Real-World Usage of Teneligliptin in Indian Patients with Type 2 Diabetes Mellitus: A Cross-Sectional Multicenter Study
by Jayesh Biyani1, Ashwin Subramaniam2, Kunta Kishore Babu3, Supratik Bhattacharya4, Manoranjan Tripathy5, Sumit Bhushan6*, Sanjay Chaudhari6, Rahee Borulkar6, Prajakta Bhosale6, Rujuta Gadkari6, Saiprasad Patil6
1Kalpana Polyclinic, Mumbai, Maharashtra, India.
2Gericare Hospital, Chennai, Tamil Nadu, India.
3Mahima Diabates Center, Hyderabad, Telangana, India.
4Rajarhat Apollo Sugar Clinic, Kolkata, West Bengal, India.
5Kalinga Hospital, Bhubaneshwar, Odisha, India.
6Glenmark Pharmaceuticals Ltd, Mumbai, Maharashtra, India.
*Corresponding author: Sumit Bhushan, Glenmark Pharmaceuticals Ltd, India.
Received Date: 17 March, 2026
Accepted Date: 23 March, 2026
Published Date: 25 March, 2026
Citation: Biyani J, Subramaniam A, Kishore Babu K, Bhattacharya S, Tripathy M, et al. (2026) Real-World Usage of Teneligliptin in Indian Patients with Type 2 Diabetes Mellitus: A Cross-Sectional Multicenter Study. Curr Trends Intern Med 10: 253. DOI: https://doi.org/10.29011/2638-003X.100253
Abstract
Background: Type 2 diabetes mellitus (T2DM) represents a major public health challenge globally and in India. Dipeptidyl peptidase-4 (DPP-4) inhibitors are widely recommended for glycemic management due to their favorable safety profile and low risk of hypoglycemia. Teneligliptin, a novel DPP-4 inhibitor, has gained widespread use in India; however, real-world data describing its prescribing patterns and patient characteristics remain limited. This study aimed to evaluate the clinical utilization of Teneligliptin in Indian patients with T2DM. Methods: This retrospective, cross-sectional, multicenter, non-interventional study analyzed medical records of adult patients with T2DM who were prescribed Teneligliptin (10 or 20 mg once daily) across 64 healthcare centers in India between September 2024 and March 2025. Descriptive statistics were used to summarize the data. Results: A total of 624 patients with T2DM were included. The mean age was 52.13 ± 11.11 years, with a male predominance (67.14%). Teneligliptin was initiated as first-line therapy in 45.19% of treatment-naïve patients, while 49.20% were switched from previous antidiabetic regimens, most commonly sulfonylurea- or metformin-based therapies. Add-on therapy was observed in 21.63% of patients. At initiation, the mean fasting blood glucose, postprandial glucose, and HbA1c were 167.42 ± 48.54 mg/dL, 236.77 ± 62.92 mg/dL, and 7.96 ± 0.94%, respectively. Hypertension (33.33%), obesity (25.32%), and dyslipidemia (18.75%) were the most common comorbidities. Conclusion: This study demonstrates the widespread use of Teneligliptin in India as initial, add-on, or switch therapy in T2DM management. The findings highlight its integration into routine clinical practice, particularly in patients with multiple comorbidities, providing valuable practice-based evidence to inform diabetes management strategies.
Keywords: Type 2 diabetes mellitus; Teneligliptin; DPP-4 inhibitors; Glycemic control; Comorbidities.
Introduction
Diabetes mellitus (DM) is a major global health burden, currently affecting about 1 in 10 adults [1]. According to the World Health Organization, the global population living with diabetes has increased substantially, rising from approximately 200 million in 1990 to around 830 million in 2022 [2]. Type 2 diabetes mellitus (T2DM) is a metabolic disorder marked by chronic hyperglycemia resulting from insulin resistance and relative insulin deficiency [3]. The global prevalence of type 2 diabetes is rising at an alarming pace, affecting populations in both developed and developing nations [4]. In India, it is estimated that approximately 77 million adults aged over 18 years are living with type 2 diabetes [5]. The increasing prevalence of diabetes in India is driven by urbanization, sedentary lifestyles, dietary transitions, obesity, and underlying genetic susceptibility [6].
