The Utility of Urine Dipstick Parameters in Excluding Bacteriuria in the Setting of Indwelling Ureteric Stents
by Matthew Wright-Smith1*, Simon Pridgeon1
Cairns Hospital, Cairns, QLD, Australia
*Corresponding Author: Matthew Wright-Smith, Cairns Hospital, Cairns, QLD, Australia
Received Date: 07 March 2026
Accepted Date: 11 March 2026
Published Date: 13 March 2026
Citation: Wright-Smith M, Pridgeon S (2026) The Utility of Urine Dipstick Parameters in Excluding Bacteriuria in the Setting of Indwelling Ureteric Stents. J Surg 11: 11581 DOI: https://doi.org/10.29011/2575-9760.011581
Abstract
Purpose: To determine the reliability of urine dipstick analysis leukocyte esterase and nitrite results in excluding pre-operative bacteriuria in patients with indwelling ureteric stents
Methods: Retrospective assessment of all patient records undergoing elective urological surgery in a single centre over a 24-month period. All patients with indwelling ureteric stents and pre-operative urine culture and dipstick test results were included. Urinary nitrite, pH, specific gravity, Leucocyte Esterase (LE), baseline patient characteristics, and stent dwell time and indication were considered.
Results: A total of 153 urine samples were included in the final analysis, of which 43 (28.1%) demonstrated significant bacteriuria. Female gender, diabetes, and increasing age, presence of infection at stent insertion, urinary nitrite and urinary leucocyte esterase were all significantly associated with increased rates of bacteriuria (p<0.05). LE activity at a cut point of “small” or greater had a sensitivity and specificity of 86% and 54%, with a NPV of 91% for the presence of bacteriuria. A more conservative LE cut point of “trace” or greater improved NPV to 100% but was only 17% specific. Incorporating nitrite results into an algorithm with LE did not improve predictive value above LE alone.
Conclusion: Readily available urinary dipstick parameter LE can exclude bacteriuria in the stented patient with reasonable reliability. However, the residual diagnostic uncertainty of this test remains too great to justify superseding routine pre-operative urine MCS.
Keywords: Bacteriuria; Pyuria; Stents; Urinalysis; Urology
Introduction
On the premise that it increases the risk of infective complications, pre-operative bacteriuria is considered a contraindication to urological surgery where the mucosa may be breached [1-5]. Urine Microscopy, Culture And Sensitivity (MCS) is the gold-standard for detection of bacteriuria [6]. The European Association of Urology Urological Infections Guidelines recommend screening for asymptomatic bacteriuria via urine MCS in all patients prior to elective endourological procedures, and if bacteriuria is present, treating pre-operatively [7]. Indwelling ureteric stents are common in urological practice, and may produce symptoms that mimic urinary tract infection (UTI) [8,9]. Furthermore, ureteric stents predispose to bacteriuria [10,11]. Thus, exclusion of pre-operative bacteriuria is especially important when indwelling ureteric stents are present. However, in everyday practice, urine MCS results are often unavailable on the day of surgery. As stent-related symptoms may be misdiagnosed as UTI, and urine MCS results take several days to return, potentially unnecessary surgical cancellations may occur, to the detriment of the healthcare system and patient. In contrast to MCS, urine dipstick analysis is a rapid and low-cost assay. The value of urine dipstick results for diagnosis of UTI in the general population has been extensively studied [6,12-15]. Pyuria is a common feature of UTI [6,16]. Leucocyte esterase (LE) assay indirectly signals pyuria through detection of proteins with esterolytic activity, which are present in lysed and intact human neutrophils [12,17]. The Griess urinary nitrite test indirectly detects the presence of nitrate-reducing bacteria, which convert excreted urinary nitrates to nitrite which is not normally found in urine [12,18]. Most, but not all, uropathogens are nitrate-reducing [12,18]. A meta-analysis of 23 studies supports the use of urinary dipstick tests to diagnose UTI in the general population – a positive urine nitrite and LE test in combination had a 96% specificity for UTI. Furthermore, a negative nitrite and LE test resulted a negative likelihood ratio of 0.22, translating to a pooled post-test probability of UTI of only 5% based on the pooled prevalence of 20% [15]. Accordingly, urologists may use urinary dipstick LE and nitrite results in daily practice to exclude bacteriuria when MCS results are unavailable. However, the predictive value of urinary dipstick results for bacteriuria in patients with indwelling ureteric stents is questionable. Leukocyturia is a common finding in stented patients [19-21]. Stent-related urinary frequency may decrease urinary dwell time, allowing insufficient time for bacteria to convert nitrate into nitrite [8,9], and non-nitrate reducing pathogens are often implicated in stent-related urinary infections [22]. To support this notion, Pooli et al (2016) analysed 122 urine specimens from patients with indwelling stents and found no significant correlation between urinary nitrite level and positive urine culture [19]. However, only 17 urine samples (13.9%) were positive on culture, and this may have underpowered the study. Thus, whilst urinary dipstick results offer a rapid and practical method of excluding pre-operative bacteriuria in patients without MCS results available, there is limited evidence to support their use in patients with indwelling ureteric stents. In this study, we aimed to determine the predictive value of urinary of rapidly available urinary dipstick results, nitrite and LE, as single tests and in combination, for the presence of bacteriuria in stented patients.
