Secondary Hyperparathyroidism in Dialysis Patients: Treatment Patterns and Parathyroidectomy Decisions Francesca Bacchetta
by Cattori1, Rafael Freire dos Santos2, Kerstin Hübel3, Marco Bonani3, Elena Cadamuro4, Domenico Cozza4, Pietro Cippà5, Antonio Bellasi4, Diana Vetter2*
1Department of Anesthesiology, Cantonal Hospital Winterthur, Winterthur, Switzerland
2Department of Abdominal and Transplantation Surgery, University Hospital Zurich (USZ), Zurich, Switzerland
3Department of Nephrology, University Hospital Zurich (UZH), Zurich, Switzerland
4Department of Nephrology, EOC Lugano, Lugano, Switzerland
5Department of Nephrology, University Hospital Basel, Switzerland
Received Date: 21 April 2026
Accepted Date: 27 April 2026
Published Date: 29 April 2026
Citation: Bacchetta-Cattori F, Santos RFD, Hübel K, Bonani M, Bellasi A, et al. (2026) Secondary Hyperparathyroidism in Dialysis Patients: Treatment Patterns and Parathyroidectomy Decisions. J Surg 11: 11619 DOI: https://doi.org/10.29011/2575-9760.011619
Abstract
Background: Secondary Hyperparathyroidism (sHPT) is a near-universal complication of end-stage renal disease, contributing significantly to Chronic Kidney Disease-Mineral And Bone Disorder (CKD-MBD). While medical therapy is the first-line treatment, a substantial proportion of patients develop refractory disease. Parathyroidectomy (PTx) is recommended for medically refractory sHPT, yet its real-world utilization remains poorly characterized.
Methods: We conducted a retrospective cohort study of 177 dialysis patients treated at two tertiary centers: the University Hospital Zurich (USZ, n=60) and the Ente Ospedaliero Cantonale (EOC, n=117). We analyzed the prevalence of sHPT, patterns of medical therapy (vitamin D analogues, phosphate binders, calcimimetics), rates of treatment success, and the clinical decision-making process regarding PTx in patients with treatment failure.
Results: sHPT was present in 93.8% (166/177) of the cohort. Medical therapy was administered to 81.4% (144/177) of patients, with vitamin D analogues being the most frequently prescribed agent (77.4%), followed by phosphate binders (49.2%) and calcimimetics (18.1%). Medical management was successful in 62.7% (111/177) of cases, while 35.0% (62/177) experienced treatment failure. The primary reasons for treatment failure were persistently elevated parathyroid hormone (PTH) > 800 pg/mL (85.7%) and hyperphosphatemia (82.1%). Among the 62 patients with refractory sHPT, only 1 patient (1.6%) underwent PTx specifically for medication failure. In the remaining patients, the primary documented reason for withholding surgery was physician judgment that PTx was “not indicated” (59.6%, 34/57), rather than patient refusal (1.8%) or surgical unfitness (7.0%).
Conclusions: Despite a high rate of medical treatment failure in sHPT, surgical referral for PTx is strikingly underutilized. The predominance of physician-driven barriers, particularly the reluctance to consider elevated phosphate as an indication for surgery, suggests a significant gap between current clinical guidelines and real-world practice. This highlights the need for structured multidisciplinary referral pathways.