Persistent hyperglycemia contributes to the development of both microvascular (retinopathy, nephropathy, neuropathy) and macrovascular complications (cardiovascular disease, stroke, peripheral arterial disease), which account for significant morbidity, mortality, and healthcare costs [7]. Optimal glycemic control remains the cornerstone of T2DM management to reduce long-term complications and improve quality of life. To ensure the optimal glycemic control the ADA 2025 guidelines recommend addition of dipeptidyl peptidase-4 (DPP-4) inhibitors [8]. The DPP-4 inhibitors act by prolonging the activity of incretin hormones such as glucagon-like peptide-1 (GLP-1), thereby stimulating insulin release and suppressing glucagon secretion in a glucose-dependent manner [9].
Teneligliptin, a novel DPP-4 inhibitor first developed in Japan, has gained widespread use in Asian countries including India due to its comparable efficacy and safety profile [10]. According to the TREAT-INDIA real-world evidence study, teneligliptin is found to be cost-effective compared with other agents in its class [11]. Its unique “J-shaped anchor-lock domain” structure confers strong binding affinity and prolonged enzyme inhibition, resulting in sustained glycemic control. The pharmacokinetic profile of teneligliptin contributes to its sustained effect. It has a half-life of approximately 24 hours and is metabolized by both cytochrome P450 (CYP) 3A4 and flavin-containing monooxygenase 3 (FMO3) and is also excreted unchanged by the kidneys. This multi-pathway elimination reduces the need for dose adjustment in patients with hepatic or renal impairment and suggests a low potential for drug-drug interactions [12]. The TREAT-INDIA (2020) study has demonstrated the benefits of Teneligliptin as monotherapy or in combination with other antidiabetic drugs such as metformin, sulfonylureas, or SGLT2 inhibitors, with significant reductions in HbA1c, fasting plasma glucose, and postprandial glucose levels [13]. Importantly, Teneligliptin has shown good tolerability in elderly patients and those with renal impairment, conditions frequently encountered in Indian clinical practice [14].
Despite its growing use, real-world evidence on prescribing patterns and patient outcomes in India remains limited. This study aims to bridge the gap by evaluating the clinical utilization of Teneligliptin in Indian patients with T2DM, focusing on demographic characteristics, treatment combinations, lifestyle factors and laboratory parameters. By generating practice-based evidence, the findings are expected to provide valuable insights for clinicians and policymakers.
Materials and Methods
This was a retrospective, cross-sectional, multicenter, non-interventional study designed to evaluate the real-world usage patterns of Teneligliptin 10 mg or 20 mg tablet once daily in adult patients with T2DM across India. The study adhered to the International Council for Harmonization (ICH) guidelines on Good Clinical Practice (GCP), national regulatory requirements, and the STROBE recommendations for observational studies. Independent ethics committee approval was obtained prior to study initiation.
The study was conducted across 64 healthcare centres in India, including tertiary care hospitals, secondary care facilities, and specialized diabetes clinics. Sites were geographically distributed across the northern, southern, eastern, western, and central regions to capture a diverse range of clinical settings. Data were collected over a six-month period from Sep 2024 to March 2025. Medical records of 624 adult patients (≥18 years) with T2DM who had been prescribed Teneligliptin as initial therapy, add-on therapy, or as a switch-over treatment were included. Patients with unavailability of prescription of Teneligliptin or deemed unsuitable by the investigator were excluded. Eligible patients were identified through systematic review of prescription and medical records at participating sites. A convenience sampling approach was used, with consecutive eligible patients included during the data collection period.
The primary variable was the pattern of Teneligliptin prescription (initial, add-on, or switch-over therapy). Secondary variables included sociodemographic characteristics (age, gender, body weight, vital signs); medical history (duration of T2DM, comorbidities, and lifestyle factors); laboratory parameters (glycemic and relevant biochemical markers); and concomitant medications.
Data were retrospectively extracted from validated medical records by trained site personnel using a structured electronic Case Report Form (eCRF) on the Electronic Data Capture (EDC) system. Role-based access and structured training ensured consistency, accuracy, and data security.Medical records of 624 adult patients with T2DM were included. Duration based sampling approach was followed, and all eligible records were included in this study, consistent with the descriptive nature of this observational study.
