Uterine Tumor Resembling Sex-Cord Tumors of the Ovary (Utrosct): A Case of Long-Term Response to Pazopanib
Giacomo Massa1*, Anna Tessari2, Silvia Damian1, Matteo Duca1, Elena Tamborini3, Federica Perrone3, Biagio Paolini4, Antonia Martinetti1, Elisa Sottotetti1, Filippo G. de Braud1, Sara Cresta1, Chiara Pircher1
1Medical Oncology Department, Fondazione IRCCS Istituto Nazionale dei Tumori. Milan, Italy
2The Ohio State University Comprehensive Cancer Center, Wexner Medical Center, Department of Cancer Biology and Genetics, College of Medicine. Columbus, Ohio
3Laboratory of Molecular Pathology, Department of Pathology, Fondazione IRCCS Istituto Nazionale dei Tumori. Milan, Italy
4Department of Pathology, Fondazione IRCCS Istituto Nazionale dei Tumori. Milan, Italy
*Corresponding author: Giacomo Massa, Medical Oncology Department, Fondazione IRCCS Istituto Nazionale dei Tumori. Milan, Italy
Received Date: 11 December 2022
Accepted Date: 15 December 2022
Published Date: 19 December 2022
Citation: Massa G, Tessari A, Damian S, Duca M, Tamborini E, et al. (2022) Uterine Tumor Resembling Sex-Cord Tumors of the Ovary (Utrosct): A Case of Long-Term Response to Pazopanib. Ann Case Report. 7: 1090. DOI: https://doi.org/10.29011/2574-7754.101090
Abstract
Background: uterine tumor resembling sex-cord tumors of the ovary (UTROSCT) is a rare form of mesenchymal uterine tumor of uncertain malignancy. Sporadic cases of advanced disease are reported, but little is known about its management in the metastatic setting. Pazopanib is a multi-tyrosine kinase inhibitor (TKI), approved for the treatment of advanced renal cell carcinoma and advanced soft tissue sarcomas, which affects several targets, including VEGFR1-2-3, PDGFRα-β and FGFR1-3. Case presentation: here we report a case of metastatic UTROSCT with strong and prolonged clinical response to pazopanib. Conclusions: these findings suggest that this anti-angiogenic approach should be considered in planning future therapeutic strategies for this rare disease. Furthermore, we noted the importance of including patients with rare diseases in clinical trials and investigating molecular pathogenesis.
Keywords: UTROSCT; Pazopanib; Therapeutic strategy
Abbreviations: ADC: Antibody-Drug Conjugate; BEP: Bleomycin, Etoposide, and cisplatin; CT: Computed Tomographic; CTCAE: Common Terminology Criteria for Adverse Events; ER: Estrogen Receptor; ESTSCLEs: Endometrial Stromal Tumors with Sex Cord-Like Elements; FDA: Food and Drug Administration; FISH: Fluorescence In Situ Hybridization; NGS: Next Generation Sequencing; PgR: Progesterone Receptor; TKI: Tyrosine Kinase Inhibitor; UTROSCT: Uterine Tumor Resembling Sex-Cord Tumors of the Ovary; WHO: World Health Organization
Background
Uterine tumor resembling sex-cord tumors of the ovary (UTROSCT) is a rare form of mesenchymal uterine cancer. First described by Morehead and Bowman in 1945 [1], its clinic-pathologic definition dates back to Clement and Scully in 1976 [2] who highlighted two different subtypes. Type 1 is pathologically similar to endometrial stromal tumors, with areas of sex chord-like structures comprising 10-40% of the total tumor mass. These cancers, known as endometrial stromal tumors with sex cord-like elements (ESTSCLEs), are associated with a significant risk of recurrence and metastases. On the other hand, Type 2 tumors (classic UTROSCTs) are composed of a predominant ovarian sex cord pattern (> 50%) and contained minimal to no stromal component. This neoplasm has a low malignant potential and typically exhibits benign biological behavior, however extrauterine or lymph node metastasis has been previously reported [3-7]. The current World Health Organization (WHO) classification includes type 1 in the low grade endometrial stromal tumors while type 2 tumors are individually categorized as UTROSCTs [8]. UTROSCTs usually occur in middle-aged women who complain pelvic pain or abnormal bleeding; occasionally these tumors are diagnosed incidentally [9]. Because of its rarity and low malignant potential, most of our knowledge on UTROSCT is limited to single case reports or small tumor series, and very little is known about the proper treatment in event of disease recurrence and distant metastasis.