Citation: Bacchetta-Cattori F, Santos RFD, Hübel K, Bonani M, Bellasi A, et al. (2026) Secondary Hyperparathyroidism in Dialysis Patients: Treatment Patterns and Parathyroidectomy Decisions. J Surg 11: 11619 DOI: 10.29011/2575-9760.011619
Keywords: Calcimimetics; Chronic Kidney Disease–Mineral And Bone Disorder; Dialysis; Parathyroidectomy; Secondary Hyperparathyroidism
Introduction
Secondary Hyperparathyroidism (sHPT) is a hallmark complication of End-Stage Renal Disease (ESRD) and a central component of Chronic Kidney Disease-Mineral And Bone Disorder (CKD-MBD) [1]. The condition is driven by a complex interplay of phosphate retention, reduced synthesis of active vitamin D (calcitriol), and subsequent hypocalcemia, which collectively stimulate the parathyroid glands to overproduce Parathyroid Hormone (PTH) in an attempt to normalize serum calcium. Secondary HPT is potentially reversible when the chronic kidney disease is addressed by kidney transplantation. However, prolonged sHPT leads to severe clinical consequences, including renal osteodystrophy [2], an increased risk of bone fractures, intractable pruritus, and accelerated vascular and valvular calcification, which independently drives cardiovascular morbidity and mortality in the dialysis population [3-5]. Furthermore, the presence of secondary hyperparathyroidism has been shown to decrease kidney graft survival and increase all-cause mortality following renal transplantation [6-8], whereas performing parathyroidectomy (PTx) prior to transplantation is protective and decreases the risk of future graft failure [9]. The management of sHPT primarily relies on pharmacological therapies. The standard armamentarium includes dietary phosphate restriction combined with phosphate binders, active vitamin D analogues to suppress PTH synthesis, and calcimimetics, which competes with calcium at the calciumsensing receptor on the parathyroid gland, resulting in reduced PTH secretion [10]. While these medical therapies are effective for many patients, a significant subset develops progressive sHPT, characterized by autonomous nodular hyperplasia of the parathyroid glands that no longer responds to pharmacological suppression. For patients with medically refractory sHPT, clinical guidelines— including those from Kidney Disease: Improving Global Outcomes (KDIGO) [11], the KDOQI US Commentary [12], and the German S2k guidelines on operative therapy of renal hyperparathyroidism [13]—recommend surgical PTx. PTx has been shown to rapidly correct biochemical abnormalities, significantly reduce the risk of fractures [14], and confer a substantial survival benefit compared to continued medical therapy in refractory cases [15-17]. However, anecdotal evidence and registry data suggest that PTx rates have declined globally since the introduction of calcimimetics [18, 19], raising concerns about the potential underutilization of surgery. The real-world decision-making process leading to referral or non-referral for PTx remains poorly understood. In particular, the relative contribution of physician-, patient-, and system-level factors has not been systematically evaluated. This study aimed to evaluate the real-world prevalence of sHPT, the patterns and efficacy of medical therapy, and the clinical decision-making process regarding PTx in a contemporary multicenter cohort of dialysis patients.
Material and Methods
Study Design and Population
We conducted a multicenter retrospective cohort study of 177 dialysis patients between 2019 and 2020 treated at two tertiary centers: the University Hospital Zurich (USZ, n=60) and the Ente Ospedaliero Cantonale (EOC, n=117) (Figure 1). The cohort included adult patients undergoing maintenance dialysis for ESRD. This study was approved by the ethical boards of the Canton Zurich and the Canton Ticino.
Data Collection and Variables
Data were extracted from electronic medical records and compiled into a standardized database. The primary variables of interest included:
- sHPT Diagnosis: The presence or absence of clinically diagnosed sHPT, defined as PTH above normal range.
- Medical Therapy: The prescription of specific pharmacological agents for sHPT, categorized into vitamin D analogues, phosphate binders, and calcimimetics.
- Treatment Outcome: The clinical failure of medical therapy, defined according to the KDIGO guidelines 2006 if PTH > 800 pg/mL (> 9x upper boundary range), hyperphosphatemia and/or hypercalcemia are present twice over a time period of more than 6 months, or if there is an increase in symptoms of secondary hyperparathyroidism such as bone pain, bone fractures, or calciphylaxis.
- Surgical Decision-Making: For patients identified as having medical treatment failure, we analyzed whether PTx was performed. If PTx was not performed, the clinical rationale was assessed from the treating nephrologist (e.g., patient refusal, surgical unfitness, physician judgment of non-indication).
Statistical Analysis
Descriptive statistics were used to summarize the cohort characteristics. Categorical variables were expressed as frequencies and percentages. The analysis focused on the cascade of care, from initial diagnosis through medical treatment to surgical referral. Differences between the two study centers were also evaluated.