Descriptive statistics were used. Categorical variables were presented as counts and percentages, while continuous variables were expressed as means ± standard deviations. Percentages were calculated based on available data for each variable. Data quality was ensured through site monitoring, source data verification, query resolution, and internal validation checks within the EDC system performed by independent study managers, in accordance with ICH-GCP and Indian regulatory guidelines.
The study protocol was approved by the Independent Ethics Committee [ECR/386/Indt/MH/2024] on September 28, 2024. The study was conducted in accordance with the Declaration of Helsinki, ICH-GCP, and applicable Indian regulations for non-interventional research. Independent ethics committee approval was obtained prior to study initiation.
Results
A total of 624 patient medical records were included from 64 clinical centres across India. All patients fulfilled the inclusion criterion of having been prescribed the fixed-dose combination (FDC) of teneligliptin plus metformin for the management of T2DM. A total of 624 patients with type 2 diabetes mellitus (T2DM) were included in the study. The mean age was 52.13 ± 11.11 years, with a male predominance (67.14%) compared to females (32.85%). The average height and weight were 160.34 ± 13.30 cm and 74.00 ± 11.83 kg, respectively. The mean systolic blood pressure was 135.61 ± 15.88 mmHg, the diastolic blood pressure was 88.75 ± 11.10 mmHg, and the average pulse rate was 84.29 ± 9.42 bpm. The mean duration of diabetes was 5.53 ± 4.94 years. The demographic and clinical characteristics of the study population are summarized in (Table 1).
|
Parameter |
Mean ± SD |
|
Age (years) |
52.13 ± 11.11 |
|
Gender n (%) Male Female |
|
|
419 (67.14) |
|
|
205 (32.85) |
|
|
Height (cm) |
160.34 ±13.30 |
|
Weight (kg) |
74.00 ±11.83 |
|
SBP (mmHg) |
135.61 ± 15.88 |
|
DBP (mmHg) |
88.75 ± 11.10 |
|
PR (bpm) |
84.29 ± 9.42 |
|
Duration of DM (years) |
5.53 ± 4.94 |
|
DBP, diastolic blood pressure; SBP, systolic blood pressure; PR, pulse rate. Data represented as Mean ± SD, otherwise specified. |
|
Table 1: Demographics and baseline characteristics of patients (N=624)
Primary Variable: Usage of Teneligliptin
The primary endpoint analysis revealed distinct prescribing patterns for teneligliptin across various outpatient departments in India. Among the 624 patients analyzed, 282 were treatment-naïve (45.19%) at the time of Teneligliptin initiation, whereas 307 (49.20%) were switched from other antidiabetic therapies (Figure 1). The most common prior regimens before switching included sulfonylurea plus metformin (60, 9.61%), metformin alone (53, 8.49%), glimepiride (51, 8.17%), sulfonylurea alone (36, 5.78%), and sitagliptin (29, 4.65%). Less frequent prior therapies included voglibose combined with glimepiride and metformin (10, 1.60%) and other miscellaneous therapies (29, 4.64%). Teneligliptin was prescribed as an add-on treatment option in 135 (21.63%) patients. Of which 107 (17.15%) received it as the first add-on, 20 (3.20%) as the second add-on, and 8 (1.29%) as the third add-on antidiabetic drug (Table 2).
