Real World Usage of Teneligliptin and Metformin Fixed Dose Combination among Indian Patients with Type 2 Diabetes Mellitus in Indian Clinical Practice
by Sanjay Arora1, Geetha Lakshmi2, Anisha Shah3, Deepak Kapoor4, Arjun Baidya5, Sumit Bhushan6*, Sanjay Choudhari6, Rahee Borulkar6, Amey Kamble6, Saiprasad Patil6
1Arora Diabetic Clinic, New Delhi, India
2Dr. Kamakshi Memorial Hospitals, Chennai, India
3Gada Clinic, Mumbai, India
4Diabeater Sugar Specialty Hospital, Hyderabad, India
5NRS Medical College & Hospital, Kolkata, India
6Glenmark Pharmaceuticals ltd, India
*Corresponding author: Sumit Bhushan, Glenmark Pharmaceuticals Limited, Andheri (E), Mumbai – 400 099, India.
Received Date: 02 April, 2026
Accepted Date: 08 April, 2026
Published Date: 10 April, 2026
Citation: Arora S, Lakshmi G, Shah A, Kapoor D, Baidya A, et al. (2026) Real World Usage of Teneligliptin and Metformin Fixed Dose Combination among Indian Patients with Type 2 Diabetes Mellitus in Indian Clinical Practice. Curr Trends Intern Med 10: 255. DOI: https://doi.org/10.29011/2638-003X.100255
Abstract
Despite the growing use of teneligliptin and metformin FDC in India, there is a significant research gap concerning its real-world usage and effectiveness within the diverse Indian population. This retrospective study aimed to evaluate real-world usage of FDC of teneligliptin and metformin in patients with T2DM who were prescribed Zita Met®. from data in medical records of Indian clinics. Demographic & detailed clinical profile of eligible patients was recorded. The primary assessment was the evaluation of treatment and clinical usage patterns of TEN/MET FDC among the patients. Independent ethics committee approval was obtained prior to study initiation. A total of 2487 patients were included in the study with a mean age of 53.57 ± 10.78 years, majority (67.95%) were males. The mean duration of diabetes was 5.55 ± 5.33 years, and frequent comorbidities were cardiovascular disease, obesity, dyslipidaemia, chronic kidney disease and anxiety/depression. Among the patients receiving TEN/MET FDC, 46.3% of the patients were treatment naïve and the remaining 53.7% had switched from other antidiabetics. Of those switched from other antidiabetics, 17.7% were previously taking monotherapy, ~32% were taking dual FDC and 10.8% were on triple FDC. Among the switchers, metformin was the most commonly prescribed antidiabetic agent closely followed by glimepiride, both with average duration of use about 3.5 years. The findings suggest that the Teneligliptin & Metformin FDC is a relevant therapeutic option for a diverse patient population, including those with significant comorbidities and those who are treatment naïve and those who are transitioning from other treatments.
Keywords: Teneligliptin, Metformin, Indian Patients, Type 2 Diabetes Mellitus
Background
Type 2 Diabetes Mellitus (T2DM) represents a major public health challenge globally, and its burden is particularly pronounced in India, which is often referred to as the "Diabetes capital of the world"[1]. With an estimated 89.8 million adults (20-79 Years) living with T2DM which is projected to exceed 156.7 million by 2050, there is a significant rise in morbidity, mortality, and economic strain [2]. The high prevalence of T2DM in India, often presenting at a younger age and lower body mass index (BMI) compared to Western populations, underscores the urgent need for effective and accessible treatment strategies to achieve optimal glycaemic control and prevent long-term complications [1,3]. The management of T2DM often requires a multi-faceted approach, with metformin being a cornerstone of initial treatment due to its proven efficacy, safety profile, and affordability. However, as the disease progresses, monotherapy often becomes inadequate to maintain glycaemic targets, necessitating the addition of other antidiabetic agents [4]. The FDC of teneligliptin and metformin provides a powerful and convenient dual-action therapy. Teneligliptin, a dipeptidyl peptidase-4 (DPP-4) inhibitor, enhances glucose-dependent insulin secretion and suppresses glucagon levels, while metformin reduces hepatic glucose production and improves insulin sensitivity. This complementary mechanism of action helps achieve significant reductions in HbA1c, fasting plasma glucose, and postprandial glucose levels, with a low risk of hypoglycemia and a weight-neutral effect, thus improving patient adherence and clinical outcomes [5,6,7]. Further, a combination of metformin and teneligliptin has been shown to have additional positive impact such as reduction in bone resorption markers in patients with T2DM [8]. Despite the growing use of teneligliptin and metformin FDC in India, there is a significant research gap concerning its real-world usage and effectiveness within the diverse Indian population. A comprehensive assessment of the prescription and treatment patterns of this FDC is therefore needed to understand how it is being utilized, and identify patient characteristics that influence its use. In this study, we aimed to evaluate the real-world usage of FDC of teneligliptin and metformin in Indian patients with T2DM.
