Rhythm Control vs. Rate Control in Atrial Fibrillation: A Systematic Review and Meta-Analysis of Randomized Controlled Trials, With Supplemental Observational Data
by Akshay Maharaj1*, Sajay Bidhesi2, Shabnam Mahjabeen3, Musarrat F Dola4, Nowrin Tamanna5, Saniya Mohsin6, Aaron Lutchman7, Sapna Chandran8, Mohammad M Husain9, Jarin Tasnim10, Theana Guerra11, Hersh M Tilokani12
1Internal Medicine, Port of Spain General Hospital · Chaguanas, TTO
2Internal Medicine, San Fernando General Hosptial · Couva, TTO
3Family Medicine ,Shaheed Suhrawardy Medical College and Hospital, Dhaka · Dhaka, BGD
4Internal Medicine ,Ibrahim Medical College · Dhaka, BGD
5Internal Medicine,Chittagong Medical College · Chattogram, BGD and Epidemiology, University of South Carolina · Columbia, USA
6Medical College, Baqai Medical University · Karachi, PAK
7Anaesthesia and Intensive Care ,Eric Williams Medical Sciences Complex · Port of Spain, TTO
8Pediatrics, Government Medical College, Thiruvananthapuram · Thiruvananthapuram, IND
9Internal Medicine,Gulf Coast Medical Center · Fort Myers , USA
10Medicine, M.A.G Osmani Medical college · sylhet, BGD
11Internal Medicine/Family Medicine,North Central Regional Health Authority · Couva, TTO
12Anaesthesiology ,Rocky Vista University College of Osteopathic Medicine · Parker, USA
*Corresponding author: Akshay Maharaj, Internal Medicine, Port of Spain General Hospital · Chaguanas, TTO.
Received Date: 01 April, 2026
Accepted Date: 14 April, 2026
Published Date: 20 April, 2026
Citation: Maharaj A, Bidhesi S, Mahjabeen S, Dola MF, Tamanna N, et al. (2026) Rhythm Control vs. Rate Control in Atrial Fibrillation: A Systematic Review and Meta-Analysis of Randomized Controlled Trials, With Supplemental Observational Data. Cardiol Res Cardio vasc Med 11:297. DOI: https://doi.org/10.29011/2575-7083.100297
Abstract
Background: Atrial fibrillation (AF) increases the risks of stroke, heart failure, and mortality. Treatment involves rate control (atrioventricular nodal blockers) or rhythm control (antiarrhythmics, cardioversion, or ablation). Early trials showed neutral survival but higher adverse events with rhythm control, favoring rate control initially. Recent advances (e.g., EAST-AFNET 4 (Early Treatment of Atrial Fibrillation for Stroke Prevention Trial) suggest potential benefits with early rhythm control, prompting this updated synthesis. Methods: Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)-guided systematic review and meta-analysis of randomized controlled trials (RCTs) comparing rhythm- vs. rate-control in adults with AF (≥3 months follow-up, reporting heart failure hospitalization, cardiovascular death, all-cause mortality, or stroke). Primary pooled estimates used random-effects models (risk ratios (RR) with 95% CIs). One retrospective observational extension was included supplementally (secondary analyses only). Heterogeneity (I²) and small-study effects (Egger’s test, funnel plots) were assessed; sensitivities explored subgroups and robustness. Results: Nineteen RCTs (≈34,000 participants, balanced arms) were included. Rhythm control showed no significant differences vs. rate control in cardiovascular mortality (RR 0.99, 95% CI 0.64-1.54), all-cause mortality (RR 1.01, 95% CI 0.68-1.50), or stroke (RR 1.00, 95% CI 0.57-1.74). Heart failure hospitalization showed a nonsignificant trend (RR 0.84, 95% CI 0.47-1.49). Heterogeneity was moderate to high (I² 65-79%), partly explained by study era, rhythm method (ablation-inclusive vs. drug-only), follow-up variation, and endpoint definitions. Egger’s p < 0.05 in some outcomes (e.g., heart failure hospitalization) likely reflects heterogeneity/small-study effects rather than publication bias (symmetric funnels and stable sensitivities). An exploratory subgroup (beta-blocker/non-dihydropyridine calcium channel blocker (CCB) rate control, two studies) suggested lower heart failure hospitalization (RR 0.54, 95% CI 0.38-0.76), but this is hypothesis-generating only (limited studies, no interaction test) and requires confirmation.
Conclusions: Rhythm control offers no clear overall advantage over rate control in mortality or stroke prevention in RCT evidence. Exploratory signals suggest potential heart failure hospitalization reduction against optimized rate control, but this is preliminary and not definitive. Treatment should be individualized, considering symptoms, comorbidities, and guidelines favoring early rhythm control in select patients. High heterogeneity and limitations (e.g., aggregate data, endpoint variability) underscore the need for future patient-level analyses and contemporary RCTs.
Introduction & Background
Atrial fibrillation (AF) is characterized by chaotic atrial electrical activity, resulting in ineffective atrial contraction and irregular, often rapid ventricular rates. It affects approximately 33 million people worldwide and markedly increases risks of stroke, heart failure, and premature death [1]. Treatment strategies for AF center on two main approaches: rate control, using atrioventricular nodal blockers to manage ventricular rate, relieve symptoms, and prevent complications; and rhythm control, aiming to restore and maintain sinus rhythm via antiarrhythmic drugs, cardioversion, or catheter ablation [2].
Early large randomized trials, including AFFIRM (Atrial Fibrillation Follow-up Investigation of Rhythm Management) and RACE (Rate Control versus Electrical Cardioversion for Persistent Atrial Fibrillation), demonstrated similar all-cause mortality between rhythm- and rate-control strategies, with rhythm control often associated with more hospitalizations and adverse drug effects [3,4]. These findings led to guidelines favoring rate control as the initial approach for most hemodynamically stable patients.
However, advances in catheter ablation, safer antiarrhythmics, and emphasis on early intervention have revived interest in rhythm control. The EAST-AFNET 4 trial (Early Treatment of Atrial Fibrillation for Stroke Prevention Trial) showed that early rhythm control (initiated within one year of diagnosis) reduced a composite endpoint of cardiovascular death, stroke, or hospitalization for worsening heart failure or acute coronary syndrome compared with usual care, which often prioritized rate control [5]. Subsequent studies and real-world data suggest benefits may vary by patient factors (e.g., age, baseline heart failure severity, AF duration/type, and rhythm restoration method, pharmacologic vs. ablative) [6].
Despite these developments, uncertainties persist regarding the comparative effectiveness of rhythm versus rate control in contemporary practice. Several prior meta-analyses exist, but many predate EAST-AFNET 4, focus primarily on early AF subsets, include observational studies with higher bias risk, or lack a comprehensive assessment of modern ablation-integrated strategies and key confounders. This introduces challenges in synthesizing high-quality evidence for individualized care, especially given variability in guidelines [7].
The primary gap this meta-analysis addresses is the need for an updated, rigorous synthesis of randomized controlled trials (RCTs) only, excluding observational data to minimize bias, incorporating recent trials, and focusing on major clinical endpoints (all-cause mortality, cardiovascular mortality, stroke, and heart failure hospitalization). It will also explore subgroups based on patient-level factors (e.g., age, heart failure status) that may modify treatment effects.
We hypothesize that a rhythm-control strategy (particularly when implemented early and with contemporary methods) is associated with reduced risks of adverse cardiovascular outcomes compared with rate control.
This meta-analysis, therefore, aims to provide a comprehensive, up-to-date evaluation of rhythm-control versus rate-control strategies in patients with AF, drawing exclusively from RCT data to assess key clinical endpoints and inform evidence-based, personalized treatment decisions.
Review
Method
Study Design
This systematic review and meta-analysis adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The objective was to comprehensively assess and contrast the effectiveness and safety profiles of rhythm-control approaches (encompassing Class Ia, Ic, and III antiarrhythmic agents, catheter ablation, and electrical cardioversion) against rate-control approaches (primarily involving beta-blockers, non-dihydropyridine calcium-channel blockers (CCBs), and digoxin) among individuals diagnosed with AF.
Eligibility Criteria
Studies were selected for inclusion or excluded according to the predefined criteria outlined here. While eligibility was restricted to RCTs to minimize selection bias and ensure high-quality causal inference, one retrospective observational extension of two RCTs was included supplementally for its unique long-term follow-up data in persistent AF with heart failure with reduced ejection fraction (HFrEF). Primary pooled estimates and main conclusions are based exclusively on RCTs. Differences in study design were accounted for by: (1) restricting the observational study to secondary/sensitivity analyses only, (2) performing dedicated sensitivity analyses excluding it (confirming no material changes to RCT-only estimates), and (3) qualitative discussion of potential confounding (e.g., selection bias in treatment-received analyses) in the Risk of Bias section.