Method
Study population
This project was reviewed and approved by the local Ethics Committee (EX/2024/QCH/105702), and was completed in accordance with the Declaration of Helsinki. Records of all patients who underwent elective urological surgery at our hospital between October 2021 to September 2023 inclusive were retrospectively reviewed. All patients who submitted a pre-operative urine sample with a documented indwelling ureteric stent were included. Incomplete records, or patients with documented recent exposure to antibiotics (within 14 days) were excluded. Urine MCS results that were considered contaminated (growth of mixed skin species, or ≥20 epithelial cells per high-powered field) were excluded from final analysis.
Investigations
Midstream urine samples were collected under nurse instruction in the pre-admission clinic. Urine samples were immediately subjected to dipstick analysis via the Clinitek Status+ Analyzer (Siemens, Germany), then sent to microbiology for standard MCS protocols. Greater than 106 colony forming units per litre of a known urological pathogen was considered positive for bacteriuria. Mixed enteric species or multiple uropathogens within a single culture were included to reflect local clinical practice which considers these urine cultures positive. Samples that were culture positive with epithelial cell counts >10 x 106/L, or grew mixed skin species, were considered contaminated and excluded from final analysis.
Data Collection
Baseline patient demographics, date and indication of stent insertion, presence of urinary tract infection at time of stent insertion, diabetes and immunocompromised status, urine MCS results and urine dipstick analysis results (pH, Specific Gravity (SG), LE activity, nitrite) were gathered from medical records.
Statistical analysis
The upper cut-off for microscopy leukocyte count on pathology reports was “>500 x 106/L”; such results were assigned a value of 500 x 106 for statistical analysis. The Clinitek Status+ Analyzer reports LE activity and nitrite presence categorically. Any trace of nitrite was considered positive. LE activity was converted to an ordinal variable as follows: negative, 0; trace, 1; small, 2; medium 3; large, 4. Urinary dipstick LE results were converted from ordinal to a dichotomous positive/negative result using a cut-off point determined by the Youden index of Receiver Operating Characteristic (ROC) curve to maximise predictive value. For descriptive data, continuous variables are described using medians and Interquartile Ranges (IQR), and categorical variables using count and percentage. The two-tailed Mann-Whitney U test was used to assess the associated between continuous variables. For categorical variables, the Chi-squared test of independence was used. P<0.05 was considered significant. Predictive values of urinary nitrite and LE were calculated using standard cross tables. Correlation between urinary nitrite, SG and pH was tested using Spearman’s rho coefficient. Statistical analysis was performed with SPSS Statistics (V31) software (IBM, 2025). Confidence intervals for sensitivity, specificity and accuracy are exact Clopper-Pearson confidence intervals, confidence intervals for predictive value are standard logit confidence intervals, assuming overall disease prevalence to be equal to prevalence in the study group (28.1%) [23] (Figure 1).