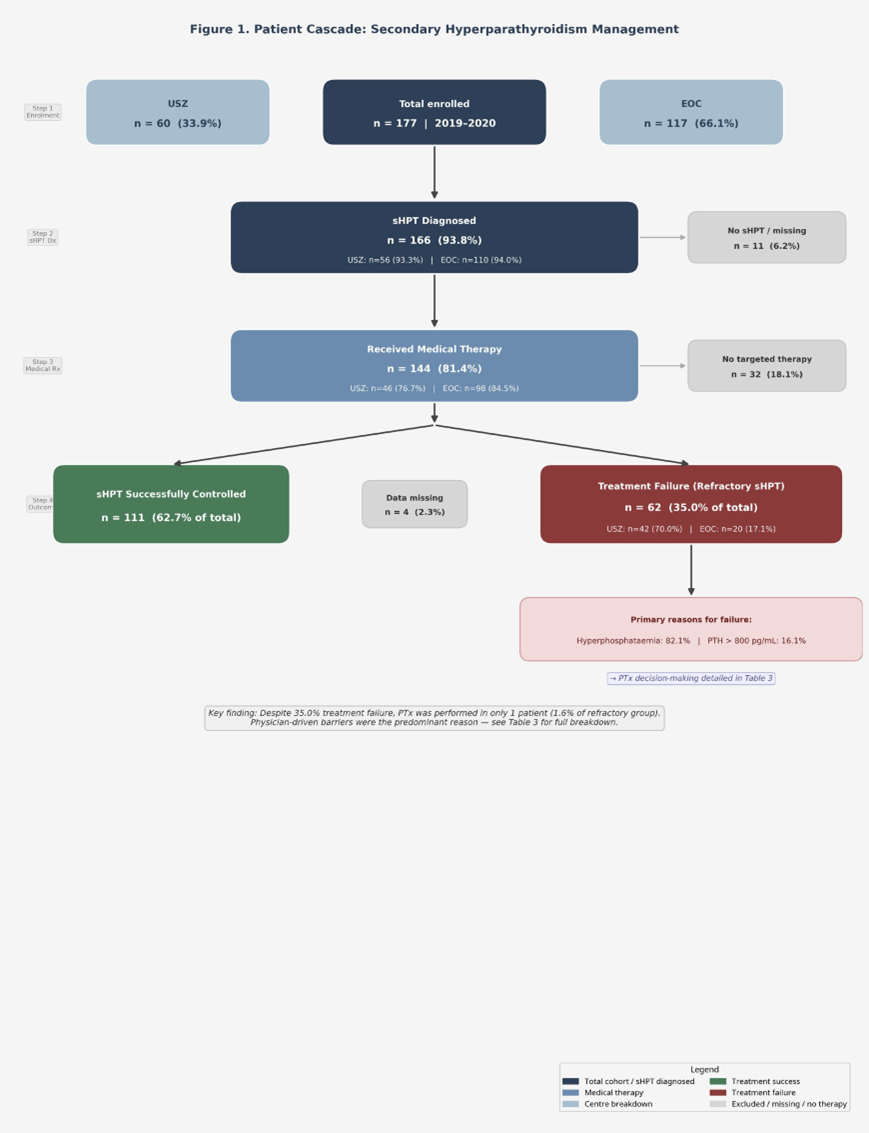
Figure 1: Patient Cascade: Secondary Hyperparathyroidism Management.
Results
Patient Characteristics
A total of 177 patients were included in the study, of whom 60 were enrolled at the University Hospital Zurich (USZ) and 117 at the Ente Ospedaliero Cantonale (EOC). The median age of the cohort was 77.2 years (IQR 68.6–82.4), with a female predominance (69.5%). The majority of patients were on haemodialysis (92.7%), with a median dialysis duration of 63.0 months (IQR 34.0–83.5). A history of prior kidney transplantation was documented in 54 patients (30.5%), and late referral to nephrology care was recorded in 15 patients (12.6% of those with available data). Full patient characteristics are presented in Table 1. Several clinically relevant differences between the two centers were identified.