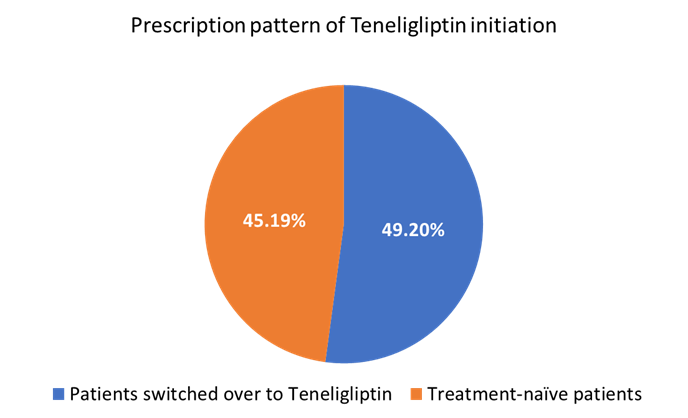
Figure 1: Usage of Teneligliptin initiation among patients with T2DM as an add-on to ongoing diabetic medication
|
Parameter |
Category |
n (%) |
|
Teneligliptin treatment initiation |
Treatment-naive patients |
282 (45.19) |
|
Patients switched over to Teneligliptin |
307 (49.20) |
|
|
Missing details |
35 (5.61) |
|
|
Therapies received prior to switching to Teneligliptin |
Sulfonylurea + Metformin |
60 (9.61) |
|
Metformin |
53 (8.49) |
|
|
Glimepiride |
51 (8.17) |
|
|
Sulfonylurea |
36 (5.78) |
|
|
Sitagliptin |
29 (4.65) |
|
|
Voglibose + Glimepiride + Metformin |
10 (1.60) |
|
|
Others |
29 (4.64) |
|
|
As an add–on to ongoing anti-diabetic treatment |
Yes |
135 (21.63) |
|
1st add-on |
107 (17.15) |
|
|
2nd add-on |
20 (3.20) |
|
|
3rd add-on |
8 (1.29) |
|
|
Data represented as n (%), otherwise specified |
||
Table 2: Treatment Initiation and Prior Therapies
Secondary Variables
Glycemic parameters at initiation of Teneligliptin
Table 2 shows that, at the time of Teneligliptin prescription, the mean FBG was 167.42 ± 48.54 mg/dl, the mean PPG was 236.77 ± 62.92 mg/dl, and the mean HbA1c was 7.96 ± 0.94%. The mean total duration of T2DM was 8.85 months. Continuous glucose monitoring was performed in 95 (15.22%) patients, among whom 59 (9.45%) demonstrated glycemic variability.
|
Parameter |
Mean ± SD (N=624) |
|
Mean FBG (mg/dl) |
167.42 ±48.54 |
|
Mean PPG (mg/dl) |
236.77 ±62.92 |
|
Mean HbA1c (%) |
7.96 ±0.94 |
|
Mean total duration of T2DM (months) |
8.85 Months |
|
Continuous glucose monitoring performed in past n (%) |
95 (15.22) |
|
Glycemic variability observed n (%) |
59 (9.45) |
|
FBG, fasting blood glucose; HbA1c, glycated hemoglobin; PPG, postprandial blood glucose; T2DM, type 2 diabetes mellitus. Data represented as Mean ± SD, otherwise specified |
|
Table 2: Mean glycemic parameters of T2DM patients at the time of prescription with Teneligliptin.
Baseline Laboratory and Clinical Parameters of Patients
The mean value of the urine albumin-creatinine ratio (UACR) was found to be 39.56 ± 42.18 mg/g, whereas serum creatinine (mg/dl) was 1.49 ± 2.78 mg/dl. The mean values of clinical and other biochemical parameters in study participants at the time of initiation of Teneligliptin are summarized in (Table 3).
|
Parameter |
Mean ± SD |
|
UACR (mg/g) |
n = 251 39.56 ± 42.18 |
|
Sr. Creatinine (mg/dl) |
n = 293 1.49 ± 2.78 |
|
BUN (mg/dl) |
n = 223 17.31 ± 8.76 |
|
Sr. Total Cholesterol (mg/dl) |
n = 308 196.09 ± 55.73 |
|
LDL (mg/dl) |
n = 274 103.37 ± 47.84 |
|
HDL (mg/dl) |
n = 273 59.63 ± 32.79 |
|
Sr. Triglycerides (mg/dl) |
n = 273 212.72 ± 190.18 |
|
ALT (U/L) |
n = 149 31.28 ± 25.06 |
|
AST (U/L) |
n = 149 27.92 ± 22.54 |
|
Sr. Bilirubin (mg/dl) |
n = 143 1.13 ± 0.82 |
|
QT/QTc interval (msec) |
n = 138 334.24 ± 130.21 |
|
ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; HDL, high density lipoprotein; LDL, low density lipoprotein; QT/QTc, corrected QT interval; Sr., serum; UACR, urine albumin-creatinine ratio. Data represented as Mean ± SD |
|
Table 3: Laboratory and Clinical Parameters of Patients at the Time of Teneligliptin Prescription
Medical History and Comorbidities of patients on teneligliptin
Figure 2 shows that hypertension was the most prevalent comorbidity, reported in 208 (33.33%) patients, followed by obesity, 158 (25.32%) and dyslipidemia, 117 (18.75%). Ischemic heart disease was observed in 36 (5.77%) patients, while chronic kidney disease was present in 33 (5.29%) patients. Obstructive sleep apnoea was identified in 29 (4.65%) patients, and other comorbid conditions were reported in 17 (2.72%) patients.