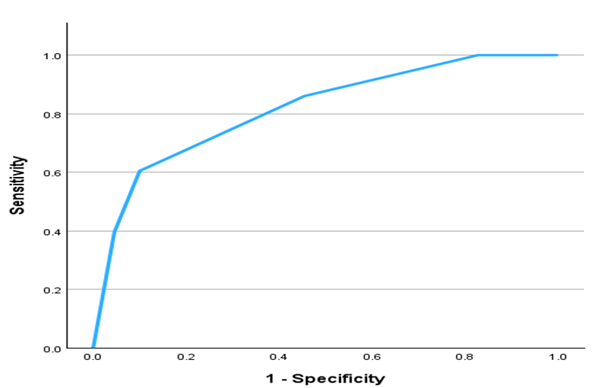
Figure 1: Urinary LE activity receiver operating characteristic (ROC) for presence of bacteriuria. Area under curve 0.82. For calculation, overall disease prevalence was assumed to be equal to prevalence in study group (28.1%).
Results
Baseline characteristics
A total of 244 urine specimens were reviewed. Ninety-one specimens were considered contaminated and excluded, leaving 153 samples for final analysis. Average age at specimen collection was 64.47yrs. Median stent dwell time was 54 days (range 3 to 1224). Table 1 summarises the baseline patient characteristics and investigations. 43 (28.1%) of urine samples demonstrated significant bacteriuria. Table 2 summarises the indications for stent insertion and pathogens isolated on MCS. Confirmed urinary tract infection was present during insertion of 31 stents. Thiry-seven stents had replaced a previously inserted stent. Only 11 urine samples (7.19%) were nitrite positive. There were 134 (87.53%) urine samples with some degree of LE activity. There was no significant correlation found between nitrite and either SG or pH.
|
Parameter (Normal range, unit) |
Sterile urine, n=110 |
Bacteriuria, n=43 |
P Value |
|
median (IQR) |
median (IQR) |
||
|
Age |
65 (23) |
72 (18.5) |
0.016 |
|
Urinary leucocyte microscopy count (<10, 106/L) |
130 (250) |
500 (0) |
<0.001 |
|
Urinary pH dipstick (4.5 - 8) |
5.5 (0.5) |
5.5 (0.75) |
0.734 |
|
Urinary specific gravity dipstick (1.005 – 1.030) |
1.02 (0.01) |
1.02 (0.01) |
0.032 |
|
Stent dwell time (days) |
52.5 (62.5) |
54 (118.5) |
0.377 |
|
Parameter |
Sterile urine |
Bacteriuria |
|
|
n (%) |
n (%) |
||
|
Gender (female) |
28 (25.45) |
22 (51.16) |
0.002 |
|
Nitrite positive |
3 (2.72) |
8 (18.6) |
<0.001 |
|
Diabetic patient |
18 (16.36) |
15 (34.88) |
0.012 |
|
Immunocompromised |
2 (1.81) |
3 (6.98) |
0.107 |
|
Stent replaced existing stent |
29 (26.36) |
8 (18.6) |
0.314 |
|
LE activity Neg |
19 (17.27) |
0 (0) |
<0.001 |
|
Trace |
41 (37.27) |
6 (13.64) |
|
|
Small |
39 (35.45) |
11 (25) |
|
|
Med |
6 (5.45) |
9 (20.45) |
|
|
Large |
5 (4.55) |
17 (38.64) |
|
|
Infection present when stent inserted |
18 (16.36) |
13 (30.23) |
0.005 |
For continuous variables, the two-tailed Mann-Whitney U test was used to calculate p-values (upper part of table). For dichotomous or discrete variables, Chi-squared test of independence was used (lower part of table).