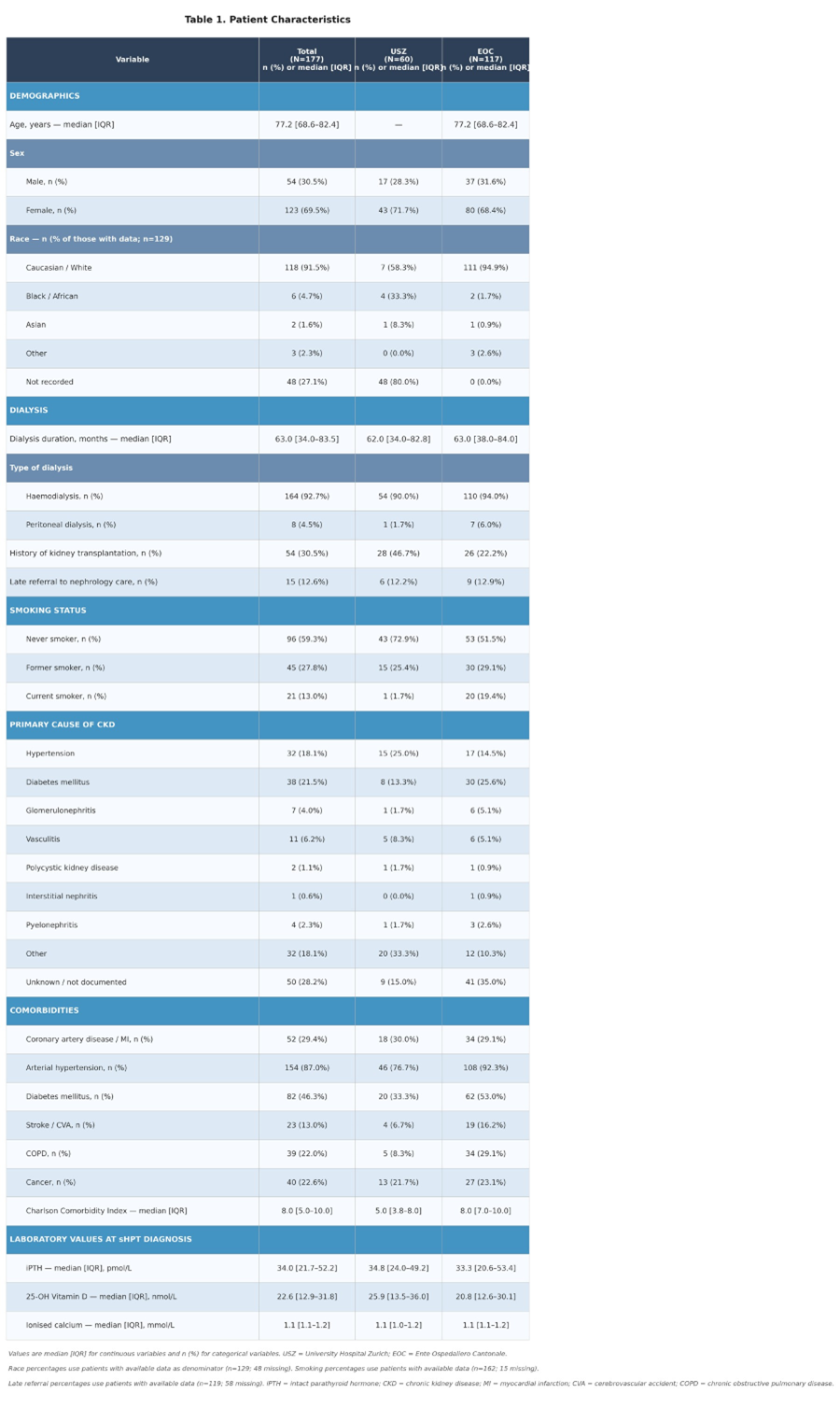
Table 1: Patient Characteristics.
The prevalence of diabetes mellitus was higher at EOC (53.0%) than at USZ (33.3%), as was the prevalence of arterial hypertension (92.3% vs. 76.7%), COPD (29.1% vs. 8.3%), and stroke (16.2% vs. 6.7%). Accordingly, the median Charlson Comorbidity Index was higher at EOC (8.0 [IQR 7.0–10.0]) than at USZ (5.0 [IQR 3.8–8.0]), suggesting a greater overall comorbidity burden in the EOC cohort. Regarding the primary cause of chronic kidney disease, hypertension (25.0% vs. 14.5%) and diabetes mellitus (13.3% vs. 25.6%) were distributed differently between centers, with a higher proportion of unknown or undocumented aetiology at EOC (35.0% vs. 15.0% at USZ). Smoking status also differed markedly: current smoking was substantially more prevalent at EOC (19.4%) than at USZ (1.7%), while USZ had a higher proportion of never-smokers (72.9% vs. 51.5%). Race data were largely unavailable at USZ (80.0% missing), precluding meaningful inter-center comparison of ethnicity. Baseline laboratory values at the time of sHPT diagnosis showed comparable 25-OH vitamin D levels (USZ: 25.9 nmol/L [IQR 13.5–36.0]; EOC: 20.8 nmol/L [IQR 12.6–30.1]) and ionised calcium (USZ: 1.1 mmol/L [IQR 1.0–1.2]; EOC: 1.1 mmol/L [IQR 1.1–1.2]) between centers. Baseline iPTH levels were comparable between the two centers (USZ: 34.8 pmol/L [IQR 24.0–49.2]; EOC: 33.3 pmol/L [IQR 20.6–53.4]).