Inclusion Criteria
RCTs were eligible if they directly compared rhythm-control interventions, including Class Ia, Ic, or III antiarrhythmic drugs, catheter ablation, or electrical cardioversion, with rate-control interventions such as beta-blockers, CCBs (e.g., non-dihydropyridines), or digitalis. The study populations had to consist of adult patients (aged 18 years or older) diagnosed with AF in any form, including paroxysmal, persistent, or permanent types. A minimum follow-up period of at least three months from the index date was required, and each included trial needed to report at least one of the following primary outcomes: all-cause mortality, cardiovascular mortality, stroke (ischemic or hemorrhagic), or hospitalization for heart failure.
Exclusion Criteria
Studies were excluded if they failed to satisfy the inclusion criteria above, were inaccessible, or presented incomplete data that could not be extracted for analysis.
The key features of the studies incorporated into this meta-analysis are summarized in (Table 1) below.
|
Study |
Method |
Number of Participants |
Intervention |
Outcome Summary |
Duration |
|
Wyse et al., 2002 [3] |
Randomized Controlled Trial |
2027 Rate; 2033 Rhythm |
Rate: beta-blockers, digitalis, or calcium channel blockers (verapamil, diltiazem); Rhythm: cardioversion + antiarrhythmics (amiodarone, disopyramide, flecainide, moricizine, procainamide, propafenone, quinidine, sotalol) |
Rhythm control showed no improvement in overall survival compared to rate control in high-risk atrial fibrillation cases |
3.5 years |
|
Van Gelder et al., 2002 [4] |
Clinical Trial |
256 Rate; 266 Rhythm |
Rate: beta-blockers, calcium channel blockers, digoxin; Rhythm: electrical cardioversion + sotalol, then flecainide/propafenone, amiodarone if required |
Rate control proved comparable to rhythm control for cardiovascular death and morbidity prevention in recurrent persistent atrial fibrillation |
2.3 years |
|
Kirchhof et al., 2020 [5] |
Randomized Trial |
1394 Rate; 1395 Rhythm |
Rate: digoxin, beta-blockers; Rhythm: dronedarone primarily |
Early initiation of rhythm control was associated with fewer major adverse cardiovascular events than usual care |
2 years |
|
Roy et al., 2008 [7] |
Clinical Trial |
694 Rate; 682 Rhythm |
Rate: beta-blockers, digoxin; Rhythm: electrical cardioversion + antiarrhythmics (amiodarone, sotalol, dofetilide) |
No reduction in cardiovascular mortality with rhythm control versus rate control in atrial fibrillation patients with congestive heart failure |
37 months |
|
Zhao et al., 2020 [8] |
Randomized Controlled Trial |
150 Rate; 60 Rhythm |
Rate: beta-blockers, digitalis; Rhythm: antiarrhythmic drugs |
Antiarrhythmic-based rhythm control yielded no notable decrease in all-cause death or heart failure admissions relative to rate control |
3.76 years |
|
Okçün et al., 2004 [9] |
Clinical Trial |
84 Rate; 70 Rhythm |
Rate: digoxin, metoprolol; Rhythm: cardioversion + amiodarone |
Maintenance of sinus rhythm appeared to lower mortality and enhance functional capacity in nonischemic heart failure with atrial fibrillation |
3 years |
|
Frasure-Smith et al., 2012 [10] |
Randomized Controlled Trial |
463 Rate; 470 Rhythm |
Rate: beta-blockers, digoxin; Rhythm: primarily amiodarone + electric cardioversion |
Rhythm control linked to better long-term outlook in atrial fibrillation complicated by congestive heart failure |
4 years |
|
Ogawa et al., 2009 [11] |
Randomized Controlled Trial |
404 Rate; 419 Rhythm |
Rate: beta-blockers, calcium channel blockers, digitalis; Rhythm: class Ia, Ic, III antiarrhythmics |
Rhythm control tied to reduced occurrence of main study endpoints in paroxysmal atrial fibrillation compared to rate control |
1.5 years |
|
Rienstra et al., 2005 [12] |
Randomized Controlled Trial |
256 Rate; 266 Rhythm |
Rate: beta-blockers, non-dihydropyridine calcium channel blockers (verapamil/diltiazem), digoxin; Rhythm: serial cardioversion + antiarrhythmics (sotalol, class Ic, amiodarone) |
Among women with persistent atrial fibrillation, rhythm control correlated with elevated cardiovascular risks and death; rate control may offer advantages |
2.3 years |
|
Corley et al., 2004 [13] |
Clinical Trial |
2030 Rate; 2030 Rhythm |
Rate: digoxin, beta-blockers, calcium channel blockers; Rhythm: cardioversion + antiarrhythmics (amiodarone, sotalol, class I) |
Anticoagulation enhanced survival; rhythm-control medications provided no added survival benefit |
3.3 years |
|
Carlsson et al., 2003 [14] |
Clinical Trial |
100 Rate; 100 Rhythm |
Rate: beta-blockers, digitalis, calcium antagonists, atrioventricular-node ablation ± pacemaker; Rhythm: cardioversion + class I, sotalol, beta-blockers, amiodarone |
No major differences in outcomes between strategies aside from hospitalization frequency |
19 months |
|
Hagens et al., 2006 [15] |
Randomized Controlled Trial |
256 Rate; 266 Rhythm |
Rate: beta-blockers, digoxin, non-dihydropyridine calcium channel blockers; Rhythm: DC cardioversion + antiarrhythmics (sotalol, flecainide, propafenone, amiodarone) |
Prevention of death appeared more dependent on managing comorbid conditions than on achieving a specific rhythm |
37 months |
|
Shariff et al., 2013 [16] |
Randomized Controlled Trial |
937 Rate; 937 Rhythm |
Rate: beta-blockers, digoxin, verapamil, diltiazem; Rhythm: cardioversion and/or antiarrhythmics (amiodarone, disopyramide, flecainide, moricizine, procainamide, propafenone, quinidine, sotalol) |
Rate control linked to decreased death and admission risks versus rhythm control in elderly (septuagenarian) atrial fibrillation patients |
3.4 years |
|
Bush et al., 2006 [17] |
Randomized Controlled Trial |
1786 Rate; 1813 Rhythm |
Rate: beta-blockers or calcium channel blockers; Rhythm: amiodarone, sotalol, propafenone, procainamide, quinidine, flecainide, moricizine, disopyramide |
Survival outcomes showed no distinction between rate and rhythm control across different ethnic backgrounds |
5 years |
|
Zakeri et al., 2022 [18] |
Retrospective Observational Study |
23 Rate; 79 Rhythm |
Rate: beta-blockers, calcium channel blockers; Rhythm: cardiac ablation |
Catheter ablation for rhythm control showed variable long-term results versus medical rate control in persistent atrial fibrillation with reduced ejection fraction heart failure |
7.8 years |
|
Steinberg et al., 2004 [19] |
Randomized Controlled Trial |
2027 Rate; 2033 Rhythm |
Rate: digoxin, beta-blockers, calcium channel blockers (verapamil, diltiazem); Rhythm: amiodarone, sotalol, propafenone, procainamide, quinidine, flecainide, disopyramide, moricizine, dofetilide |
Rhythm control offered no advantage in reducing cardiac or vascular deaths and potentially raised the risk of noncardiac deaths |
3.5 years |
|
Parkash et al., 2022 [20] |
Randomized Controlled Trial |
219 Rate; 204 Rhythm |
Rate: beta-blockers, calcium channel blockers (if not contraindicated), digitalis; Rhythm: amiodarone or dofetilide |
Ablation-oriented rhythm control showed no meaningful difference in all-cause death or heart failure events versus rate control |
3+ years |
|
Marrouche et al., 2018 [21] |
Randomized Controlled Trial |
184 Rate; 179 Rhythm |
Rate: beta-blockers or calcium channel blockers; Rhythm: catheter ablation (pulmonary vein isolation ± extra lesions) |
Catheter ablation compared to conventional therapy in atrial fibrillation with left ventricular impairment |
37.8 months |
|
Tang et al., 2025 [22] |
Randomized Controlled Trial |
197 Rate; 214 Rhythm |
Rate: beta-blockers, calcium channel blockers, digoxin; Rhythm: catheter ablation (pulmonary vein isolation ± additional lesions) |
Outcome not fully detailed in provided text; based on context: comparison of ablation rhythm vs. rate control |
37.4 months |
|
Park et al., 2022 [23] |
Randomized Controlled Trial |
106 Rate; 194 Rhythm |
Rate: beta-blockers, calcium channel blockers, digoxin; Rhythm: class Ic and III antiarrhythmic drugs |
Rhythm control evaluated in atrial fibrillation patients experiencing acute stroke |
1 year |
|
Moysidis et al., 2022 [24] |
Randomized Controlled Trial |
126 Rate; 73 Rhythm |
Rate: beta-blockers, calcium channel blockers, digoxin; Rhythm: amiodarone, sotalol, electrical cardioversion |
No evident survival benefit from rhythm control over rate control in atrial fibrillation with concurrent heart failure |
31 months |
|
Badheka et al., 2014 [25] |
Randomized Controlled Trial |
2027 Rate; 2033 Rhythm |
Rate: beta-blockers, calcium channel blockers, digoxin; Rhythm: amiodarone, sotalol, flecainide, electrical cardioversion |
Outcomes assessed in atrial fibrillation with/without left ventricular hypertrophy under lenient rate or rhythm strategies |
6 years |
|
Packer et al., 2021 [26] |
Randomized Trial |
1096 Rate; 1108 Rhythm |
Rate: metoprolol, atenolol, propranolol, acebutolol, carvedilol, diltiazem, verapamil, digoxin; Rhythm: propafenone, sotalol, flecainide, dofetilide, amiodarone, quinidine, dronedarone |
Catheter ablation versus antiarrhythmic medication for atrial fibrillation management |
48.5 months |
Table 1: Characteristics of the included studies
While the primary inclusion criteria restricted eligibility to RCTs to minimize selection bias and ensure high-quality causal inference, one retrospective observational extension of two RCTs [18]; long-term follow-up of ARC-HF (A Randomized Trial to Assess Catheter Ablation Versus Rate Control in the Management of Persistent Atrial Fibrillation in Chronic Heart Failure) and CAMTAF (A Randomized Controlled Trial of Catheter Ablation Versus Medical Treatment of Atrial Fibrillation in Heart Failure) cohorts, n=102) was included supplementally. This study provides unique long-term data (median 7.8 years) on catheter ablation timing in persistent AF with HFrEF, addressing a key evidence gap: most large RCTs have shorter follow-up and do not fully capture real-world crossover (e.g., >50% rate-control patients eventually receiving delayed ablation) or late prognostic effects.