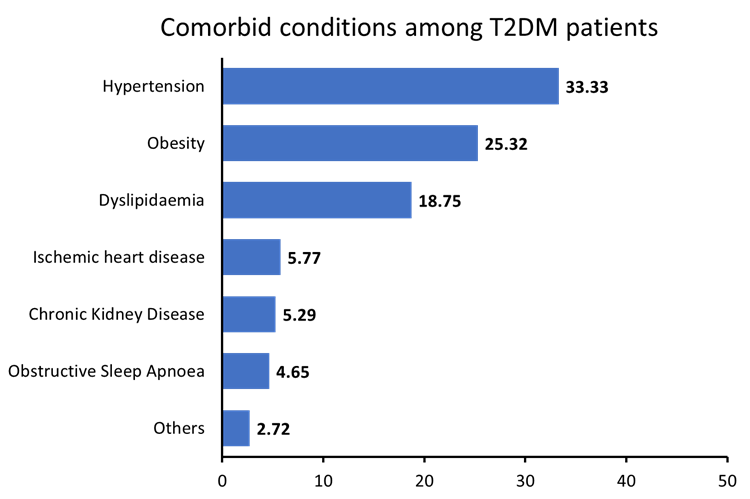
Figure 2: Distribution of comorbid conditions among T2DM patients prescribed Teneligliptin
Regarding past medical history, diabetic neuropathy was reported in 93 (14.90%) patients, while diabetic retinopathy was noted in 82 (13.14%) patients. A total of 67 (10.73%) patients had a history of hypoglycemic episodes, and 26 (4.16%) reported proteinuria. Liver disease was documented in 25 (4.00%) patients, whereas myocardial infarction (MI) and stroke/transient ischemic attack were reported in 23 (3.69%) and 12 (1.44%) patients, respectively. None of the patients had a history of dialysis.
Concomitant Medications and Lifestyle modifications
Lifestyle modification was recommended for 297 (47.60%) patients, whereas 327 (52.40%) did not receive any lifestyle-related counselling. Among 297 patients, dietary recommendations were provided to 159 (25.48%) patients, followed by physical activity [113 (18.11%)], yoga [18 (2.88%)], and meditation [7 (1.12%)]. (Figure 3) depicts that antidiabetic drugs were the most commonly prescribed concomitant medications, with metformin being the most frequent (157, 25.16%), followed by glimepiride (35, 5.61%) and glimepiride + metformin (25, 4.60%). Among cardiovascular agents, telmisartan was the most prescribed (77, 12.34%), followed by telmisartan + hydrochlorothiazide (15, 2.40%) and telmisartan + amlodipine (9, 1.44%). For dyslipidemic agents, atorvastatin (17, 2.72%) and rosuvastatin (14, 2.24%) were the most commonly used. The details of concomitant medications are summarized in (Table 4).