Table 1: Patient characteristics and investigation findings in patients with and without bacteriuria.
|
Indication for stent insertion |
n (%) |
Pathogens isolated |
n (%) |
|
Ureteric stricture or extrinsic compression |
61 (39.9) |
E. coli |
12 (27.9) |
|
Ureteric stone |
59 (38.6) |
Klebsiella pneumoniae |
6 (14) |
|
Renal stone |
16 (10.5) |
Proteus mirabilis |
5 (11.6) |
|
Diagnostic ureteropyeloscopy |
9 (5.9) |
Candida spp. |
5 (11.6) |
|
TUR resection of ureteric orifice |
7 (4.6) |
Mixed enteric |
5 (11.6) |
|
Ureteric tumor |
3 (2) |
E. faecalis |
3 (7) |
|
Ureteric clot |
1 (0.7) |
E. cloacae |
3 (7) |
|
Traumatic perinephric urinoma |
1 (0.7) |
P. aeruginosa |
2 (4.7) |
|
Major colorectal surgery |
1 (0.7) |
Morganella morganii |
2 (4.7) |
|
Serratia marcescens |
2 (4.7) |
||
|
Othera |
4 (9.3) |
aOne each of: Klebsiella aerogenes, achromobacter spp, stenotrophomas m, streptococcus agalactiae. Some urine specimens contained more than one pathogen.
Table 2: Indications for stent insertion and pathogens isolated.
Exploration of predictive value
The optimal LE activity cut point was calculated as “small” or greater (Youden index 0.406). The area under the ROC curve (AUC) was 0.82, (95% CI [0.74, 0.89], SE = 0.04, p < .001). This indicates that LE activity does significantly discriminate the presence of bacteriuria better than chance. The LE activity cut point “small” or greater was termed the “balanced” model. However, a NPV of 91% may not be considered robust enough in this context. To maximise negative predictive value of LE, a more restrictive cut point of “trace” or greater was also defined, titled the “conservative” model. The predictive values of urinary nitrite, and urinary LE activity at the two cut points are in Table 3. A predictive algorithm to exclude bacteriuria was designed using both nitrite and LE activity in combination. A combined negative nitrite and LE activity result was considered negative for bacteriuria, whereas any other combination of results was considered positive. The predictive values of the combined algorithm were determined for both the balanced and conservative LE activity cut points (Table 4). Adding nitrite results into a predictive algorithm did not improve the negative predictive value of LE activity and worsened the positive predictive value.
|
Nitrite |
LE Activity Balanced Modela |
LE Activity Conservative Modelb |
|
|
Percent (95% CI) |
Percent (95% CI) |
Percent (95% CI) |
|
|
Sensitivity |
18.60 (8.39-33.40) |
86.05 (72.07-94.70) |
100 (91.78-100) |
|
Specificity |
97.27 (92.24-99.43) |
54.55 (44.77-64.07) |
17.27 (10.73-25.65) |
|
Positive predictive value |
72.73 (42.59-90.55) |
42.53 (36.85-48.41) |
32.08 (30.25-33.97) |
|
Negative predictive value |
75.35 (72.54-77.97) |
90.91 (82.36-95.54) |
100 (82.35-100) |
|
Accuracy |
75.16 (67.54-81.79) |
63.40 (55.24-71.03) |
40.52 (32.67-48.75) |
aPositive LE cut point “small” or greater. bPositive LE cut point “trace” or greater
Table 3: Independent sensitivity, specificity and predictive values of urinary dipstick parameters for the presence of bacteriuria.
|
Nitrite plus balanced LE modela |
Nitrite plus conservative LE modelb |
|
|
Percent (95% CI) |
Percent (95% CI) |
|
|
Sensitivity |
86.5 (72.07-94.7) |
100 (91.78-100) |
|
Specificity |
52.73 (42.98-62.37) |
16.36 (10-24.62) |
|
Positive predictive value |
41.57 (36.08-47.27) |
31.85 (30.08-33.67) |
|
Negative predictive value |
90.63 (81.84-95.40) |
100 (81.47-100) |
|
Accuracy |
62.09 (53.90-69.80) |
39.87 (32.05-48.08) |
aPositive LE cut point “small” or greater. bPositive LE cut point “trace” or greater
Table 4: Sensitivity, specificity and predictive values of combined urinary nitrite and leukocyte esterase predictive algorithm for the presence of bacteriuria.