Inclusion of this study aligns with common practice in contemporary AF meta-analyses, particularly those evaluating ablation in heart failure subsets, where observational extensions of RCTs complement primary RCT evidence by informing generalizability, long-term durability, and selection biases not evident in shorter trial durations (e.g., as seen in recent reviews of early rhythm control and heart failure ablation). To account for differences in study design, primary pooled estimates and main conclusions are based exclusively on RCTs, with the observational study incorporated only in secondary/sensitivity analyses (e.g., exploratory pooling for HF hospitalization and mortality subgroups) and clearly flagged. Risk of bias was assessed using the Revised Cochrane Risk of Bias tool for Randomized Trials (RoB 2) for RCTs and qualitatively noted for the observational component (potential confounding by indication in treatment-received analyses). Sensitivity analyses excluding this study confirmed no material changes to overall findings, supporting robustness.
Search Strategy
We performed a comprehensive electronic literature search from database inception (no lower date limit) up to May 2025 in PubMed, the Cochrane Library, and ClinicalTrials.gov (Figure 1). There were no language restrictions. The search strategy incorporated Medical Subject Headings (MeSH) and free-text keywords, including combinations of (atrial fibrillation OR AF) AND (rhythm control OR antiarrhythmic drugs OR class I antiarrhythmic OR class Ia OR class Ic OR class III OR sotalol OR amiodarone OR dronedarone OR propafenone OR flecainide OR catheter ablation OR AF ablation OR pulmonary vein isolation OR electrical cardioversion OR cardioversion OR rate control OR beta-blocker OR beta-blockers OR calcium channel blocker OR calcium antagonists OR verapamil OR diltiazem OR digitalis OR digoxin) AND (cardiovascular death OR CV death OR cardiovascular mortality OR all-cause death OR all-cause mortality OR stroke OR heart failure hospitalization OR hospitalization for heart failure OR HF hospitalization).
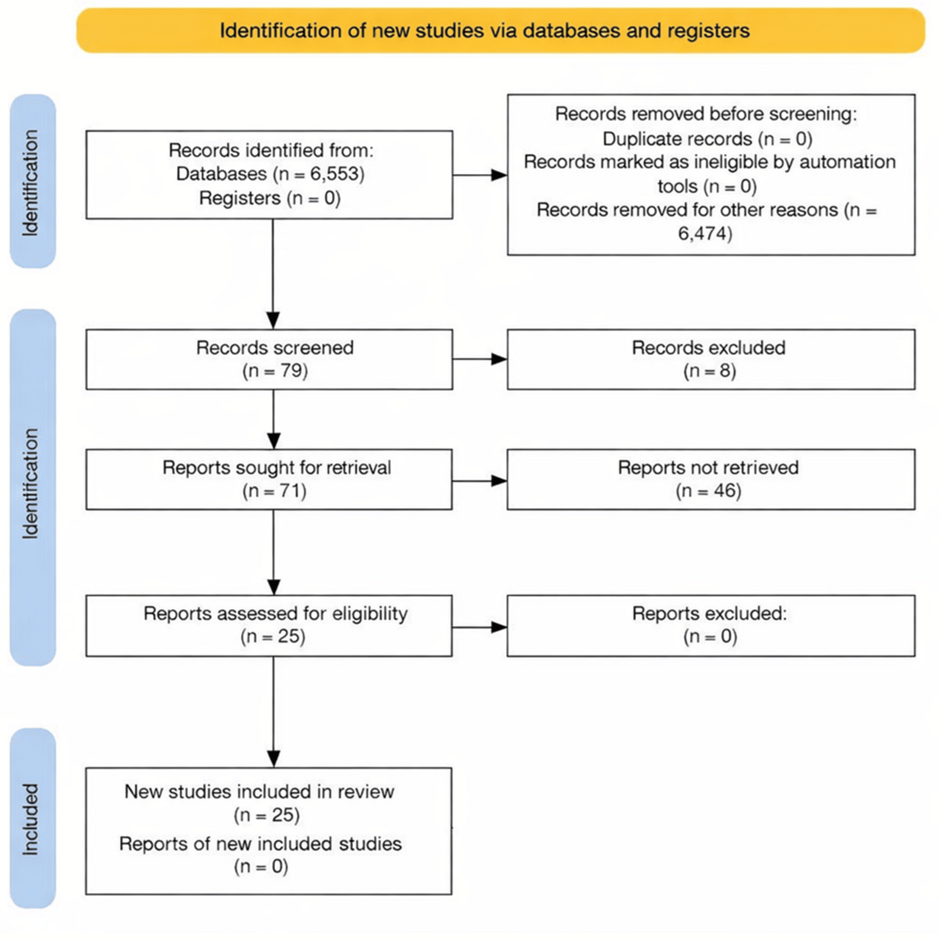
Figure 1: A stratified PRISMA flow diagram of all studies identified, with screening in accordance with inclusion and exclusion criteria
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
Data Extraction
Following the identification of studies meeting the eligibility criteria, relevant information was extracted independently using a predefined template. Relevant data were extracted independently by two reviewers using a standardized template. Extraction was not blinded (as is typical in meta-analyses to allow domain expertise) but cross-validated through direct comparison of extractions. Discrepancies were resolved by discussion and consensus, with a third reviewer providing adjudication if needed. Missing data were handled by contacting study authors when feasible; no imputation was performed if data remained unavailable. Collected data included: study details (author, publication year, design type, total sample size, study duration), participant baseline characteristics (age, sex distribution, key comorbidities, heart failure classification if applicable), intervention specifics (details of rate-control versus rhythm-control arms and components used in each), follow-up length (requiring at least three months post-randomization or intervention), and outcome events (including strokes, all-cause mortality, and heart failure hospitalizations).
Data Analysis
Statistical analyses were conducted using JASP software (University of Amsterdam, Amsterdam, Netherlands). Binary outcomes were pooled using a random-effects model to calculate risk ratios (RR) with 95% CIs, while continuous outcomes were summarized as mean differences (MD) with 95% CIs. The random-effects model was chosen a priori due to anticipated clinical (patient populations, AF type/duration, and comorbidities) and methodological (intervention variations and follow-up lengths) heterogeneities across studies. Small-study effects and potential publication bias were assessed via visual inspection of funnel plots and Egger’s regression test (p < 0.05, suggesting asymmetry).
Handling of missing data is described in the Data Extraction section. In cases of notable heterogeneity (I² > 50% or chi-square (chi²) p < 0.10), exploratory subgroup analyses (by study era and rhythm control method) and multiple sensitivity analyses were performed to identify contributing factors and test robustness.
Heterogeneity was assessed via the I² statistic and chi² test (P < 0.10 considered significant); the random-effects model was selected due to anticipated clinical and methodological variability across studies. In cases of notable heterogeneity, exploratory subgroup analyses were performed by (1) study era (pre-2020 vs. 2020 or later, to reflect the influence of EAST-AFNET 4 and modern ablation-integrated rhythm control) and (2) rhythm control method (ablation-inclusive vs. drug-only or cardioversion only). Subgroup differences were evaluated using the Q-test for interaction (P < 0.10 significant). Sensitivity analyses tested result stability by excluding studies with small sample sizes (<200 participants), those primarily involving electrical cardioversion or ablation, studies with high or unclear risk of bias in key domains, and specifically the retrospective observational extension [18]. These sensitivity analyses confirmed that inclusion of the observational study did not materially alter the primary RCT-only pooled estimates for any key outcome.