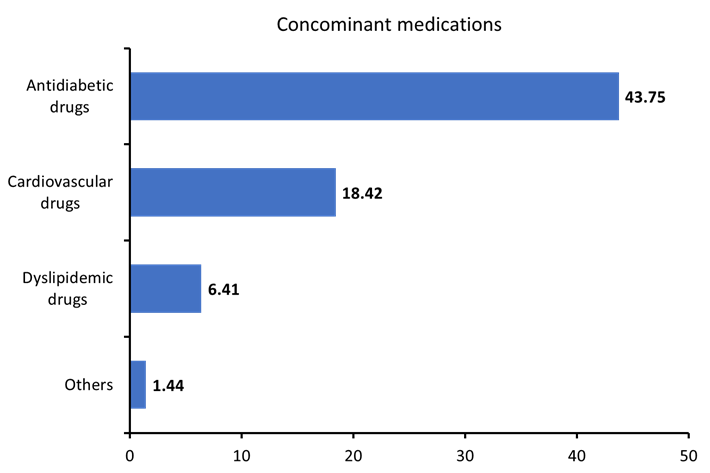
Figure 3: Distribution of concomitant medications prescribed with Teneligliptin
|
Concomitant Medication |
n (%) |
|
Antidiabetic drugs |
273 (43.75) |
|
Metformin |
157 (25.16) |
|
Glimepiride |
35 (5.61) |
|
Glimepiride + Metformin |
25 (4.60) |
|
Gliclazide + Metformin |
14 (2.24) |
|
Sitagliptin + Dapagliflozin + Metformin |
11 (1.76) |
|
Sulfonylureas |
9 (1.44) |
|
Others |
22 (3.52) |
|
Cardiovascular drugs |
115 (18.42) |
|
Telmisartan |
77 (12.34) |
|
Telmisartan + Hydrochlorothiazide |
15 (2.40) |
|
Telmisartan + Amlodipine |
9 (1.44) |
|
Others |
14 (2.24) |
|
Dyslipidemic drugs |
31 (4.96) |
|
Rosuvastatin |
14 (2.24) |
|
Atorvastatin |
17 (2.72) |
|
Others |
9 (1.44) |
Table 4: Concomitant medications prescribed to T2DM patients with Teneligliptin
Discussion
This retrospective, multicentric, real-world study offers valuable insights into the clinical use of Teneligliptin in managing T2DM among Indian patients. The findings demonstrate its widespread adoption across diverse clinical settings, both as initial therapy in treatment-naïve patients (45%) and as a switch strategy in previously treated individuals (49.20%). The study population, with a mean age of 52.13 years and a male predominance (67.14%), mirrors the epidemiology of T2DM in India. Nearly half of patients-initiated Teneligliptin early in their disease course (mean duration, 5.53 years), reflecting a growing preference for DPP-4 inhibitors as first-line or early add-on therapy, which aligns with global ADA guidelines recommending these agents for patients requiring therapies with minimal hypoglycemic risk [8].
Teneligliptin offers a strong pharmacological rationale for its growing use in Indian patients with T2DM. As a highly selective DPP-4 inhibitor, it enhances glucose-dependent insulin secretion while suppressing glucagon release, thereby addressing the dual defects of β-cell dysfunction and impaired incretin response that characterize T2DM. Its unique chemical structure with five consecutive rings confers a prolonged 24-hour half-life, enabling convenient once-daily dosing and sustained glycemic control [15,16]. The dual elimination pathway, involving hepatic metabolism via CYP3A4/FMO3 and renal excretion, allows flexible use without dose adjustment in patients with mild-to-moderate renal impairment, an important consideration given the early nephropathy observed in this cohort. These attributes are particularly relevant in this cohort, where 4.1% had proteinuria and the mean serum creatinine was elevated (1.49 mg/dL). Emerging evidence of teneligliptin’s neutral or beneficial renal effects positions it as a rational choice in this patient cohort [17].
Teneligliptin as monotherapy offers clinical advantages by providing effective glycemic control with a low risk of hypoglycemia, weight neutrality, and suitability in patients with renal impairment [8]. Its initiation in 45% of treatment-naïve patients in our cohort reflects an increasing preference for early use of DPP-4 inhibitors, consistent with recommendations that support individualized therapy beyond metformin, particularly when tolerability, history, or comorbidities influence treatment choice. This approach addresses therapeutic inertia, enables earlier achievement of glycemic targets, and offers a pragmatic solution in resource-constrained healthcare settings. Comparable prescribing patterns have been observed in large real-world studies, including the TREAT-INDIA registry of 4,305 patients, which reported significant HbA1c and PPG reductions with Teneligliptin, and the TREAT-INDIA-2 study of 10,623 patients, which confirmed consistent glycemic improvements across diverse Indian populations [11,13]. Teneligliptin was prescribed to nearly half of treatment-naive patients as initial therapy, while 49.20% were switched from existing regimens, most commonly metformin or sulfonylurea combinations. Real-world evidence from the TREAT-INDIA registry (n = 4,305) supports this trend, demonstrating significant glucose control across diverse patient populations. In the TREAT-INDIA study, 186 (4.32%) patients received Teneligliptin as monotherapy, while 3,858 (89.60%) patients received it in combination with other agents [11].
