× objective.
Further examination of hypodermal tissue from the hand demonstrated marked vascular enlargement and surrounding fibrosis, consistent with significant vascular remodeling. H&E staining revealed multiple enlarged and irregular vessels with thickened walls and increased vascular density within the hypodermis, indicative of angiogenesis and abnormal vascular remodeling (Figure 5A). CD31 immunohistochemistry confirmed the endothelial identity of these structures and highlighted prominent vessel wall thickening (Figure 5B), while adjacent lymphatic vessels appeared morphologically preserved. Masson’s trichrome staining demonstrated dense collagen deposition within the vessel walls and surrounding stroma, consistent with perivascular fibrosis and extracellular matrix expansion (Figure 5C). Higher-magnification sections illustrated a markedly enlarged hypodermal vessel with substantial wall hypertrophy (Figure 5D). CD31 staining confirmed the endothelial lining (Figure 5E), and corresponding trichrome staining demon.
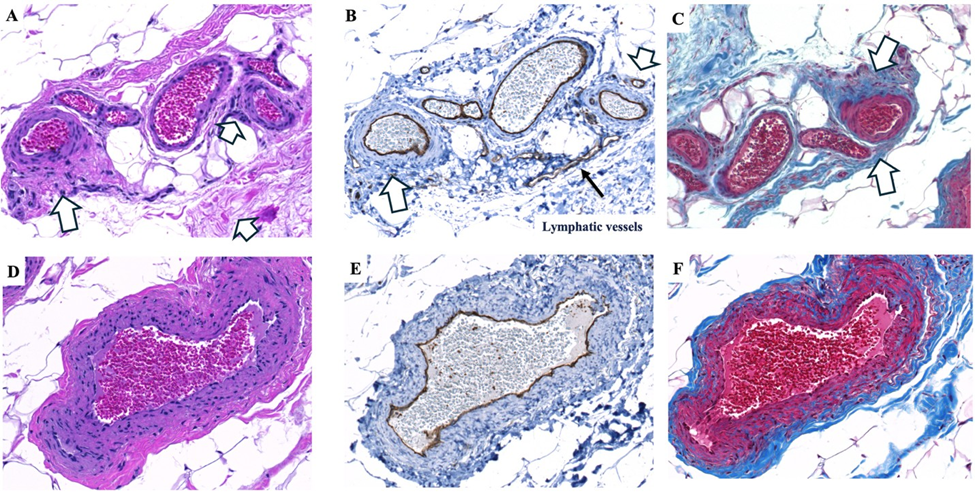
Figure 5: Histopathological and immunohistochemical analysis of lipedema hypodermis from the hand. H&E staining (A) shows enlarged blood vessels in the hypodermis, indicated by white arrows. CD31 immunohistochemistry (B) confirms these structures are blood vessels. Masson’s trichrome staining (C) demonstrates increased collagen deposition (blue) surrounding the enlarged vessels. A large hypodermal vessel (~400 µm in diameter) is shown with H&E staining (D) and confirmed by CD31 immunohistochemistry (E). Masson’s trichrome staining (F) highlights increased fibrosis within the thickened vessel wall. White arrows indicate blood vessels or areas of collagen deposition as shown. Images were acquired using a 20× objective.
Nerve Inflammation in Lipedema Hand and Foot
Immunohistochemical analysis revealed inflammatory cell infiltration in both hand and foot tissues. In particular, CD68-positive macrophages were observed surrounding peripheral nerves, localized to the perineural and epineural regions, indicating focal inflammatory changes within the nerve microenvironment. Similar patterns were observed with CD163 staining, confirming the presence of M2-polarized macrophages around peripheral nerves (data not shown). In the hand, CD68-positive macrophages surrounded small nerve fascicles, consistent with mild perineural infiltration (Figure 6A), while larger nerves showed macrophage accumulation along the perineural and epineural regions (Figure 6B). Similar patterns were observed in the foot, with clusters of macrophages surrounding peripheral nerve fascicles (Figure 6C). Additional sections demonstrated macrophages along the outer nerve sheath, highlighting persistent perineural inflammation (Figure 6D).