Prevalence of sHPT
Secondary hyperparathyroidism was nearly universal in this dialysis cohort. Of the 177 patients evaluated, 166 (93.8%) had a confirmed diagnosis of sHPT. Only 8 patients (4.5%) did not have sHPT, and data were missing for 3 patients (1.7%) (Figure 1).
Patterns of Medical Therapy
The majority of the cohort received pharmacological treatment for sHPT. Medical therapy was prescribed to 144 patients (81.4%), while 32 patients (18.1%) received no targeted sHPT medication (Table 1). The overall rate of medical therapy was similar between the two centers, with 76.7% (46/60) of patients at USZ and 84.5% (98/116) of patients at EOC receiving pharmacological treatment (Table 2). The utilization of specific drug classes varied significantly, though the patterns were largely consistent across both centers (Table 2):
- Vitamin D analogues were the most frequently prescribed agents, utilized by 137 patients (77.4% of the total cohort). This included 76.7% of patients at USZ and 79.1% at EOC.
- Phosphate binders were prescribed to 87 patients (49.2%). Utilization was slightly higher at USZ (53.3%) compared to EOC (47.4%).
- Calcimimetics were the least utilized class, prescribed to only 32 patients (18.1%). Prescription rates were comparable between centers (20.0% at USZ vs. 17.4% at EOC).
- Combination therapy was common, reflecting the standard stepwise approach to managing CKD-MBD.
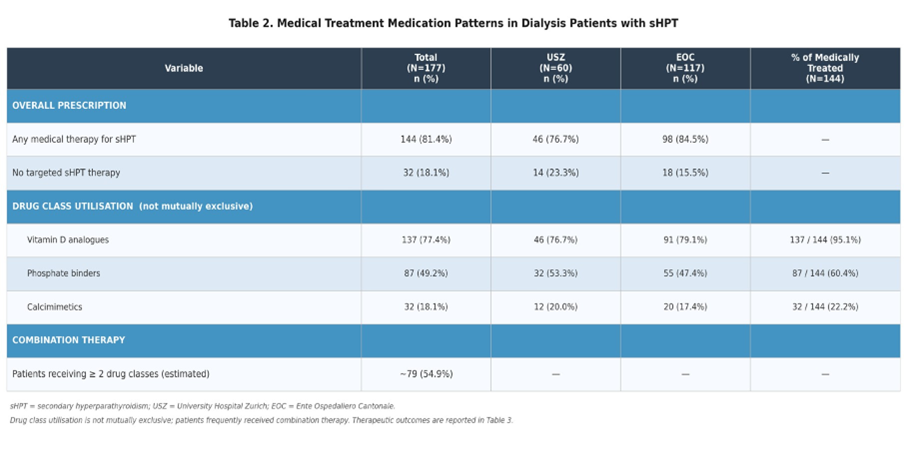
Table 2: Medical Treatment Medication Patterns in Dialysis Patients with Shpt.
Efficacy of Medical Management
Medical therapy achieved successful control of sHPT in 111 patients, representing 62.7% of the total cohort. However, a substantial proportion of patients—62 individuals (35.0%)—experienced treatment failure, defined as refractory sHPT despite medical intervention. Outcome data were missing for 4 patients (2.3%) (Table 3).
The primary reasons for treatment failure among these 62 patients were hyperphosphatemia (46/56 with available data, 82.1%) and persistently elevated PTH > 800pg/mL (9/56, 16.1%). Clinical symptoms such as bone pain (4/56, 7.1%) and bone fractures (1/56, 1.8%) were less frequently documented as the primary cause of failure (Table 3). Treatment failure rates differed significantly between the two centers. At USZ, 70.0% (42/60) of patients experienced treatment failure, compared to only 17.1% (20/117) at EOC (Table 3).