Pazopanib (Votrient®, GlaxoSmithKline, Brentford, U.K.) is an oral ATP-competitive multi-tyrosine kinase inhibitor (TKI), whose targets include VEGFR1-2-3, PDGFRα-β, and FGFR1-3. It received the Food and Drug Administration (FDA) approval in 2009 for the treatment of advanced renal cell carcinoma [10] and subsequently in 2012 for the treatment of patients with advanced pretreated soft tissue sarcoma [11]. Pazopanib showed clinical efficacy also in other tumor types, such as thyroid cancer [12], ovarian cancer [13], and lung cancer [14].
Herein we report the first case in literature of metastatic UTROSCT that benefited from treatment with TKI pazopanib.
Case Presentation
A 56-year-old woman presented to the National Cancer Institute (Milan, Italy) in December 2009. The past medical and family history was unremarkable until 2001, when the patient underwent simple hysterectomy for multiple uterine leiomyomas. The resected specimen contained, among leiomyomas, a white-yellowish elastic nodule with no whorling appearance. The nodule was composed by atypical tumor cells with eosinophilic cytoplasm and prominent nucleoli, arranged in trabeculae and cord of gland-like structures, in a jaline stroma (Figure 1).
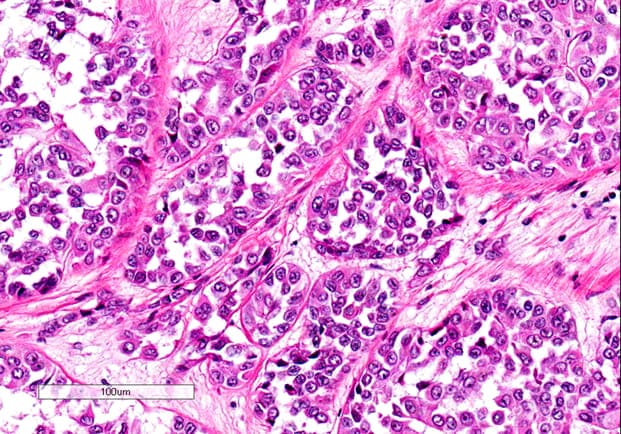
Figure 1: UTROSCT shows in this case a solid/nested pattern of growth. Tumor cells are uniform and epithelioid, with vesicular nuclei and distinct to prominent nucleoli (hematoxylin & eosin section, 20x).
Tumor cells focally infiltrated the adjacent myometrium, also causing vascular invasion. By immunohistochemistry, neoplastic cells were positive for cytokeratin pool AE1-AE3, vimentin, and calretinin, and negative for smooth muscle actin, alpha-inhibin, and thyroid transcription factor-1. A final diagnosis of UTROSCT was thus rendered.