Discussion
Since urine MCS results are sometimes unavailable at the time of surgery, we assessed the utility of point of care urinary dipstick analysis parameters LE and nitrite for excluding bacteriuria in patients with indwelling ureteric stents. In contrast to the findings of Pooli et al (2016), a statistical correlation between urinary nitrite and bacteriuria was detected. However, the sensitivity (18.6%) in the study population was significantly lower than in the general unstented population (sensitivity approximately 54%) [15]. Decreased urinary dwell time due to stent irritation, a generally low median urinary pH of 5.5, and non-nitrate-reducing pathogens (present in 23.3% of positive culture results) may have accounted for the poor sensitivity of nitrite assay results in this cohort [8,9,12,17,18]. Whilst nitrite was highly specific for bacteriuria, the poor NPV results in an unacceptably high false negative rate of 25%, making nitrite a poor rule-out test in isolation. On the contrary, urinary LE activity was a reasonably reliable indicator of bacteriuria. The “balanced” model yielded a NPV of 91%. The poor specificity is due to the propensity for stents to induce pyuria even in the absence of infection, as demonstrated by the presence of leucocytes on direct microscopy for both sterile and infected urine. The “conservative” LE activity model yielded perfect negative predictive value (100%), at the expense of terrible specificity (17%), with a false-positive rate of 67%. The combined nitrite-LE algorithm yielded poorer overall predictive reliability when compared to LE alone, at both balanced and conservative cutoff points.
There was significant concordance between urinary LE and nitrite levels (Chi-squared association p=0.002), thus incorporating nitrite results did not improve upon the NPV of LE, and the high false-positive rate of nitrite worsened the PPV. In keeping with previous literature regarding ureteric stent infection [10,24,25], female gender, diabetes, and increasing age were all significantly associated with increased rates of bacteriuria. The presence of urinary infection at time of stent insertion was associated with subsequent bacteriuria – presumably due to immediate colonisation of stents inserted into a non-sterile urinary tract. Stent dwell time did not correspond to bacteriuria risk, in contrast to the findings of ssother authors [10,25]. Increased leucocyte count on microscopy was associated with bacteriuria, however was also a common feature in sterile urine specimens, due to the local inflammatory reaction caused by ureteric stents [19-21]. There was a statistically significant difference between the distribution of SG between the groups, despite the same median and IQR. Urinary SG may influence nitrite assay results. However, no significant correlation was detected between SG and nitrite, so it is unlikely that difference in SG distribution between groups was clinically meaningful. Given that several clinical parameters were found to significantly correlate with bacteriuria, it could be surmised that a predictive algorithm incorporating these parameters may further refine the predictive value of urinary dipstick assay results. Indeed, Weber and colleagues (2018) analysed the clinicopathological parameters of 101 positive urine cultures in 462 stented patients and generated both a multiple logistic regression and recursive portioning model to predict bacteriuria. Both models were similarly robust at excluding bacteriuria with a negative predictive value of 92%, with a reasonable false positive rate of 42% and 39% respectively [20].
The main drawback of this approach is the requirement to impute between four to six variables into a specific model function, making their use ponderous. Additionally, both models require serum C-reactive protein and urine leukocyte count results as input parameters, neither of which are readily available at the bedside. Thus, we chose to focus only on urinary LE and nitrite assay results as simple and immediate rule-out tests for bacteriuria, rather than generate a complex multivariate model, as this is reflective of their use in day-to-day clinical practice. Urinary LE activity appears to be the most reliable parameter for the exclusion of bacteriuria in stented patients in our study, with the performance of the “balanced” cut-point closely resembling that of the predictive models created by Weber et al, at 91% NPV. However, the simplicity of our single-parameter test carries the trade-off of a high false positive rate of 57%. Given that patients with pre-operative bacteriuria are at significantly higher risk of developing sepsis [26], and severe sepsis and shock may lead to a mortality rate of 30-40% [27], the diagnostic uncertainty of urinary LE in stented patients is too great to justify it superseding the routine collection of urine MCS pre-operatively. This is in accordance with