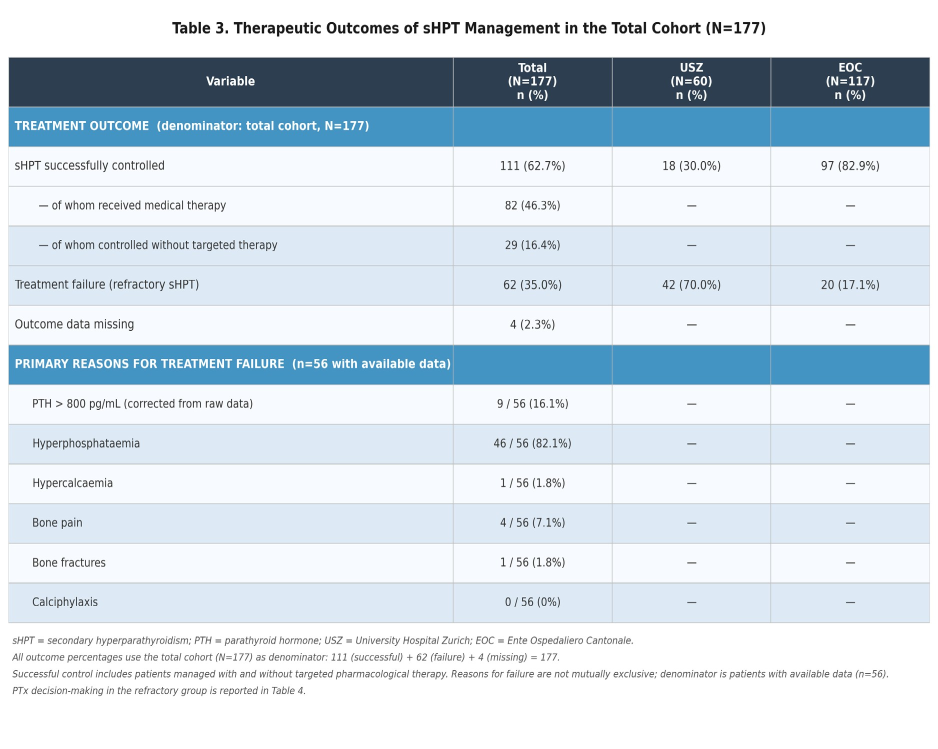
Table 3: Therapeutic Outcomes of sHPT Management in the Total Cohort (N=177).
Parathyroidectomy Decision-Making in Refractory sHPT
The 62 patients with medical treatment failure represented the primary population with a potential indication for PTx. Within this refractory group, surgical intervention was exceedingly rare. Only 1 patient (1.6% of the refractory group) underwent PTx specifically due to medication failure. For the remaining patients who did not undergo surgery, a documented reason for withholding PTx was available for 57 patients (91.9%), while 4 patients (6.5%) had no documented rationale. An analysis of the documented reasons for withholding PTx revealed that physician-driven decisions were the primary barrier to surgical referral. The reasons were distributed as depicted in Table 4. Notably, no cases were withheld due to a lack of access to a trusted surgeon, the option not being considered, or concerns regarding excessive surgical complications. Significant differences in decision-making were observed between the two centers. At USZ, the overwhelming majority of withheld surgeries (33/38, 86.8%) were due to the physician deeming PTx “not indicated”. In contrast, at EOC, this reason was cited in only 1 of 19 cases (5.3%), with “other reasons” being the most common justification (14/19, 73.7%) (Table 4).