The patient was followed until 2008, when a computed tomographic (CT) scan revealed widespread abdominal relapse of disease. Bilateral oophorectomy, appendicectomy, resection of multiple peritoneal, rectal, ascendant colon nodules, and peri-bladder mass were performed, due to the presence of more than 10 infiltrative nodules with diameters up to 8 cm. Appendix and fallopian tubes were free of disease. Histologic features were similar to those previously described, together with myxoid areas, cystic dilations, and mitotic activity up to 9/10 high power fields. Abdominal effusion was positive for tumor cells. Neoplastic cells stained positive for cytokeratin pool AE1-AE3, vimentin, calretinin, WT1, CD99, and focal smooth muscle actin, and negative for EMA, CD117, alpha-inhibin, MelanA/MART1, synaptophysin, CD10, CD56, CEA, and desmin. More than 95% of tumor cells expressed estrogen receptor (ER) and progesterone receptor (PgR). The proliferation index was <10%. Pathologic examination was consistent with metastasis of UTROSCT. No residual disease was evident upon surgery. The patient was then treated with three cycles of first line chemotherapy with cisplatin, etoposide, and bleomycin, complicated by ototoxicity and skin toxicity. On November 2008, at the end of the treatment, no evidence of disease relapse was seen at the CT scan.
On July 2009, PET scan documented two sub-glissonian nodules and abdominal nodal involvement, for which the patient underwent omentectomy and multiple peritoneal nodules resection (at least 9 nodules, maximum diameter 4 cm), still pathologically consistent with UTROSCT.
Three months later, the patient was enrolled in a phase I clinical trial at our Institution, evaluating the combination of carboplatin, paclitaxel, and ombrabulin. Ombrabulin is a synthetic analogue of combrestatin A4 that acts as vascular disrupting agent [15]. The treatment schedule consisted in the administration of ombrabulin 25 mg/mq intravenously on day 1, followed by carboplatin AUC 5 and paclitaxel 175 mg/mq on day 2 every 3 weeks. The patient experienced grade-2 arthralgia and myalgia, peripheral sensory neuropathy, asthenia, alopecia, and grade-1 itching, according to Common Terminology Criteria for Adverse Events (CTCAE) v3.0 [16]. All adverse events were transient and completely recovered after the end of treatment. After 6 cycles of therapy (last administration on April 2010), the CT scan documented stable disease, while the PET examination showed a complete metabolic remission. Due to liver disease progression not judged susceptible to surgery, on August 2010 we proposed to the patient a phase I combination of pazopanib plus anthracyclines [17]. The experimental treatment with epirubicin (90 mg/mq intravenously on day 3) in combination with pazopanib 800 mg/die per os from day 1 to day 8 in a three-week schedule was started on September 2010. After 6 cycles the CT scan showed stable disease and we suspended the administration of the anthracycline. Patient continued the treatment with pazopanib. The TKI weekly regimen was moved to a continuous one at the same dose of 800 mg/die, as for protocol. During the combination phase of the study, the patient experienced mild toxicities lasting up to three days, such as asthenia, nausea and myalgia, and transient asymptomatic grade 4 neutropenia. Pazopanib as monotherapy was well tolerated, with mild short-term adverse events. The CT scan after the 8th cycle of treatment revealed the achievement of partial response of disease (as per RECIST v1.0) [18]. This response was confirmed at subsequent evaluations until November 2013, when the patient experienced abdominal progression of disease.
Due to the strong ER and PgR positivity of the metastatic lesions, endocrine treatment with letrozole 2.5 mg/die per os was started. In January 2014, at the first disease evaluation, the patient had further abdominal progressive disease.
We performed two more lines of chemotherapy treatment (trabectedin and weekly paclitaxel) without clinical and radiological benefit.
On December 2015 the patient was enrolled in a phase I clinical trial at our Institution evaluating the combination of BI860585 (mTORC1 and mTORC2 serine/threonine kinase inhibitor) and exemestane 25 mg/die [19]. The treatment was well tolerated except for grade-2 stomatitis. Despite an initial disease stability, after 10 cycles of therapy the patient experienced liver progression.
On December 2016 we proposed a rechallenge with epirubicin, which was interrupt due to grade-3 pneumonia.
In the absence of further therapeutic strategies, the patient was enrolled in a phase I-II clinical trial with the combination of BMS-986148, a mesothelin-directed antibody drug conjugate (ADC), and Nivolumab (April 2017) [20]. After three cycles of therapy the patient experienced grade-3 ascites. Therefore, BMS-986148 was discontinued and the patient was treated with Nivolumab in monotherapy until progression occurred in December 2017. The patient dead on March 2018.