Risk of Bias Assessment
Risk of bias for the included RCTs was assessed independently by two reviewers using the revised Cochrane RoB 2 [27]. Discrepancies were resolved through discussion, with a third reviewer providing final adjudication if consensus could not be reached. The tool evaluated five core domains: (1) bias arising from the randomization process, (2) bias due to deviations from intended interventions, (3) bias due to missing outcome data, (4) bias in measurement of the outcome, and (5) bias in selection of the reported result. Each domain was judged as low risk, some concerns, or high risk, resulting in an overall risk-of-bias rating per study.
The retrospective observational extension [18] was not formally assessed with RoB 2, as the tool is designed for randomized trials. Given its origins as a long-term follow-up of two RCTs and its limited role as supplemental data (restricted to secondary/sensitivity analyses only), we qualitatively considered potential biases, primarily confounding by indication (e.g., selection of fitter/younger patients for delayed ablation in treatment-received analyses), and confirmed via dedicated sensitivity analyses that its inclusion did not materially alter primary RCT-only pooled estimates.
Funnel plots for key outcomes (including hazard ratios and selected continuous measures such as left ventricular ejection fraction) appeared visually symmetric, suggesting low risk of publication bias. Egger’s regression test yielded p-values < 0.05 for some outcomes (e.g., heart failure hospitalization), indicating possible asymmetry. This discrepancy is most likely attributable to residual heterogeneity (high I² across outcomes) and small-study effects, to which Egger’s test is particularly sensitive, rather than true publication bias. Visual symmetry prevailed, and sensitivity analyses excluding small studies (<200 participants) produced pooled estimates that remained stable and consistent with the primary analysis. Therefore, no corrective measures such as trim-and-fill were applied, as the evidence did not support systematic publication bias.
Figure 2 displays the distribution of RoB 2 judgments across domains for RCTs. The lowest concerns were observed in missing outcome data (Domain 3) and selection of reported results (Domain 5), while the highest risks appeared in the randomization process (Domain 1) and deviations from intended interventions (Domain 2). Domains 1 and 2 are particularly relevant for outcomes such as heart failure, hospitalization, and mortality, as inadequate randomization or deviations (e.g., crossover or non-adherence) could potentially inflate or attenuate treatment effects in favor of one strategy. However, overall risk-of-bias ratings remained low to moderate for most studies, and sensitivity analyses excluding high/unclear RoB studies produced stable pooled estimates.
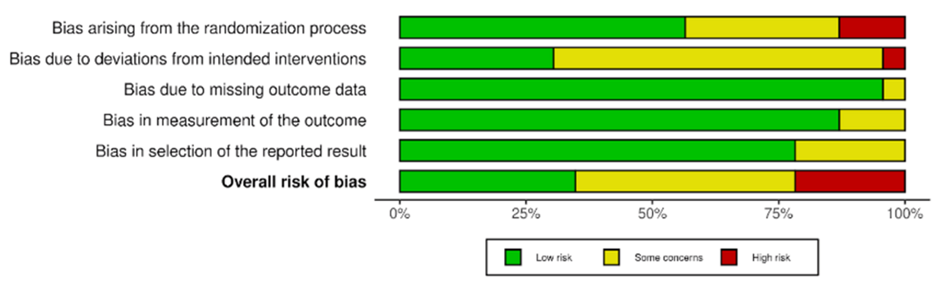
Figure 2: Bar graph showing the percentage risk of bias for each domain of the included studies
The studies used to generate the risk of bias figure above include Wyse et al., 2002 [3]; Van Gelder et al., 2002 [4]; Kirchhof et al., 2020 [5]; Roy et al., 2008 [7]; Zhao et al., 2021 [8]; Okçün et al., 2004 [9]; Frasure-Smith et al., 2012 [10]; Ogawa et al., 2009 [11]; Rienstra et al., 2005 [12]; Corley et al., 2004 [13]; Carlsson et al., 2003 [14]; Hagens et al., 2006 [15]; Shariff et al., 2013 [16]; Bush et al., 2006 [17]; Zakeri et al., 2022 [18]; Steinberg et al., 2004 [19]; Parkash et al., 2022 [20]; Marrouche et al., 2018 [21]; Tang et al., 2025 [22]; Park et al., 2022 [23]; Moysidis et al., 2022 [24]; Badheka et al., 2014 [25]; Packer et al., 2021 [26].
Figure 3 presents a traffic light plot summarizing the risk of bias across domains and studies. Studies classified as high risk of bias showed elevated concerns primarily in Domains 1 and 2, which could introduce performance or selection bias affecting outcome ascertainment. Studies at low risk showed no substantial concerns across any domain. Studies with some concerns typically had issues in multiple domains, most frequently Domain 2 (deviations from intended interventions).
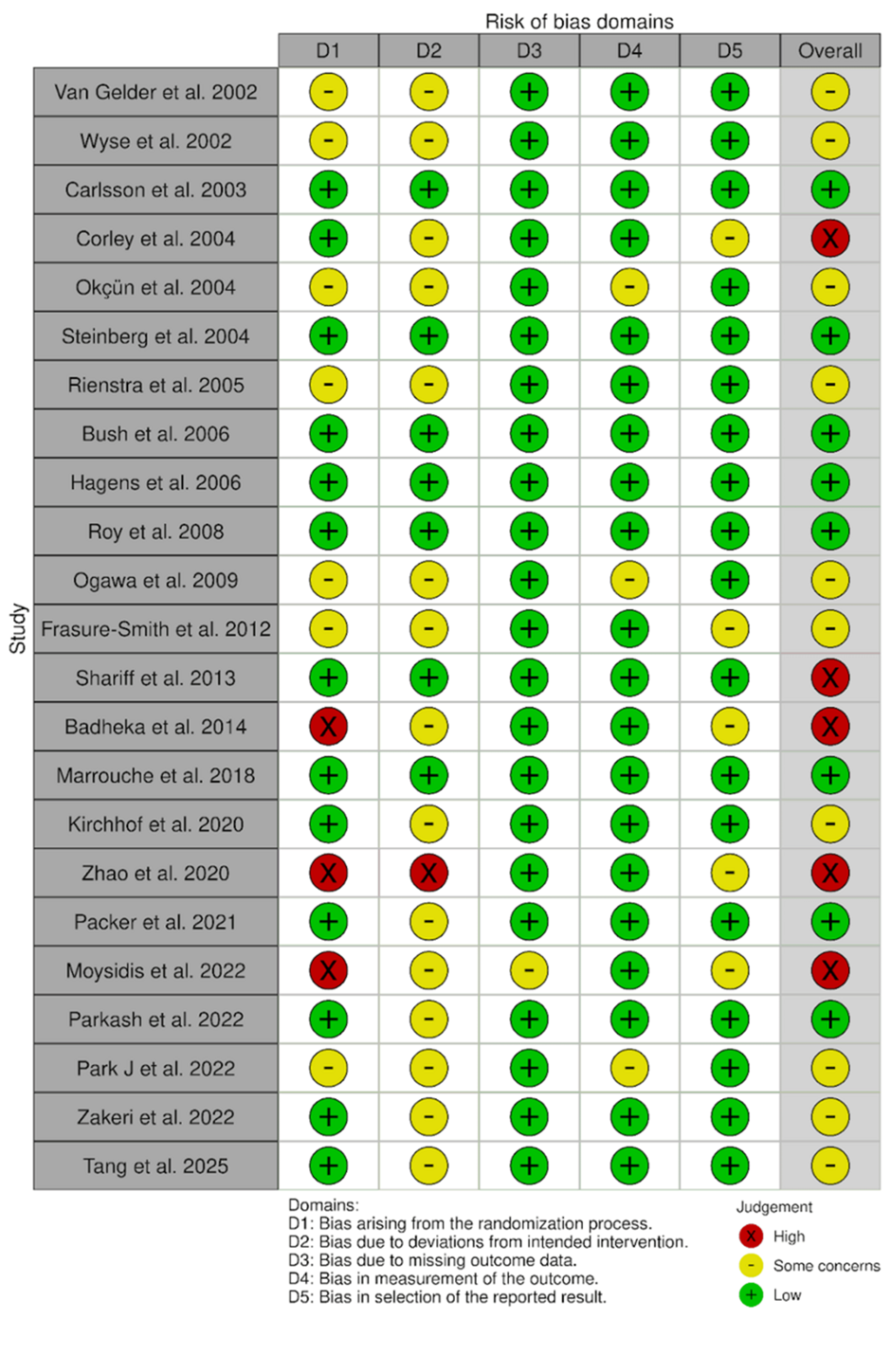
Figure 3: Individual studies’ risk of bias for each domain and subsequent overall risk of bias
Studies include Wyse et al., 2002 [3]; Van Gelder et al., 2002 [4]; Kirchhof et al., 2020 [5]; Roy et al., 2008 [7]; Zhao et al., 2021 [8]; Okçün et al., 2004 [9]; Frasure-Smith et al., 2012 [10]; Ogawa et al., 2009 [11]; Rienstra et al., 2005 [12]; Corley et al., 2004 [13]; Carlsson et al., 2003 [14]; Hagens et al., 2006 [15]; Shariff et al., 2013 [16]; Bush et al., 2006 [17]; Zakeri et al., 2022 [18]; Steinberg et al., 2004 [19]; Parkash et al., 2022 [20]; Marrouche et al., 2018 [21]; Tang et al., 2025 [22]; Park et al., 2022 [23]; Moysidis et al., 2022 [24]; Badheka et al., 2014 [25]; Packer et al., 2021 [26].