other authors [19,20] and current guidelines [28]. Additionally, MCS results are required to direct appropriate prophylactic antibiotic therapy [29]. However, in cases where urine MCS results are unavailable, trace or absent LE activity on dipstick analysis may be used to support a decision to proceed with surgery, especially in the presence of other favourable patient characteristics, such as male gender, absence of diabetes, negative nitrite, and absence of infection at time of initial stent insertion. For even higher certainty, the conservative LE cut point may be used instead, where any level of LE activity is considered positive for bacteriuria. While the NPV of this cut point is perfect at 100%, it classifies 86% of samples as positive, with a false positive rate of 68%, meaning most patients are misclassified. However, this conservative approach may be suitable in higher risk patients, in whom surgery should otherwise definitely be postponed if urine MCS results are unavailable. There are several limitations to this our study that must be considered. The data are subject to the inherit weaknesses of retrospective collection in a single centre and external validation would improve its reliability. There was no study-specific standardized methodology for collection of the urine samples or dipstick testing. A high rate of MCS contamination weakened the power of the study. Symptoms of UTI were not captured, and this data may have had diagnostic value. Other authors have demonstrated discordance between urinary and stent cultures, with positive stent culture associated with infectious complications following ureteroscopy [22]. Stent cultures were not performed, thus MCS results may not reflex true urinary tract colonization rates. LE levels were based on the Clinitek Status+ Analyzer outputs and may not correlate to results from alternative urinalysis machines. Post-operative complications were not captured, meaning that urine dipstick parameters could not be directly compared to complication rates - the key outcome of concern. Nonetheless, we have demonstrated that urinary LE activity can exclude bacteriuria in stented patients with high reliability. This is in contrast to previous studies which have either demonstrated poor correlation of dipstick results to culture [19], or required multiple parameters, including blood tests, to power a predictive model [20].
Conclusion
Bacteriuria in the setting of ureteric stents is common and requires exclusion prior to elective endourological surgery. Unavailable urine MCS results on the day of surgery may result in unnecessary cancellations. Urinary dipstick LE activity can quickly and easily exclude bacteraemia with reasonable reliability. However, given the high risk of infective complications following ureteroscopy and the high false positive rate of urine LE activity in stented patients, urine dipstick analysis should not be routinely substituted for standard pre-operative MCS, and clinicians should strive to ensure culture results are always available on the day of surgery.
Ethical Considerations: This project was reviewed and approved by the local Ethics Committee (EX/2024/QCH/105702) and was completed in accordance with the Declaration of Helsinki. A waiver to informed consent was granted on the grounds of negligible risk research. All relevant privacy regulations were followed.
Conflicts Of Interest: The authors have no conflicts of interest to disclose
Funding: No funding was provided for this research
Acknowledgements: none
Authorship Contribution: Dr Matthew Wright-Smith: investigation, formal analysis, writing – original draft, writing – review and editing. Dr Simon Pridgeon: conceptualization, methodology, supervision, writing – review and editing.
Data Sharing: Data sharing was not approved by the Far North Queensland Human Research Ethics Committee
References
- Nicolle LE, Bradley S, Colgan R, (2005) Infectious Diseases Society of America Guidelines for the Diagnosis and Treatment of Asymptomatic Bacteriuria in Adults. Clinical Infectious Diseases 40: 643-654.
- Grabe M, Forsgren A, Hellsten S (1984) The Effect of a Short Antibiotic Course in Transurethral Prostatic Resection. Scandinavian Journal of Urology and Nephrology 18: 37-42.
- Grabe M, Forsgren A, Björk T (1987) Controlled trial of a short and a prolonged course with ciprofloxacin in patients undergoing transurethral prostatic surgery. Eur J, Clin Microbiol 6: 11-17.
- Murphy DM, Falkiner FR, Carr M (1983) Septicemia after transurethral prostatectomy. Urology 22: 133-135.
- Grabe M (1987) Antimicrobial Agents in Transurethral Prostatic Resection. The Journal of Urology 138: 245-252.
- Schmiemann G, Kniehl E, Gebhardt K (2010) The Diagnosis of Urinary Tract Infection. Dtsch Arztebl Int 107: 361-367.
- Bonkat G, Bartoletti R, Bruyere F (EAU Guidelines on Urological Infections [Internet]. Uroweb - European Association of Urology.