A metastatic nodule was biopsied (in January 2017, prior to the BMS-986148 trial) and evaluated by next generation sequencing (NGS) in order to identify any targeted mutations and explain the clinical benefit obtained from pazopanib treatment. Briefly, 10 ng/μl tumor DNA was isolated from paraffin sections using GeneRead DNA kit (Qiagen, Hilden, Germany, http://www.qiagen.com Cat. n. 180134) and analyzed according to the Hotspot Cancer Panel v2 (Life Technologies), which includes hotspot regions of 50 oncogenes and oncosuppressor genes frequently altered in human cancers, by using 316 chips on Ion Torrent Personal Genome Machine platform (Life Technologies). NGS analysis did not show any somatic mutation, but only two polymorphisms (aminoacidic substitution I391M due to single nucleotide polymorphism (SNP) in the exon 7 of the PIK3CA gene with 56% frequency, and aminoacidic substitution P72R due to a SNP in the exon 4 of the TP53 gene, with 100% frequency).
Discussion and Conclusions
UTROSCT is a rare uterine tumor with low malignant potential, for which clinical data are still limited. In the majority of cases primary surgical resection is curative [21-23]. Extensive surgical management based on hysterectomy plus salpingo-oophorectomy, parametrium resection, and pelvic and para-aortic lymphadenectomy should be considered in cases with higher disease burden and in recurrent disease [3-7, 22, 24]. No effective systemic therapy regimen has been still identified. However, in case of not resectable metastatic disease, chemotherapy with 3 cycles of bleomycin, etoposide, and cisplatin (BEP), as for treatment of ovarian sex cord malignancies, could be administered [5]. Endocrine treatment with the aromatase inhibitor anastrozole is another strategy to consider, in light of the frequent estrogen and progesterone receptors positivity of UTROSCT [25].
In contrast with these findings, we did not observe any clinical benefit from the aromatase inhibitor letrozole despite the high expression of ER and PgR. However, our patient obtained prolonged stable disease with exemestane and BI860585.
Conversely, the clinical response to pazopanib was impressive: its cytotoxic effect resulted in a confirmed reduction of tumor burden greater than 30%, lasting more than 3 years.
In keeping with previous publications, no alterations of JAZF1, SUZ12, EPC1 and PHF1 were found by fluorescence in situ hybridization analysis (FISH), indicating that UTROSCT are unlikely to be of endometrial stromal origin [26]. This entity was recently reported to exhibit recurrent gene fusions involving NCOA1, NCOA2, NCOA3, ESR1 and GREB1 discovered by FISH and targeted RNA sequencing [27-28]. At DNA-based NGS analysis we found P72R polymorphism on TP53 gene. It shows differences in different ethnicities, nothing is known about the association between this SNP and UTROSCTs. In the general population this polymorphism is not associated with an increased cancer risk if p53 is wild type. However, if p53 of cancer cells is mutated, the P72R SNP influences the expression of genes involved in metabolism (e.g., Warburg effect), tumor proliferation, and epithelial to mesenchymal transition [29-30]. To the best of our knowledge, there are no data in literature about the association of PIK3CA I391M and gynecological tumors.
In this rare disease, a genetic study with a large panel is essential, as is the enrollment of patients in clinical trials. Further studies are needed to determine predictive markers of response to personalized treatments.
In conclusion, we report the first documented response to pazopanib of this rare form of tumor. Both the magnitude and the duration of the clinical benefit (which lasted more than 3 years), together with the low toxicity profile of the treatment, suggest that this anti-angiogenic approach should be considered in planning future therapeutic strategies for this rare disease. Furthermore, we noted the importance of including patients with rare diseases in clinical trials and investigating molecular pathogenesis.