The baseline glycemic profile in this cohort highlights a considerable gap in achieving recommended targets at the time of teneligliptin initiation. The mean HbA1c (7.96%), FPG (167.4 mg/dL), and PPG (236.8 mg/dL) were all above ADA-recommended thresholds, underscoring suboptimal glycemic control and the presence of therapeutic inertia in routine practice [18]. Importantly, glycemic variability was observed in nearly half of the patients (62.10%) on continuous glucose monitoring, a parameter increasingly recognized as a key determinant of diabetic complications. Evidence shows that fluctuations in glucose levels increase the odds of complications with an increased risk of mortality, cardiovascular disease, renal disease, and peripheral neuropathy [19]. This highlights the clinical relevance of teneligliptin, which provide consistent glucose control.
The baseline laboratory and clinical parameters in this cohort of T2DM patients provide a comprehensive overview of metabolic, hepatic, renal, and cardiovascular status. The findings indicate suboptimal lipid control, with values exceeding recommended targets (LDL <55 mg/dL and triglycerides <150 mg/dL), highlighting the need for more aggressive lipid management. These baseline abnormalities further underscore the importance of implementing renoprotective strategies in this population [20].
Patient characteristics highlight the substantial burden of comorbidities, with hypertension, obesity, and dyslipidemia commonly observed, alongside complications such as neuropathy and retinopathy, reflecting the chronicity of T2DM. A notable proportion reported hypoglycemic episodes (10.73%), likely due to prior sulfonylurea. Concomitant prescribing patterns, including metformin, glimepiride, and telmisartan, underscore the dual need to manage both glycemic and cardiovascular risks [21]. The relatively low use of a combination of newer agents may reflect cost barriers or limited access in this setting, a common challenge in resource-limited healthcare systems. These findings suggest that teneligliptin provides effective glycemic control with minimal risk of hypoglycemia, renal safety, and suitability for use in patients with multiple comorbidities [22].
A key strength of this study is its large sample size and comprehensive characterization of a real-world T2DM cohort, providing valuable insights into Teneligliptin’s prescribing patterns and patient profiles. Future prospective, comparative studies are suggested to evaluate Teneligliptin’s long-term impact on glycemic control, cardiovascular and renal outcomes, safety, and cost-effectiveness in diverse Indian populations, alongside strategies to improve lifestyle counselling and access to newer therapies.
Study Limitations
The cross-sectional design precludes assessment of temporal relationships and clinical outcomes. The retrospective data collection may have introduced selection bias. The study was sponsored by the manufacturer of the studied medication, which could potentially influence site selection and data interpretation. The absence of a control group limits comparative effectiveness assessments.
Conclusion
This study highlights the widespread and growing use of Teneligliptin in India for the management of T2DM, where it is employed as initial, add-on, or switch-over therapy, particularly in patients with comorbidities and renal impairment, and is frequently prescribed in combination with metformin and cardiovascular medications, reflecting a pragmatic, patient-centred approach to addressing the complex challenges of diabetes care. The demographic comorbidities and clinical patterns observed in this study underscore the increasing integration of Teneligliptin into routine clinical practice, providing practice-based evidence to guide clinicians, researchers, and policymakers in optimizing comprehensive diabetes management strategies.
Ethical approval statement
The work presented in this study was in accordance with the study protocol, the New Drugs and Clinical Trials Rules 2019 issued by the Government of India, the ethical principles that have their origin in the Declaration of Helsinki, International Council for Harmonisation (ICH) Good Clinical Practice (GCP), and all applicable local regulatory requirements. Independent Ethics Committee approval was obtained prior to study initiation and data collection
Acknowledgements
We would like to extend our thanks to all the institutes and respective investigators and team members for their support. We also appreciate the CRO support by IR Innovate Research for clinical trial management. We also thank VeritasQ Research and Analytics Pvt. Ltd. for medical writing support in this study.
Funding
This study was funded by Glenmark Pharmaceuticals Limited. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interest
Dr Sumit Bhushan, Dr Sanjay Chaudhari, Dr Rahee Borulkar, Ms Prajakta Bhosale, Ms Rujuta Gadkari and Dr Saiprasad Patil are employees of Glenmark. All other investigators/authors have no conflicts of interest that are directly relevant to the content of this article.