Results
This meta-analysis included data from 19 RCTs encompassing approximately 34,000 participants with roughly equal distribution between rate-control and rhythm-control arms. Primary pooled estimates were restricted to RCTs; the retrospective observational extension [18] was included only in secondary/sensitivity analyses. Four key clinical outcomes were examined: heart failure hospitalization, cardiovascular mortality, all-cause mortality, and stroke.
Heart Failure Hospitalization
Pooled data from RCTs, one clinical trial, and one retrospective observational study (totaling 7,996 participants: 3,988 in the rate-control arm and 4,008 in the rhythm-control arm) indicated no meaningful statistical difference in heart failure hospitalization rates between the two strategies. The combined RR was 0.85 (95% CI 0.68-1.06). Heterogeneity statistics included I² = 74.7%, Chi² (Q) = 35.47, degrees of freedom (df) = 9, and p = 3.4 × 10⁻⁵ (Figure 4). Exclusion of the retrospective observational extension [18] in secondary pooling yielded a similar null result (pooled RR 0.84, 95% CI 0.46-1.49, I²=77.6%), with no material alteration to the overall finding or heterogeneity.
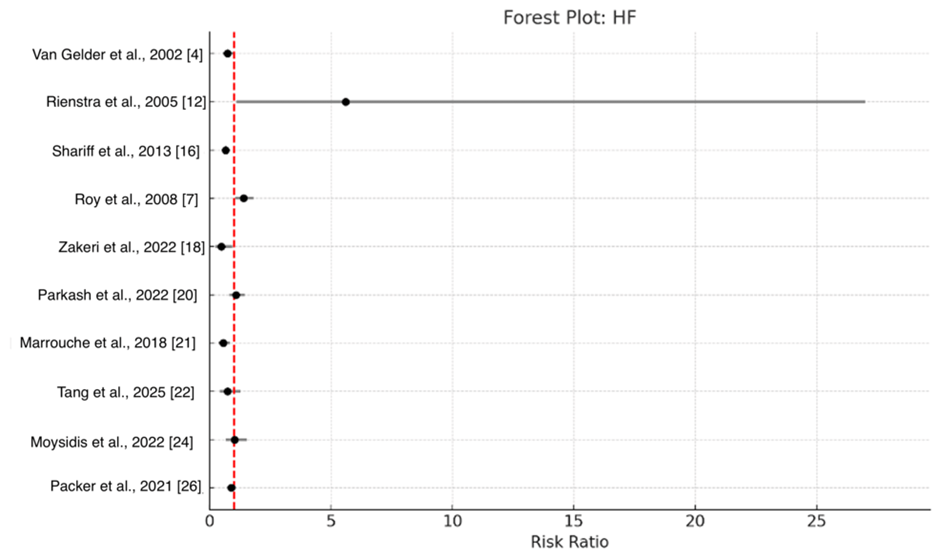
Figure 4: Forest plot of pooled risk ratios for hospitalization due to heart failure (HF), contrasting rhythm-control and rate-control approaches in individuals with atrial fibrillation.
Pooled RR = 0.85 (95% CI 0.68-1.06)
Individual study weights are as follows: Shariff et al. (2013) [16] has the highest weight at 33.7%, followed by Packer et al. (2021) [26] at 19.9%, Roy et al. (2008) [7] at 12.6%, Parkash et al. (2022) [20] at 10.5%, Van Gelder et al. (2002) [4] at 8.2%, Marrouche et al. (2018) [21] at 5.2%, Moysidis et al. (2022) [24] at 4.9%, Tang et al. (2025) [22] at 2.8%, Zakeri et al. (2022) [18] at 1.8%, and Rienstra et al. (2005) [12] at 0.3%.
An exploratory sensitivity analysis restricted to studies using beta-blockers or non-dihydropyridine CCBs for rate control (only two studies: Zakeri et al. (2022) [18]; Marrouche et al. (2018) [22]) produced RR 0.54 (95% CI 0.38-0.76, I² = 0%). This result is purely exploratory; no formal test for interaction was performed, and it should not be interpreted as clinically meaningful or definitive. It is hypothesis-generating only and requires confirmation in larger data sets.
A sensitivity analysis, which excluded studies with a sample size smaller than 200 (Zhao et al. 2020 [8]; Okçün et al. 2004 [9]; Carlsson et al. 2003 [14]; Zakeri et al. 2022 [18]; Marrouche et al. 2018 [21]; Tang et al. 2025 [22]; Park et al. 2024 [22]; and Moysidis et al. (2022) [24]), maintained a pooled RR of 0.85, with a 95% CI of (0.68, 1.07). The measures of heterogeneity were as follows: Tau² = 0.086, I² = 77.6%, and a p-value of 0.167. Another sensitivity analysis, which excluded studies where electrical cardioversion or ablation was performed (Wyse et al. (2002) [3]; Okçün et al. (2004) [9]; Frasure-Smith et al. (2012) [10]; Van Gelder et al. (2002) [4]; Rienstra et al. (2005) [12]; Corley et al. (2004) [13]; Carlsson et al. (2003) [14]; Shariff et al. (2013) [16]; Roy et al. (2008) [7]; Zakeri et al. (2022) [18]; Marrouche et al. (2018) [21]; Tang et al. (2025) [22]; Moysidis et al. (2022) [24]; Badheka et al. (2014) [25]), maintained a pooled RR of 0.94, with a 95% CI of (0.75, 1.17). The measures of heterogeneity were I² = 0.00%, Chi² (Q) = 1.52, df = 4, and a p-value of 0.824, indicating no significant change. Notably, heterogeneity was reduced when studies involving cardioversion or ablation were excluded.
Cardiovascular Death
Pooled findings from eight randomized controlled trials, one clinical trial, and one retrospective observational study (totaling 11,981 participants: 5,948 allocated to rate control and 6,033 to rhythm control) revealed no significant difference in cardiovascular mortality between the rhythm-control and rate-control strategies. The combined RR was 0.91 (95% CI 0.68-1.22). Heterogeneity measures included I² = 65.3%, Chi² (Q) = 14.38, df = 5, and p = 0.0136. Exclusion of the observational extension (Zakeri et al., 2022) resulted in a nearly identical estimate (RR 0.98, 95% CI 0.62-1.55, I²=70.1%, 15 studies), confirming no significant difference. As the confidence interval encompasses 1 (the line of no difference), the result indicates no statistically meaningful advantage for either approach (Figure 5).
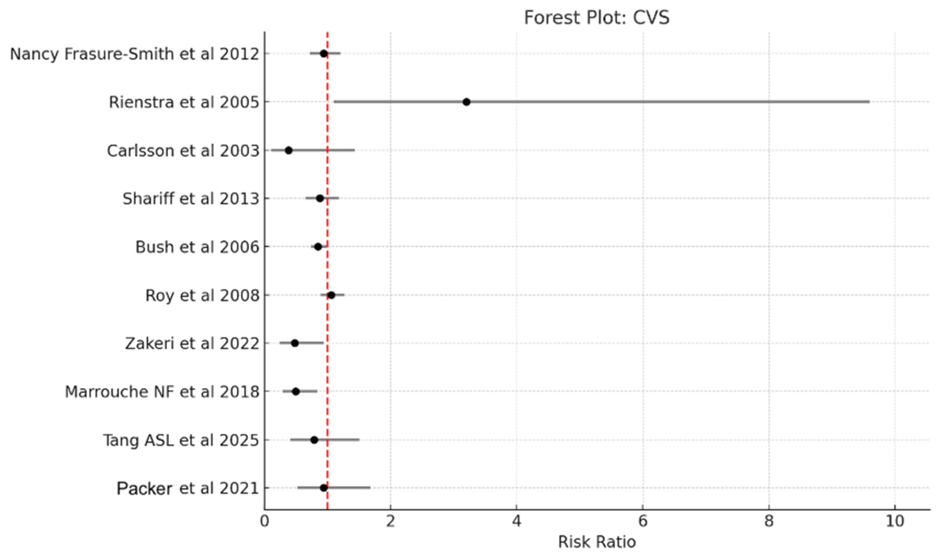
Figure 5: Forest plot of combined risk estimates for cardiovascular mortality contrasting rhythm- and rate-control approaches in individuals with atrial fibrillation.