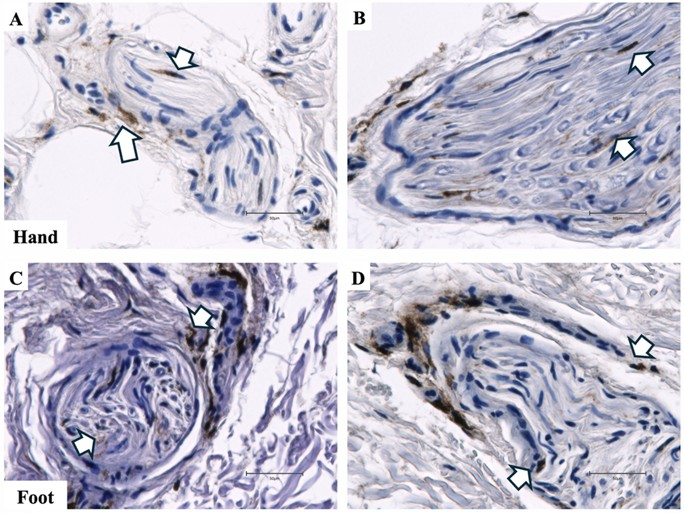
Figure 6: CD68 immunohistochemical staining demonstrating nerve inflammation in hand and foot tissue from a patient with lipedema. A. CD68 immunohistochemical stain shows macrophages (indicated by white arrows) surrounding nerve fascicles in the hand. B. CD68 immunohistochemical stain revealed macrophages (indicated by white arrows) in the epineural regions of the peripheral nerve in the hand. C. CD68 immunohistochemical stain of macrophages in the perineural and epineural regions of a nerve in the foot. D. CD68 immunohistochemical stain of macrophages in the perineural and epineural regions of a nerve in the foot. Images were acquired using a 40× objective.
Adipocyte Cysts in Lipedema Back Adipose Tissue
Hypertrophic adipose tissue is a hallmark of lipedema and is generally associated with fibrosis and inflammation throughout affected areas. Histologic evaluation of back subcutaneous adipose tissue revealed markedly enlarged cyst-like adipocyte structures associated with pericystic inflammation and fibrosis (Figure 7A). Masson’s trichrome staining showed collagen deposition along the cyst boundary, consistent with fibrotic remodeling of the surrounding tissue (Figure 7B). CD31 immunohistochemistry revealed endothelial & macrophage-associated staining around cells lining the cyst-like structures, suggesting vascular components within the microenvironment (Figure 7C). CD68 staining identified dense macrophage accumulation along the cyst perimeter, indicating prominent inflammatory infiltration (Figure 7D), and CD163 immunohistochemistry confirmed the presence of M2-polarized macrophages (Figure 7E). Highermagnification sections demonstrated fibrosis interspersed among macrophages lining the cyst wall (Figure 7F), CD31-positive staining surrounding the macrophage-lined boundary (Figure 7G), and CD163-positive macrophages forming part of the cellular lining of the cyst-like structures (Figure 7H), highlighting the combined inflammatory, vascular, and fibrotic components of these adipose tissue changes.
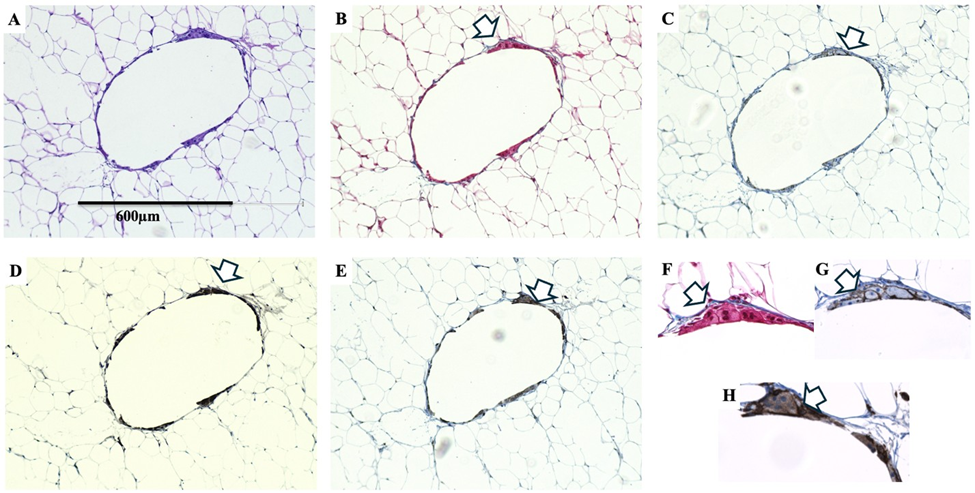
Figure 7: Histologic and immunohistochemical characterization of a large cyst-like adipocyte structures in lipedema adipose tissue. H&E staining (A) shows an enlarged cyst-like structure in the hypodermis. Masson’s trichrome staining (B) demonstrates fibrosis surrounding the cyst (white arrow). CD31 immunohistochemistry (C) shows positive endothelial-associated staining around the cystadjacent macrophages. CD68 staining (D) reveals macrophages surrounding the cyst (white arrow), and CD163 immunohistochemistry (E) confirms the presence of M2-polarized macrophages around the structure. Higher-magnification sections show fibrosis between macrophages lining the cyst wall (F), CD31-positive staining around the macrophage-lined boundary (G), and CD163-positive macrophages forming part of the cyst lining (H). Images were acquired using a 10× objective (A-E) and a 40× objective (F-G).