References
- International Diabetes Federation (2025) IDF Diabetes Atlas: Global diabetes data and statistics.
- World Health Organization (2025) Diabetes.
- Damanik J, Yunir E (2021) Type 2 Diabetes Mellitus and Cognitive Impairment. Acta Med Indones 53:213-220.
- Van Dieren S, Beulens JWJ, Van Der Schouw YT, Grobbee DE, Neal B (2010) The global burden of diabetes and its complications: An emerging pandemic. Eur J Cardiovasc Prev Rehabil, 17: S3–S8.
- World Health Organization (2025) Diabetes in India.
- Misra A, Tandon N, Ebrahim S, Sattar N, Alam D, et al. (2017) Diabetes, cardiovascular disease, and chronic kidney disease in South Asia: Current status and future directions. BMJ 357: j1420.
- Mansour A, Mousa M, Abdelmannan D, Tay G, Hassoun A, et al. (2023) Microvascular and macrovascular complications of type 2 diabetes mellitus: Exome wide association analyses. Front Endocrinol (Lausanne) 14: 1143067.
- American Diabetes Association Professional Practice Committee (2025) Pharmacologic approaches to glycemic treatment: Standards of care in diabetes—2025. Diabetes Care 48: S181–S206.
- Ahrén B (2007) DPP-4 inhibitors. Best Pract Res Clin Endocrinol Metab 21: 517–533.
- Kadowaki T, Kondo K (2013) Efficacy, safety and dose-response relationship of teneligliptin, a dipeptidyl peptidase-4 inhibitor, in Japanese patients with type 2 diabetes mellitus. Diabetes Obes Metab 15: 810–818.
- Ghosh S, Trivedi S, Sanyal D, Modi KD, Kharb S (2016) Teneligliptin real-world efficacy assessment of type 2 diabetes mellitus patients in India (TREAT-INDIA study). Diabetes Metab Syndr Obes 9: 347–353.
- Ceriello A, De Nigris V, Iijima H, Matsui T, Gouda M (2019) The unique pharmacological and pharmacokinetic profile of teneligliptin: Implications for clinical practice. Drugs 79: 733–750.
- Ghosh S, Tiwaskar M, Chawla R, Jaggi S, Asirvatham A, et al. (2020) Teneligliptin real-world effectiveness assessment in patients with type 2 diabetes mellitus in India: A retrospective analysis (TREAT-INDIA 2). Diabetes Ther 11: 2257–2268.
- Sharma SK, Panneerselvam A, Singh KP, Parmar G, Gadge P, et al. (2016) Teneligliptin in management of type 2 diabetes mellitus. Diabetes Metab Syndr Obes 9: 251–260.
- Goda M, Kadowaki T (2013) Teneligliptin for the treatment of type 2 diabetes. Drugs Today (Barc) 49: 615–629.
- Kutoh E, Hirate M, Ikeno Y (2014) Teneligliptin as an initial therapy for newly diagnosed drug-naïve subjects with type 2 diabetes. J Clin Med Res 6: 287–294.
- Shah K (2017) Teneligliptin in early diabetic kidney disease: An observation in Asian Indian patients with type 2 diabetes mellitus in real-life scenario. J Clin Diagn Res 11: OC22–OC25.
- American Diabetes Association Professional Practice Committee (2025) Improving care and promoting health in populations: Standards of care in diabetes—2025. Diabetes Care 48: S14–S26.
- Krinsley JS (2008) Glycemic variability: A strong independent predictor of mortality in critically ill patients. Crit Care Med 36: 3008–3013.
- KDBH Lipid Management (2025) Lipid management guidelines.
- Schmieder RE, Bakris G, Weir MR (2011) Telmisartan in incipient and overt diabetic renal disease. J Nephrol 24: 263–273.
- Kadowaki T, Inagaki N, Kondo K, Nishimura K, Kaneko G, et al. (2018) Long-term safety and efficacy of canagliflozin as add-on therapy to teneligliptin in Japanese patients with type 2 diabetes. Diabetes Obes Metab 20: 77–84.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.