- Joshi HB, Stainthorpe A, MacDONAGH RP ( 2003) Indwelling Ureteral Stents: Evaluation of Symptoms, Quality of Life and Utility. Journal of Urology 169: 1065-1069.
- Lee C, Kuskowski M, Premoli J (2005) Randomized Evaluation of Ureteral Stents Using Validated Symptom Questionnaire. Journal of Endourology 19: 990-993.
- Farsi HM, Mosli HA, Al-Zemaity MF (1995) Bacteriuria and colonization of double-pigtail ureteral stents: long-term experience with 237 patients. J Endourol 9: 469-472.
- Aydin HR, Irkilata L, Aydin M (2016) Incidence of bacterial colonisation after indwelling of double-J ureteral stent. Arch Ital Urol Androl 87: 291-294.
- Mambatta AK, Jayarajan J, Rashme VL (2015) Reliability of dipstick assay in predicting urinary tract infection. J Family Med Prim Care 4: 265-268.
- Ohly N, Teece S (2003) Accuracy of negative dipstick urine analysis in ruling out urinary tract infection in adults. Emerg Med J 20: 362-363.
- Devillé WLJM, Yzermans JC, van Duijn NP (2004) The urine dipstick test useful to rule out infections. A meta-analysis of the accuracy. BMC Urol 4: 4.
- St John A, Boyd JC, Lowes AJ (2006) The use of urinary dipstick tests to exclude urinary tract infection: a systematic review of the literature. Am J Clin Pathol 126: 428-436.
- Jarvis TR, Chan L, Gottlieb T (2014) Assessment and management of lower urinary tract infection in adults. Australian Prescriber 37: 7-9
- Simerville JA, Maxted WC, Pahira JJ (2005) Urinalysis: A Comprehensive Review. Afp 71: 1153-1162.
- Andriole VT (1987) Urinary Tract Infections: Recent Developments. The Journal of Infectious Diseases 156: 865-869.
- Pooli A, Cook G, Isharwal S (2016) Urinalysis findings are not predictive of positive urine culture in patients with indwelling stents. Can J Urol 23: 8446-8450.
- Weber A, Zumstein V, Betschart P (2018) Prediction of Bacteriuria Based on Clinical or Laboratory Parameters in Patients with Indwelling Ureteral Stents Before Ureterorenoscopy Should Not Substitute for Urine Cultures. J Endourol 32: 739-745.
- Lau CT, Stavropoulos SW (2011) Chapter 35 - Genitourinary and Gastrointestinal Interventional Radiology. In: Pretorius ES, Solomon JA, editors. Radiology Secrets Plus (Third Edition) [Internet]. Philadelphia: p. 251-256.
- Nevo A, Mano R, Schreter E (2017) Clinical Implications of Stent Culture in Patients with Indwelling Ureteral Stents Prior to Ureteroscopy. The Journal of Urology 198: 116-121.
- Schoonjans F (2020) MedCalc’s Diagnostic test evaluation.
- Akay AF, Aflay U, Gedik A (2007) Risk factors for lower urinary tract infection and bacterial stent colonization in patients with a double J ureteral stent. Int Urol Nephrol 39: 95-98.
- Kehinde EO, Rotimi VO, Al-Awadi KA (2002) Factors predisposing to urinary tract infection after J ureteral stent insertion. J Urol 167: 1334-1337.
- Blackmur JP, Maitra NU, Marri RR (2026) Analysis of Factors’ Association with Risk of Postoperative Urosepsis in Patients Undergoing Ureteroscopy for Treatment of Stone Disease. J Endourol 30: 963-969.
- Wagenlehner FME, Lichtenstern C, Rolfes C (2013) Diagnosis and management for urosepsis. Int J Urol 20: 963-970.
- Lightner DJ, Wymer K, Sanchez J (2020) Best Practice Statement on Urologic Procedures and Antimicrobial Prophylaxis. J Urol 203: 351-356.
- Cai T, Verze P, Palmieri A (2017) Is Preoperative Assessment and Treatment of Asymptomatic Bacteriuria Necessary for Reducing the Risk of Postoperative Symptomatic Urinary Tract Infections After Urologic Surgical Procedures? Urology 99: 100-105.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.