Discussion
The hands and feet are usually unaffected in lipedema as is the upper torso, however, it has been reported that 22% of women with lipedema have nodular tissue on the hands and up to 42% had nodular fat on the feet [27]. The data from our current case supports the tissue from the hand, foot and back are consistent with lipedema tissue. Histologic analysis of lipedema tissue from the hand and foot revealed consistent microvascular remodeling, including increased vascular density, enlarged vessels, thickened vascular walls, and prominent perivascular fibrosis. CD31 immunohistochemistry confirmed abundant endothelial-lined vessels, some extending toward the epidermal-dermal junction in skin samples and showing marked wall hypertrophy in hypodermal vessels. These findings align with prior reports describing dilated capillaries, endothelial changes, and interstitial fibrosis in lipedema adipose tissue [17,22,26].
In addition to vascular alterations, a prominent inflammatory component was observed. CD68- and CD163-positive macrophages localized to perivascular regions and along peripheral nerve fascicles, including the perineural and epineural layers. This neuroimmune interaction supports a histopathologic correlate for the neuropathic pain frequently reported in lipedema patients [15] and suggests that immune-mediated remodeling extends beyond adipose tissue into the peripheral nerve microenvironment [28]. To confirm that nerve-associated inflammation is also present proximal tissue, peripheral nerves from five lipedema and five non-lipedema thigh samples were analyzed. Quantitative analysis demonstrated a higher average number of CD68-positive cells per nerve in lipedema tissue, although this difference did not reach statistical significance in this small cohort (Figure 8). These findings reinforce the concept of systemic neuroinflammation in lipedema. The involvement of distal tissues, including the hand and foot, expands the traditionally recognized anatomical distribution of lipedema and supports the concept of a systemic connective tissue disorder.
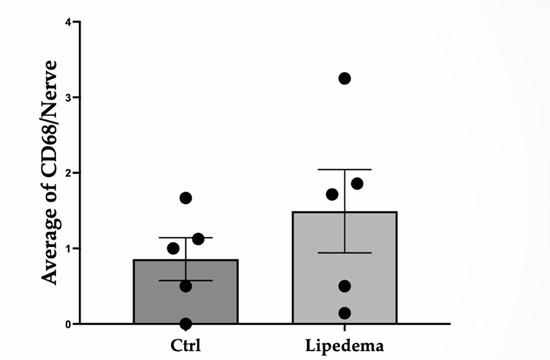
Figure 8: Average number of CD68-positive cells per nerve in non-lipedema (n = 5) and lipedema (n = 5) thigh adipose tissue. Nerves were identified using neurofilament immunostaining.
Notably, large cyst-like adipocyte structures in back adipose tissue, surrounded by macrophages and fibrotic tissue, indicate ongoing adipocyte degeneration and macrophage-mediated remodeling [29]. CD31 immunoreactivity was observed in macrophages surrounding these cysts. While CD31 is traditionally considered an endothelial marker, subsets of macrophages including tumorassociated or proangiogenic macrophages can express CD31 and promote vascular remodeling and angiogenesis in pathologic tissue microenvironments [30,31]. The presence of CD31-positive macrophages in lipedema tissue suggests that these cells may adopt a proangiogenic phenotype, contributing to local vascular hypertrophy, perivascular fibrosis, and extracellular matrix remodeling.