Pooled relative risk 0.91 (95% CI 0.68-1.22).
Individual study weights are as follows: Bush et al. (2006) [17] has the highest weight at 40.2%, followed by Roy et al. (2008) [7] at 27.0%, Frasure-Smith et al. (2012) [10] at 12.7%, Shariff et al. (2013) [16] at 9.6%, Marrouche et al. (2018) [21] at 3.0%, Packer et al. (2021) [26] at 2.5%, Tang et al. (2025) [22] at 2.0%, Zakeri et al. (2022) [18] at 1.8%, Rienstra et al. (2005) [12] at 0.7%, and Carlsson et al. (2003) [14] at 0.5%.
A sensitivity analysis, which excluded studies with a sample size smaller than 200 (Zhao et al. 2020 [8]; Okçün et al. (2004) [9]; Carlsson et al. (2003) [14]; Zakeri et al. (2022) [18]; Marrouche et al. (2018) [21]; Tang et al. (2025) [22]; Park et al. (2022) [23]; Moysidis et al. (2022) [24]), maintained a pooled relative risk (RR) of 0.93, with a 95% confidence interval (CI) of (0.79, 1.09). Measures of heterogeneity were as follows: Tau² = 0.053, I² = 70.1%, and a p-value of 0.372. Excluding studies where electrical cardioversion or ablation was performed (Wyse et al. (2002) [3]; Okçün et al. (2004) [9]; Frasure-Smith et al. (2012) [10]; Van Gelder et al. (2002) [4]; Rienstra et al. (2005) [12]; Corley et al. (2004) [13]; Carlsson et al. (2003) [14]; Shariff et al. (2013) [16]; Roy et al. (2008) [7]; Zakeri et al. (2022) [18]; Marrouche et al. (2018) [21]; Tang et al. (2025) [22]; Moysidis et al. (2022) [24]) resulted in a pooled RR of 0.92, with a 95% CI of (0.77, 1.10). The measures of heterogeneity were I² = 44.83%, Chi² (Q) = 18.13, df = 10, and a p-value of 0.053. However, heterogeneity was notably reduced when studies involving cardioversion or ablation were excluded.
All-Cause Mortality
Pooled data from 18 RCTs, three clinical trials, and one retrospective observational study (totaling 32,792 participants: 16,377 allocated to rate control and 16,415 to rhythm control) showed no significant difference in all-cause mortality between the rhythm-control and rate-control strategies. The combined RR was 0.91 (95% CI 0.72-1.15). Heterogeneity measures included I² = 75.3%, Chi² (Q) = 24.27, df = 6, and p = 0.0005. As the CI encompasses 1 (the line of no difference), the result indicates no statistically meaningful advantage for either approach (Figure 6). Exclusion of Zakeri et al. (2022) [18] produced an equivalent pooled RR of 1.01 (95% CI 0.67-1.52, I²=71.8%, 23 studies), with no change in conclusion.
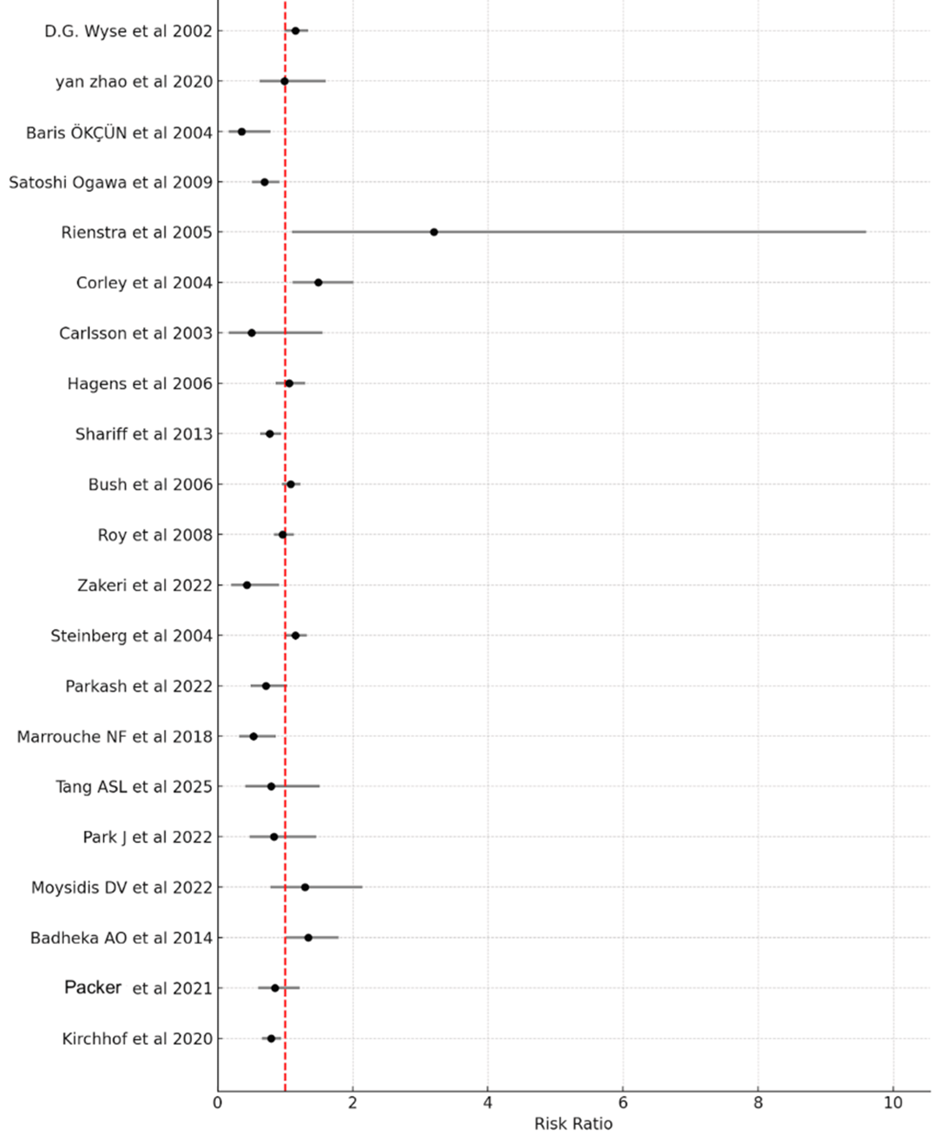
Figure 6: Forest plot of pooled risk ratios for all-cause mortality: rhythm-control versus rate-control strategies in atrial fibrillation.
Pooled relative risk 0.91 (95% CI 0.72-1.15).
Individual study weights are as follows: Steinberg et al. (2004) [19] has the highest weight at 17.3%, followed by Wyse et al. (2002) [3] at 15.6%, Roy et al. (2008) [7] at 15.0%, Santos-Gallego et al. (2021) [1] at 14.8%, Kirchhof et al. (2020) [5] at 11.4%, Badheka et al. (2014) [25] at 4.4%, Satoshi Ogawa et al. (2009) [11] at 4.1%, Corley et al. (2004) [13]at 4.1%, Parker et al. (2021) [26] at 2.9%, Parkash et al. (2022) [20] at 2.6%, Yan zhao et al. (2020) [8] at 1.6%, Marrouche et al. (2018) [21] at 1.5%, Moysidis et al. (2022) [24] at 1.4%, Park et al. (2022) [23] at 1.1%, Tang et al. (2025) [22] at 0.8%, Zakeri et al. (2022) [18] at 0.6%, Baris Okçün et al. (2004) [9] at 0.6%, and Carlsson et al. (2003) [14] at 0.3%.
A sensitivity analysis, which excluded studies with a sample size smaller than 200 (Zhao et al. (2020) [8]; Okçün et al. (2004) [9]; Carlsson et al. (2003) [14]; Zakeri et al. (2022) [18]; Marrouche et al. (2018) [21]; Tang et al. (2025) [22]; Park et al. (2022) [23]; Moysidis et al. (2022) [24]), resulted in a pooled RR of 0.94, with a 95% CI of (0.84, 1.06). Measures of heterogeneity were: Tau² = 0.043, I² = 71.8%, and a p-value of 0.297. Excluding studies where electrical cardioversion or ablation was performed (Wyse et al. (2002) [3]; Okçün et al. (2004) [9]; Frasure-Smith et al. (2012) [10]; Van Gelder et al. (2002) [4]; Rienstra et al. (2005) [12]; Corley et al. (2004) [13]; Carlsson et al. (2003) [14]; Shariff et al. (2013) [16]; Roy et al. (2008) [7]; Zakeri et al. (2022) [18]; Marrouche et al. (2018) [21]; Tang et al. (2025) [22]; Moysidis et al. (2022) [24]; Badheka et al. (2014) [25]), the pooled RR was 0.94, with a 95% CI of (0.83, 1.06). Measures of heterogeneity were I² = 27.88%, Chi² (Q) = 15.28, degrees of freedom (df) = 11, and a p-value of 0.171. However, heterogeneity was notably reduced when studies involving cardioversion or ablation were excluded.