Declarations
Consent for publication: the patient died before the decision to write the case report. The caregiver is unavailable, despite several attempts. As demonstrated by the clinical history, the patient believed in scientific research, participated in several clinical trials and was proactive about cancer research.
Ethicsapproval: this case report obtained the approval from the Ethics Committee of Fondazione IRCCS Istituto Nazionale Tumori of Milan, Italy.
Competing interests: FdB receives consultant fees from Roche, EMD Serono, NMS Nerviano Medical Science, Sanofi, MSD, Novartis, Incyte, BMS, Menarini, Astra Zeneca, Pierre Fabre, speaker fees from BMS, Healthcare Research & Pharmacoepidemiology, Merck Group, MSD, Pfizer, Servier, Sanofi, Roche, AMGEN, Incyte, Dephaforum, Seagen, research fees from Novartis, F.Hoffmann-LaRoche Ltd, BMS, Ignyta Operating INC, Merck Sharp & Dohme Spa, Kymab, Pfizer, Tesaro, MSD, MedImmune LCC, Exelixis Inc., LOXO Oncology Incorporated, DAICHI SANKIO Dev. Limited, Basilea Pharmaceutica International AG, Janssen-Cilag International NV, Merck KGAA. The other authors have no conflicts of interest to disclose.
Availability of data and materials: we are available to discuss the case, the documents are made anonymous.
References
- Morehead RP, Bowman MC (1945) Heterologous Mesodermal Tumors of the Uterus: Report of a Neoplasm Resembling a Granulosa Cell Tumor. Am J Pathol 21: 53-61.
- Clement PB, Scully RE (1976) Uterine tumors resembling ovarian sex-cord tumors. A clinicopathologic analysis of fourteen cases. Am J Clin Pathol 66: 512-25.
- Biermann K, Heukamp LC, Buttner R (2008) Uterine tumor resembling an ovarian sex cord tumor associated with metastasis. Int J Gynecol Pathol; 27: 58-60.
- Kantelip B, Cloup N, Dechelotte P (1986) Uterine tumor resembling ovarian sex cord tumors: report of a case with ultrastructural study. Hum Pathol; 17: 91-4.
- O'Meara AC, Giger OT, Kurrer M (2009) Case report: Recurrence of a uterine tumor resembling ovarian sex-cord tumor. Gynecol Oncol; 114: 140-2.
- Umeda S, Tateno M, Miyagi E (2014) Uterine tumors resembling ovarian sex cord tumors (UTROSCT) with metastasis: clinicopathological study of two cases. Int J Clin Exp Pathol; 7: 1051-9.
- Macak J, Dundr P, Dvorackova J (2014) Uterine tumors resembling ovarian sex cord tumors (UTROSCT). Report of a case with lymph node metastasis. Cesk Patol; 50: 46-9.
- Kurman RJ, Carcangiu ML, Herrington CS, Young RH (2014) WHO Classification of Tumours of Female Reproducitve Organs. Lyon: International Agency for Research on Cancer; 2014.
- Czernobilsky B (2008) Uterine tumors resembling ovarian sex cord tumors: an update. Int J Gynecol Pathol 27: 229-35.
- Sternberg CN, Davis ID, Mardiak J (2010) Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J Clin Oncol; 28: 1061-8.
- van der Graaf WT, Blay JY, Chawla SP (2012) Pazopanib for metastatic soft-tissue sarcoma (PALETTE): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet 379: 1879-86.
- Bible KC, Suman VJ, Molina JR (2010) Efficacy of pazopanib in progressive, radioiodine-refractory, metastatic differentiated thyroid cancers: results of a phase 2 consortium study. Lancet Oncol; 11: 962-72.
- Friedlander M, Hancock KC, Rischin D (2010) A Phase II, open-label study evaluating pazopanib in patients with recurrent ovarian cancer. Gynecol Oncol; 119: 32-7.