Together, these findings support a model in which lipedema reflects coordinated vascular, immune, and neural remodeling, rather than isolated adipose expansion, with capillary thickening, perivascular fibrosis, and nerve-associated inflammation as central pathologic features driving pain and tissue dysfunction. Recognition of this complex tissue microenvironment provides a framework for future studies targeting vascular dysfunction, immune activation, and neuroinflammatory signaling in lipedema.
Conclusion
Lipedema tissue from the hand and foot exhibits coordinated vascular, immune, and neural remodeling, including vessel enlargement, perivascular fibrosis, and macrophage infiltration along vessels and peripheral nerves. These findings provide a histopathologic correlate for neuropathic pain and support the concept of lipedema as a systemic connective tissue disorder.
Acknowledgments
The authors would like to thank Drs. Mojtaba Ghods, Philipp Kruppa (Klinikum Ernst von Bergmann, Potsdam) and Sabrina Gohlke (German Institute of Human Nutrition) for providing the materials used in this case report. We also thank Drs. Xin Li and Brant Isakson (University of Virginia) for their careful review of the manuscript.
Funding
Funding was provided by the National Institutes of Health (NIH) National Heart, Lung, and Blood Institute (NHLBI) grant R35HL184172. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Ethical Guidelines
Written informed consent was obtained from the patient prior to tissue collection and for publication of the associated clinical information and images. All procedures were conducted in accordance with institutional guidelines and the ethical principles outlined in the Declaration of Helsinki.
Conflict of Interest
The authors declare that they have no conflicts of interest related to this work.
References
- Herbst KL, Kahn LA, Iker E, Ehrlich C, Wright T, et al. (2021). Standard of care for lipedema in the United States. Phlebology. 36: 779-796.
- Kruppa P, Crescenzi R, Faerber G, Forner-Cordero I, Cornely M, et al. (2026) Lipedema World Alliance Delphi Consensus-Based Position Paper on the Definition and Management of Lipedema: Results from the 2023 Lipedema World Congress in Potsdam. Nat Commun. 17: 427.
- Al-Ghadban S, Evancio JV, Alfiscar PEF, Herbst KL. (2025). New Characterization of Lipedema Stages: Focus on Pain, Water, Fat and Skeletal Muscle. Life. 15: 1397.
- Buso G, Depairon M, Tomson D, Raffoul W, Vettor R, et al. (2019). Lipedema: A Call to Action! Obesity (Silver Spring) 27: 1567-1576.
- Poojari A, Dev K, Rabiee A. (2022). Lipedema: Insights into Morphology, Pathophysiology, and Challenges. Biomedicines. 10: 3081.
- Luta X, Buso G, Porceddu E, Psychogyiou R, Keller S, et al. (2025). Clinical characteristics, comorbidities, and correlation with advanced lipedema stages: A retrospective study from a Swiss referral centre. PLoS One. 20: e0319099.
- Beltran K, Herbst KL. (2017). Differentiating lipedema and Dercum’s disease. Int J Obes (Lond). 41: 240-245.
- Herbst KL. (2012). Rare adipose disorders (RADs) masquerading as obesity. Acta Pharmacol Sin. 33: 155-172.
- Dean SM, Valenti E, Hock K, Leffler J, Compston A, et al. (2020). The clinical characteristics of lower extremity lymphedema in 440 patients. J Vasc Surg Venous Lymphat Disord. 8: 851-859.
- Al-Wardat M, Clarke M, Alwardat N, Kassab M, Salimei C, et al. (2022). The Difficulties in Emotional Regulation among a Cohort of Females with Lipedema. Int J Environ Res Public Health. 19: 13679.
- Fiengo E, Sbarbati A. (2025). Lipedema and Hypermobility Spectrum Disorders Sharing Pathophysiology: A Cross-Sectional Observational Study. J Clin Med. 14: 7195.
- Wang TJ, Schubart JR, Wagner W, Mills S, Joyce RL, et al. (2025). Intersection between hypermobile Ehlers-Danlos syndrome and adipose disorders: investigating fascial remodeling with ultrasound imaging. Journal of Rare Diseases. 4: 52.
- Vazirnia A, Smart DR, Mohseni Y, Amron DM. (2026). Lipedema Diagnosis, Clinical Manifestations, and Therapeutics: A Systematic Review. Int J Dermatol. ijd.70227.