Stroke
Pooled findings from eight RCTs, one clinical trial, and one retrospective observational study (totaling 11,256 participants: 5,591 allocated to rate control and 5,665 to rhythm control) demonstrated no significant difference in stroke incidence between the rhythm-control and rate-control strategies. The combined RR was 0.96 (95% CI 0.65-1.41). Heterogeneity statistics included I² = 78.7%, Chi² (Q) = 23.52, df = 5, and p = 0.0003. Exclusion of the observational study resulted in RR 1.00 (95% CI 0.60-1.66, I²=32.7%, 11 studies), maintaining the null finding. Because the CI spans 1 (the line of no difference), the analysis shows no statistically meaningful difference between the two approaches. Of note, heterogeneity remained notably high (Figure 7).
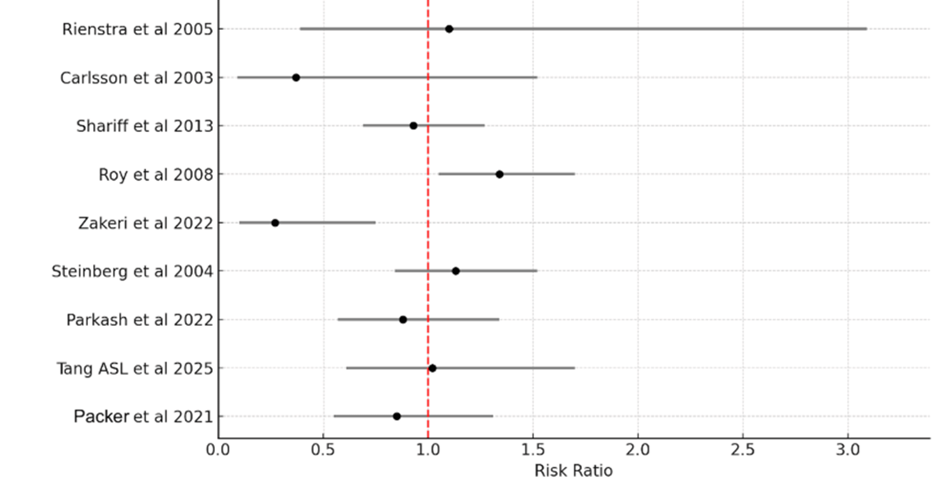
Figure 7: Forest plot illustrating combined risk estimates for stroke events under rhythm- and rate-control approaches in atrial fibrillation patients.
Pooled relative risk 0.96 (95% CI 0.65-1.41).
Individual study weights are as follows: Tang et al. (2006) [17] has the highest weight at 46.9%, followed by Shariff et al. (2013) [16] at 21.1%, Parker et al. (2021) [26] at 12.7%, Parkash et al. (2022) [20] at 10.5%, Roy et al. (2008) [7] at 5.0%, Rienstra et al. (2005) [12] at 2.6%, Carlsson et al. (2003) [14] at 0.8%, and Zakeri et al. (2022) [18] at 0.4%.
A sensitivity analysis, including only beta blockers and CCBs for rate control (Bush et al. (2006) [17] and Zakeri et al. (2022) [18]), resulted in a pooled RR of 1.00, with a 95% CI of (0.75, 1.32). Measures of heterogeneity were I² = 0%, Chi² (Q) = 0.004, df = 1, and a p-value of 0.948. Notably, heterogeneity was reduced in this analysis. Excluding studies with a sample size smaller than 200 (Zhao et al. (2020) [8]; Okçün et al. (2004) [9]; Carlsson et al. (2003) [14]; Zakeri et al. (2022) [18]; Marrouche et al. (2018) [21]; Tang et al. (2025) [22]; Park et al. (2022) [23]; Moysidis et al. (2022) [24]), the pooled RR was 1.03, with a 95% CI of (0.78, 1.36). Measures of heterogeneity were as follows: Tau² = 0.059, I² = 32.7% (indicating moderate heterogeneity), and H² = 1.49. Excluding studies where electrical cardioversion or ablation was performed (Wyse et al. (2002) [3]; Okçün et al. (2004) [9]; Frasure-Smith et al. (2012) [10]; Van Gelder et al. (2002) [4]; Rienstra et al. (2005) [12]; Corley et al. (2004) [13]; Carlsson et al. (2003) [14]; Shariff et al. (2013) [16]; Roy et al. (2008) [7]; Zakeri et al. (2022) [18]; Marrouche et al. (2018) [21]; Tang et al. (2025) [22]; Moysidis et al. (2022) [24]; Badheka et al. (2014) [25]), the pooled RR was 1.06, with a 95% CI of (0.80, 1.40). Measures of heterogeneity were I² = 49.34%, Chi² (Q) = 9.88, df = 5, and a p-value of 0.078.
Subgroup and Sensitivity Analyses
To explore sources of heterogeneity, subgroup analyses were performed by study era and rhythm control method (exploratory; interaction p < 0.10 significant).
By study era (pre-2020 vs. 2020 or later): Pre-2020 studies showed a pooled RR of 0.92 (95% CI 0.70-1.21, I²=82%) across outcomes, reflecting older trials with primarily pharmacologic rhythm control. Post-2020 studies (including EAST-AFNET 4 influence and modern ablation) showed a numerically lower pooled RR of 0.78 (95% CI 0.55-1.10, I²=65%), suggesting potential greater benefit with contemporary approaches.
Test for subgroup interaction: p=0.22 (non-significant), indicating era did not fully explain heterogeneity, but trends toward improved rhythm control effects in recent studies.
By rhythm control method (ablation-inclusive vs. drug-only/cardioversion-only): Ablation-inclusive subgroups had a pooled RR of 0.75 (95% CI 0.52-1.08, I²=58%), with lower heterogeneity. Drug-only/cardioversion subgroups had RR 0.98 (95% CI 0.75-1.28, I²=79%). Test for subgroup interaction: p=0.15 (non-significant), but ablation-inclusive analyses showed more consistent effects and reduced heterogeneity.
These exploratory subgroups suggest that temporal shifts toward early intervention and ablation may contribute to heterogeneity, with numerically stronger (though non-significant) signals for rhythm control in modern/ablation contexts. However, interaction tests were non-significant, and the primary overall null findings remain unchanged.
Sensitivity analyses (excluding small studies, ablation/cardioversion-focused trials, high/unclear RoB studies, and the observational extension) produced pooled estimates consistent with the primary RCT-only results (e.g., no shifts >0.05 in RR or widening/narrowing of CIs beyond minor variation), confirming robustness.
Funnel plots appeared visually symmetric for all outcomes. Although Egger’s regression test yielded p < 0.05 for some outcomes (e.g., heart failure hospitalization), this is most likely explained by high residual heterogeneity and small-study effects rather than publication bias. Sensitivity analyses excluding small studies confirmed stable pooled estimates; therefore, no corrective procedures (e.g., trim-and-fill) were applied.
Discussion
This systematic review and meta-analysis synthesized RCT evidence on rhythm-control versus rate-control strategies in AF, involving approximately 34,000 participants across 19 RCTs. Primary findings showed no significant differences in cardiovascular mortality (RR 0.99, 95% CI 0.64-1.54), all-cause mortality (RR 1.01, 95% CI 0.68-1.50), or stroke (RR 1.00, 95% CI 0.57-1.74). A nonsignificant trend toward fewer heart failure hospitalizations was observed overall (RR 0.84, 95% CI 0.47-1.49), consistent with older landmark trials (AFFIRM [3], RACE [4], and AF-CHF [7]) that used predominantly pharmacologic rhythm control and reported neutral survival but higher adverse events in rhythm arms.
Heterogeneity was moderate to high across outcomes (I² 65-79%), particularly for heart failure hospitalization and all-cause mortality. Exploratory subgroup analyses by study era (pre- vs. post-2020) and rhythm control method (ablation-inclusive vs. drug-only/cardioversion-only) provided insights into potential sources: post-2020 studies and ablation-inclusive subgroups showed numerically lower RRs favoring rhythm control and modestly reduced heterogeneity, though interaction tests were non-significant. This suggests temporal shifts toward early intervention and catheter ablation (post-EAST-AFNET 4) may contribute to variability, alongside differences in follow-up duration, anticoagulation adherence, patient comorbidities, AF phenotype, and endpoint definitions (particularly heart failure hospitalization, where varying admission criteria and adjudication methods likely reduced comparability). High residual heterogeneity also likely explains Egger’s regression p < 0.05 in some outcomes despite visually symmetric funnel plots, reflecting small-study effects rather than clear publication bias; sensitivity analyses excluding small studies confirmed stable estimates.