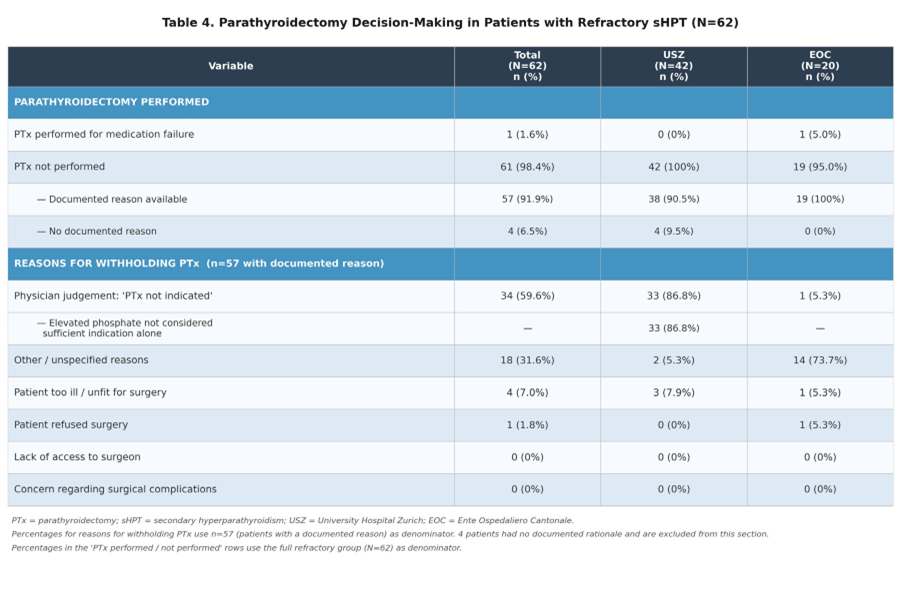
Discussion
This multicenter retrospective cohort study provides critical insights into the real-world management of sHPT in dialysis patients. Our findings confirm that sHPT remains a near-universal complication in this population (93.8% prevalence) [20]. While medical therapy is widely deployed and successful in approximately two-thirds of patients, a significant 35% of the cohort developed refractory disease. The most striking finding of this analysis is the profound underutilization of parathyroidectomy in this refractory group, driven primarily by physician judgment rather than patientrelated factors. The pharmacological management observed in our cohort aligns with historical prescribing patterns, heavily favoring vitamin D analogues (77.4%) and phosphate binders (49.2%). The relatively low utilization of calcimimetics (18.1%) is noteworthy. This may reflect local prescribing preferences, cost constraints, or tolerability issues (such as gastrointestinal side effects or hypocalcemia) that frequently limit the titration of calcimimetics in clinical practice. Overall prescribing rates and drug class preferences were remarkably similar between the two study centers, indicating a shared approach to initial medical management.
Despite the availability of these medical therapies, 62 patients experienced treatment failure, primarily driven by persistently elevated PTH and hyperphosphatemia. According to established clinical guidelines, including KDIGO [11] and the German S2k guidelines [13], medically refractory sHPT accompanied by hypercalcemia, hyperphosphatemia, or severe clinical symptoms is a Class 1 indication for surgical parathyroidectomy. However, in our cohort, only 1 of these 62 patients underwent PTx for this indication.
When examining the barriers to surgery, patient-related factors— such as surgical unfitness (7.0%) or outright refusal of the procedure (1.8%)—accounted for only a small minority of cases. Instead, the dominant reason for withholding surgery was the treating physician’s assessment that PTx was “not indicated” (59.6%). This finding suggests a significant divergence between guideline recommendations and real-world clinical thresholds for surgical referral. This divergence was particularly pronounced at the University Hospital Zurich (USZ), where 86.8% of withheld surgeries were attributed to the physician deeming PTx “not indicated”. For USZ, the main reason was that the nephrologists did not consider elevated phosphate as a sufficient reason for parathyroidectomy. However, according to both international and national guidelines, while hyperphosphatemia alone is not an indication for surgery, the combination of severe, medically refractory sHPT (persistently elevated PTH) accompanied by hyperphosphatemia or hypercalcemia constitutes a clear indication for parathyroidectomy. Several factors may contribute to this physician-driven barrier. Nephrologists may exhibit “clinical inertia,” preferring to exhaust all pharmacological combinations or tolerate sub-optimal biochemical control rather than refer a patient for surgery. Furthermore, 35% of patients undergoing calcimimetic therapy discontinue treatment within one year [21]. Also for this reason, PTx could become a relevant and important treatment option. There may also be an underappreciation of the long-term cardiovascular and skeletal benefits of PTx compared to the perceived immediate perioperative risks, even though modern parathyroid surgery in ESRD patients carries a low morbidity profile when performed by experienced endocrine surgeons [15]. The clinical implications of these findings are substantial. Patients with refractory sHPT who are denied definitive surgical therapy remain at high risk for progressive vascular calcification, fragility fractures, and cardiovascular mortality [14]. The data strongly advocate for the implementation of structured, multidisciplinary CKD-MBD pathways. Routine case discussions involving nephrologists and endocrine surgeons could help standardize the definition of “medically refractory” disease and ensure that eligible patients are offered PTx in a timely manner.
Limitations
This study is limited by its retrospective design, the inter-center and cohort heterogeneity, the lack of standardized PTx indication criteria, and the reliance on documented clinical rationales, which may not fully capture the nuance of shared decision-making between the physician and patient.