- Aday AW, Mc Donahue P, Garza M, Crain VN, Patel NJ, et al. (2024). National survey of patient symptoms and therapies among 707 women with a lipedema phenotype in the United States. Vasc Med. 29: 36-41.
- Chakraborty A, Crescenzi R, Usman TA, Reyna AJ, Garza ME, et al. (2022). Indications of Peripheral Pain, Dermal Hypersensitivity, and Neurogenic Inflammation in Patients with Lipedema. Inte J Mol Sci. 23: 10313.
- Patton L, Ricolfi L, Bortolon M, Gabriele G, Zolesioet P, et al. (2024). Observational Study on a Large Italian Population with Lipedema: Biochemical and Hormonal Profile, Anatomical and Clinical Evaluation, Self-Reported History. Int J Mol Sci. 25.
- Al-Ghadban S, Cromer W, Allen M, Ussery C, Badowski M, et al. (2019). Dilated Blood and Lymphatic Microvessels, Angiogenesis, Increased Macrophages, and Adipocyte Hypertrophy in Lipedema Thigh Skin and Fat Tissue. J Obes. 2019: 8747461.
- Felmerer G, Stylianaki A, Hägerling R, Wang A, Ströbel P, et al. (2020). Adipose Tissue Hypertrophy, An Aberrant Biochemical Profile and Distinct Gene Expression in Lipedema. J Surg Res. 253: 294-303.
- Grewal T, Kempa S, Buechler C. (2025). Lipedema: A Disease Triggered by M2 Polarized Macrophages? Biomedicines. 13: c561.
- Duhon BH, Phan TT, Taylor SL, Crescenzi RL, Rutkowski JM. (2022). Current Mechanistic Understandings of Lymphedema and Lipedema: Tales of Fluid, Fat, and Fibrosis. Int J Mol Sci. 23.
- Rasmussen JC, Aldrich MB, Fife CE, Herbst KL, Sevick-Muraca EM. (2022). Lymphatic function and anatomy in early stages of lipedema. Obesity. 30: 1391-1400.
- Michelini S, Greco S, Vaia N, Puleo V, Pellegrino P, et al. (2025). Endothelial cell alterations in capillaries of adipose tissue from patients affected by lipedema. Obesity (Silver Spring). 33: 695-708.
- Suga H, Araki J, Aoi N, Kato H, Higashino T, et al. (2009). Adipose tissue remodeling in lipedema: adipocyte death and concurrent regeneration. J Cutan Pathol. 36: 1293-1298.
- Kruppa P, Gohlke S, Łapiński K, Garcia-Carrizo F, Soultoukis GA, et al. (2023). Lipedema stage affects adipocyte hypertrophy, subcutaneous adipose tissue inflammation and interstitial fibrosis. Front Immunol. 14: 1223264.
- Wolf S, Rannikko JH, Virtakoivu R, Cinelli P, Felmerer G, et al. (2022). A distinct M2 macrophage infiltrate and transcriptomic profile decisively influence adipocyte differentiation in lipedema. Front Immunol. 13: 1004609.
- Allen M, Schwartz M, Herbst KL. (2020). Interstitial Fluid in Lipedema and Control Skin. Womens Health Rep. 1: 480-487.
- Herbst KL, Mirkovskaya L, Bharhagava A, Chava Y, Te C. (2015). Lipedema Fat and Signs and Symptoms of Illness, Increase with Advancing Stage. Archives of Medicine. 7: 10.
- Szél E, Kemény L, Groma G, Szolnoky G. (2014). Pathophysiological dilemmas of lipedema. Med Hypotheses. 83: 599-606.
- Camastra S, Vitali A, Anselmino M, Gastaldelli A, Bellini R, et al. (2017). Muscle and adipose tissue morphology, insulin sensitivity and beta-cell function in diabetic and nondiabetic obese patients: effects of bariatric surgery. Sci Rep. 7: 9007.
- McKenney JK, Weiss SW, Folpe AL. (2001). CD31 expression in intratumoral macrophages: a potential diagnostic pitfall. Am J Surg Pathol. 25: 1167-1173.
- Kim OH, Gun-Hyung K, Noh H, Ji-Young C, Ho-Jae L, et al. (2013). Proangiogenic TIE2(+)/CD31 (+) macrophages are the predominant population of tumor-associated macrophages infiltrating metastatic lymph nodes. Mol Cells. 36: 432-438.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.