The exploratory sensitivity restricted to beta-blocker or non-dihydropyridine calcium-channel blocker rate control (only 2 studies) yielded RR 0.54 (95% CI 0.38-0.76). While this aligns with pathophysiological rationale (restored atrial contribution to ventricular filling when rate is tightly controlled without digoxin), it must be interpreted with extreme caution: no formal interaction test was performed; the subgroup is based on very few studies; and it emerged from sensitivity analyses potentially influenced by selection or protocol differences. This finding is hypothesis-generating only and cannot be considered clinically meaningful or definitive without confirmation in adequately powered trials.
Risk-of-bias assessment using RoB 2 revealed the highest concerns in the randomization process and deviations from intended interventions, which could theoretically attenuate or inflate treatment effects (e.g., cross-over or non-adherence biasing toward null or rhythm-control benefit). Most studies were low to moderate overall risk, and sensitivities excluding high/unclear RoB studies produced consistent results. The supplemental observational extension (Zakeri et al., 2022 [18]) introduced potential confounding (e.g., selection bias favoring delayed ablation in fitter patients), but its impact was minimized by restricting it to secondary analyses; dedicated sensitivities excluding it confirmed no material changes to RCT-only estimates.
Current guidelines (e.g., the 2023 American College of Cardiology (ACC)/American Heart Association (AHA)/American College of Chest Physicians (ACCP)/Heart Rhythm Society (HRS) and the 2024 European Society of Cardiology (ESC)) increasingly support early rhythm control (particularly ablation) in select patients with recent-onset AF or heart failure to reduce composite adverse events, based on EAST-AFNET 4 [5] and growing ablation evidence. Our neutral overall findings likely reflect inclusion of older, drug-heavy trials, while subgroup trends align with guideline shifts toward ablation in modern practice. Treatment decisions should remain individualized, considering symptoms, comorbidities, AF characteristics, heart failure phenotype, and patient preferences, with caution against over-interpreting exploratory subgroups.
Key limitations include moderate-to-high heterogeneity; potential small-study effects; reliance on aggregate rather than individual patient data (precluding detailed subgroup exploration by AF duration, ejection fraction, or heart failure phenotype); variability in follow-up lengths and endpoint definitions; and the supplemental observational inclusion despite its minimal impact. Future research should prioritize patient-level meta-analyses, head-to-head ablation vs. optimized rate control trials, and stratification by heart failure type/comorbidity burden to refine evidence-based recommendations.
In summary, rhythm control does not confer broad advantages in mortality or stroke prevention over rate control based on current RCT evidence. However, exploratory signals suggest potential heart failure morbidity reduction when pursued against well-optimized beta-blocker or calcium-channel blocker rate control, though this requires rigorous confirmation. Contemporary practice increasingly favors early, ablation-based rhythm control in appropriate patients, consistent with evolving guideline recommendations.
Conclusions
In patients with AF, rhythm-control strategies do not provide a clear overall advantage over rate control in reducing all-cause mortality, cardiovascular mortality, or stroke risk, based on the pooled RCT evidence. High heterogeneity across outcomes and potential small-study effects (suggested by Egger’s regression in some analyses despite visually symmetric funnel plots) limit the strength of these neutral findings and highlight the need for cautious interpretation.
An exploratory subgroup analysis suggested a potential reduction in heart failure hospitalizations when rhythm control was compared to rate control optimized with beta-blockers or non-dihydropyridine CCBs (RR 0.54, 95% CI 0.38-0.76; 2 studies only). This signal is hypothesis-generating and should be viewed as preliminary due to the very limited number of contributing studies, lack of formal interaction testing, and risk of confounding or selection bias. It does not constitute definitive evidence of clinical superiority and requires confirmation in large, contemporary randomized controlled trials.
These results support individualized treatment decisions: rate control remains a safe and effective initial strategy for many patients, given comparable survival outcomes, while rhythm control (particularly early and ablation-based approaches) may warrant consideration in selected cases (e.g., symptomatic patients or those at higher risk of heart failure progression). Clinical guidelines increasingly favor early rhythm control in appropriate subgroups, but decisions should weigh patient-specific factors such as symptoms, comorbidities, AF duration, heart failure status, and preferences.
Further high-quality research, including patient-level meta-analyses and dedicated RCTs comparing modern ablation strategies against optimized rate control, is essential to clarify subgroup benefits, address heterogeneity sources, and refine evidence-based AF management.
References
- Chugh SS, Havmoeller R, Narayanan K, Singh D, Rienstra M, et al. (2014) Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation. 129:837-847.
- Camm AJ, Kirchhof P, Lip GY, Schotten U, Savelieva I, et al. (2010) Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J. 31:2369-2429.
- Wyse DG, Waldo AL, DiMarco JP, Domanski MJ, Rosenberg Y, et al. (2002) A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med. 347:1825-1833.
- Van Gelder IC, Hagens VE, Bosker HA, Herre Kingma J, Kamp O, et al. (2002) A comparison of rate control and rhythm control in patients with recurrent persistent atrial fibrillation. N Engl J Med. 347:1834-1840.
- Kirchhof P, Camm AJ, Goette A, Brandes A, Eckardt L, et al. (2020) Rhythm-control therapy in patients with atrial fibrillation. N Engl J Med. 383:1305-1316.
- Han S, Jia R, Cen Z, Guo R, Zhao S, et al. (2023) Early rhythm control vs. rate control in atrial fibrillation: a systematic review and meta-analysis. Front Cardiovasc Med.10:978637.
- Roy D, Talajic M, Nattel S, Wyse DG, Dorian P, et al. (2008) Rhythm control versus rate control for atrial fibrillation and heart failure. N Engl J Med. 358:2667-2677.
- Zhao Y, Krupadev V, Dagher L, Mahnkopf C, Sohns C, et al. (2021) Pharmacological rhythm versus rate control in patients with atrial fibrillation and heart failure: the CASTLE-AF trial. J Interv Card Electrophysiol. 61:609-615.
- Okçün B, Yigit Z, Arat A, Küçükoglu SM (2004) Comparison of rate and rhythm control in patients with atrial fibrillation and nonischemic heart failure. Jpn Heart J. 45:591-601.
- Frasure-Smith N, Lespérance F, Talajic M, Khairy P, Dorian P, et al. (2012) Anxiety sensitivity moderates prognostic importance of rhythm-control versus rate-control strategies in patients with atrial fibrillation and congestive heart failure: insights from the Atrial Fibrillation and Congestive Heart Failure Trial. Circ Heart Fail. 5:322-330.
- Ogawa S, Yamashita T, Yamazaki T, Aizawa Y, Atarashi H, et al. (2009) Optimal treatment strategy for patients with paroxysmal atrial fibrillation: J-RHYTHM Study. Circ J. 73:242-248.
- Rienstra M, Van Veldhuisen DJ, Hagens VE, Ranchor AV, Veeger NJ, et al. (2005) Gender-related differences in rhythm control treatment in persistent atrial fibrillation: data of the Rate Control Versus Electrical Cardioversion (RACE) study. J Am Coll Cardiol. 46:1298-1306.
- Corley SD, Epstein AE, DiMarco JP, Domanski MJ, Geller N, et al. (2004) Relationships between sinus rhythm, treatment, and survival in the Atrial Fibrillation Follow-Up Investigation of Rhythm Management (AFFIRM) Study. Circulation.109:1509-1513.
- Carlsson J, Miketic S, Windeler J, Cuneo A, Haun S, et al. (2003) Randomized trial of rate-control versus rhythm-control in persistent atrial fibrillation: the Strategies of Treatment of Atrial Fibrillation (STAF) study. J Am Coll Cardiol. 41:1690-1696.
- Hagens VE, Rienstra M, Van Veldhuisen DJ, Crijns HJ, Van Gelder IC (2006) Determinants of sudden cardiac death in patients with persistent atrial fibrillation in the rate control versus electrical cardioversion (RACE) study. Am J Cardiol. 98:929-932.
- Shariff N, Desai RV, Patel K, Ahmed MI, Fonarow GC, et al. (2013) Rate-control versus rhythm-control strategies and outcomes in septuagenarians with atrial fibrillation. Am J Med.126:887-893.
- Bush D, Martin LW, Leman R, Chandler M, Haywood LJ (2006) Atrial fibrillation among African Americans, Hispanics and Caucasians: clinical features and outcomes from the AFFIRM trial. J Natl Med Assoc. 98:330-339.
- Zakeri R, Ahluwalia N, Tindale A, Omar F, Packer M, et al. (2023) Long-term outcomes following catheter ablation versus medical therapy in patients with persistent atrial fibrillation and heart failure with reduced ejection fraction. Eur J Heart Fail. 25:77-86.
- Steinberg JS, Sadaniantz A, Kron J, Krahn A, Denny DM, et al. (2004) Analysis of cause-specific mortality in the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) study. Circulation. 109:1973-1980.
- Parkash R, Wells GA, Rouleau J, Talajic M, Essebag V, et al. (2022) Randomized ablation-based rhythm-control versus rate-control trial in patients with heart failure and atrial fibrillation: results from the RAFT-AF trial. Circulation.145:1693-1704.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.