- Koinis F, Agelaki S, Karavassilis V (2017) Second-line pazopanib in patients with relapsed and refractory small-cell lung cancer: a multicentre phase II study of the Hellenic Oncology Research Group. Br J Cancer 117: 8-14.
- Bahleda R, Sessa C, Del Conte G (2014) Phase I clinical and pharmacokinetic study of ombrabulin (AVE8062) combined with cisplatin/docetaxel or carboplatin/paclitaxel in patients with advanced solid tumors. Invest New Drugs 32: 1188-96.
- The Common Therminology Criteria for Adverse Events (CTCAE) version 3.0. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcaev3.pdf. Accessed 26 May 2022.
- Fasolo A, Sessa C, Bauer JA (2010) Phase Ib clinical and pharmacological study of multiple schedules of pazopanib (P) and epirubicin (EPI) in patients with advanced solid tumors. Journal of Clinical Oncology 28: 3047-3047.
- Jang GS, Kim MJ, Ha HI (2013) Comparison of RECIST version 1.0 and 1.1 in assessment of tumor response by computed tomography in advanced gastric cancer. Chin J Cancer Res; 25: 689-694.
- de Braud F, Machiels JH, Boggiani D (2020) A Phase 1 Study of mTORC1/2 Inhibitor BI 860585 as a Single Agent or with Exemestane or Paclitaxel in Patients with Advanced Solid Tumors. Cancers (Basel) 12.
- Rottey S, Clarke J, Aung K (2022) Phase I/IIa Trial of BMS-986148, an Anti-mesothelin Antibody-drug Conjugate, Alone or in Combination with Nivolumab in Patients with Advanced Solid Tumors. Clin Cancer Res 28: 95-105.
- Pradhan D, Mohanty SK (2013) Uterine tumors resembling ovarian sex cord tumors. Arch Pathol Lab Med 137: 1832-6.
- Gomes JR, Carvalho FM, Abrao M (2015) Uterine tumors resembling ovarian sex-cord tumor: A case-report and a review of literature. Gynecol Oncol Rep; 15: 22-4.
- Liu CY, Shen Y, Zhao JG (2015) Clinical experience of uterine tumors resembling ovarian sex cord tumors: a clinicopathological analysis of 6 cases. Int J Clin Exp Pathol; 8: 4158-64.
- Endo D, Todo Y, Okamoto K (2016) A case of recurrent group II uterine tumor resembling ovarian sex-cord tumors, against which two hormonal agents were ineffective. Taiwan J Obstet Gynecol; 55: 751-753.
- Blinman P, Tattersall MH (2009) A case of uterine tumour resembling ovarian sex cord tumour responding to second-line, single agent anastrazole. Intern Med J; 39: 617-9.
- Chiang S, Ali R, Melnyk N (2011) Frequency of known gene rearrangements in endometrial stromal tumors. Am J Surg Pathol; 35: 1364-72.
- Goebel EA, Hernandez Bonilla S, Dong F (2020) Uterine Tumor Resembling Ovarian Sex Cord Tumor (UTROSCT): A Morphologic and Molecular Study of 26 Cases Confirms Recurrent NCOA1-3 Rearrangement. Am J Surg Pathol; 44: 30-42.
- Lee CH, Kao YC, Lee WR (2019) Clinicopathologic Characterization of GREB1-rearranged Uterine Sarcomas With Variable Sex-Cord Differentiation. Am J Surg Pathol; 43: 928-942.
- Barnoud T, Parris JLD, Murphy ME (2019) Common genetic variants in the TP53 pathway and their impact on cancer. J Mol Cell Biol; 11: 578-585.
- De Souza C, Madden JA, Mann D (2020) The P72R Polymorphism in R248Q/W p53 Mutants Modifies the Mutant Effect on Epithelial to Mesenchymal Transition Phenotype and Cell Invasion via CXCL1 Expression. Int J Mol Sci; 21: 8025.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.