Conclusions
Secondary hyperparathyroidism remains highly prevalent in the dialysis population, with over one-third of patients failing medical management. Despite this high rate of refractory disease, parathyroidectomy is rarely performed. The primary barrier to surgical intervention appears to be physician judgment— specifically the reluctance to consider elevated phosphate as an indication for surgery—rather than patient frailty or refusal. Bridging the gap between clinical guidelines and real-world practice through structured surgical referral pathways is essential to improve the long-term outcomes of dialysis patients with severe sHPT.
References
- Ketteler M, Block GA, Evenepoel P (2017) Executive summary of the 2017 KDIGO Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD) Guideline Update: what’s changed and why it matters. Kidney Int 92: 26-36.
- Damasiewicz MJ, Nickolas TL (2018) Rethinking Bone Disease in Kidney Disease. JBMR Plus 2: 309-322.
- De Boer IH, Gorodetskaya I, Young B (2002) The severity of secondary hyperparathyroidism in chronic renal insufficiency is GFR-dependent, race-dependent, and associated with cardiovascular disease. J Am Soc Nephrol 13: 2762-2769.
- Block GA, Klassen PS, Lazarus JM (2004) Mineral metabolism, mortality, and morbidity in maintenance hemodialysis. J Am Soc Nephrol 15: 2208-2218.
- Ganesh SK, Stack AG, Levin NW (2001) Association of elevated serum PO(4), Ca x PO(4) product, and parathyroid hormone with cardiac mortality risk in chronic hemodialysis patients. J Am Soc Nephrol 12: 2131-2138.
- Pihlstrøm H, Dahle DO, Mjøen G (2015) Increased risk of all-cause mortality and renal graft loss in stable renal transplant recipients with hyperparathyroidism. Transplantation 99: 351-359.
- Heaf J, Tvedegaard E, Kanstrup IL (2003) Hyperparathyroidism and long-term bone loss after renal transplantation. Clin Transplant 17: 268-274.
- Perrin P, Caillard S, Javier RM (2013) Persistent hyperparathyroidism is a major risk factor for fractures in the five years after kidney transplantation. Am J Transplant 13: 2653-2663.
- Callender GG, Malinowski J, Javid M (2017) Parathyroidectomy prior to kidney transplant decreases graft failure. Surgery 161: 44-50.
- Tsai SH, Kan WC, Jhen RN (2024) Secondary hyperparathyroidism in chronic kidney disease: A narrative review focus on therapeutic strategy. Clin Med 24: 100238.
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group (2017) KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl 7: 1-59.
- Isakova T, Nickolas TL, Denburg M (2017) KDOQI US Commentary on the 2017 KDIGO Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney DiseaseMineral and Bone Disorder (CKD-MBD). Am J Kidney Dis 70: 737-751.
- Weber T, Dotzenrath C, Dralle H, (2021) Management of primary and renal hyperparathyroidism: guidelines from the German Association of Endocrine Surgeons (CAEK). Langenbecks Arch Surg 406: 571-585.
- Rudser KD, de Boer IH, Dooley A (2007) Fracture risk after parathyroidectomy among chronic hemodialysis patients. J Am Soc Nephrol 18: 2401-2407.
- Komaba H, Taniguchi M, Wada A (2015) Parathyroidectomy and survival among Japanese hemodialysis patients with secondary hyperparathyroidism. Kidney Int 88: 350-359.
- Sharma J, Raggi P, Kutner N (2012) Improved long-term survival of dialysis patients after near-total parathyroidectomy. J Am Coll Surg 214: 400-407.
- Kestenbaum B, Andress DL, Schwartz SM (2004) Survival following parathyroidectomy among United States dialysis patients. Kidney Int 66: 2010-2016.
- Kim SM, Shu AD, Long J (2016) Declining Rates of Inpatient Parathyroidectomy for Primary Hyperparathyroidism in the US. PLoS One 11: e0161192.
- Lou I, Schneider DF, Leverson G (2016) Parathyroidectomy is underused in patients with tertiary hyperparathyroidism after renal transplantation. Surgery 159: 172-179.
- Wang Y, Liu J, Fang Y (2024) Estimating the global prevalence of secondary hyperparathyroidism in patients with chronic kidney disease. Front Endocrinol 15: 1400891.
- Fuller DS, Hallett D, Dluzniewski PJ (2019) Predictors of cinacalcet discontinuation and reinitiation in hemodialysis patients: results from 7 European countries. BMC Nephrol 20: 169.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.