Methods
This multi-centre, retrospective study collected data from the medical records of outpatients with T2DM across 467 Indian clinics. Clinical data was collected using a predesigned structured proforma and entered into an electronic Case Report Form. The study was conducted in accordance with applicable regulations, Good Clinical Practices, and Standard Operating Procedures. All documents, including the protocol and the data collection format, related to this retrospective study were submitted to the Independent Ethics Committee, and the study was conducted as per the approved protocol.
Patients who were prescribed Zita Met® (Teneligliptin 20 mg + Metformin 500 mg /1000 mg) FDC (henceforth referred to as TEN/MET FDC) by treating physician were included in the study. Any patients with incomplete medical records or missing key data were excluded. The demographic and clinical characteristics, including medical history, concomitant medications, and the duration of diabetes, of patients taking TEN/MET FDC were recorded. The primary assessment was the evaluation of treatment and clinical usage patterns of TEN/MET FDC among the patients. Descriptive statistics were used to analyse all results. Categorical variables were presented as counts and percentages, and continuous variables were reported as means with standard deviations.
Results
A total of 2487 patients were included with a mean age of 53.57 ± 10.78 years, majority (67.95%) were males (Table1). The mean duration of diabetes was 5.55 ± 5.33 years, and mean systolic and diastolic blood pressure (BP) was 136.42 ± 22.27 and 93.38 ± 26.60, respectively. About one fifth of the patients were current smokers or tobacco users. Nearly half (46.88%) of the patients had family history of T2DM. More than 55% of the patients had a history of hypoglycaemic event in the recent one year.
|
Characteristic |
Statistics (N=2487) |
|
Age, Years |
53.57 ± 10.78 |
|
Gender, n (%) |
|
|
Male |
1,690 (67.95) |
|
Female |
782 (31.44) |
|
Missing |
15 (0.61) |
|
Body Weight, Kg |
77.02 ± 13.83 |
|
Height, cm |
164.60 ± 10.58 |
|
Blood Pressure, mmHg |
|
|
Diastolic |
93.38 ± 24.60 |
|
Systolic |
136.42 ± 22.27 |
|
Pulse Rate, bpm |
79.52 ± 12.74 |
|
Duration of T2DM, Years |
5.55 ± 5.33 |
|
Smoking or tobacco use, n (%) |
|
|
Yes |
526 (21.15) |
|
Quit |
103 (4.14) |
|
Smoking since, years |
11.06 (7.43) |
|
History of any other substance abuse, n (%) |
87 (3.50) |
|
Substance abuse since, Years |
9.53 ± 5.95 |
|
Other Complaints |
|
|
Sleep, n (%) |
429 (17.25) |
|
Sleep Complaint Since, Years |
2.28 ± 2.65 |
|
Appetite, n (%) |
267 (10.74) |
|
Appetite Complaint Since, Years |
2.94 ± 2.29 |
|
Bowel, n (%) |
218 (8.77) |
|
Bowel Complaint Since, Years |
3.35 ± 3.97 |
|
Bladder/urinary, n (%) |
98 (3.94) |
|
Bladder/urinary complaint Since, Years |
2.79 ± 5.85 |
|
Family History of T2DM, n (%) |
1166 (46.88) |
|
Relevant Medical history, n (%) |
|
|
Limitation of physical activity |
389 (15.64) |
|
Hyperosmotic hyperosmolar coma |
64 (2.57) |
|
Lactic acidosis |
59 (2.37) |
|
Hospitalizations due to any causes in past year |
175 (7.03) |
|
Hypoglycemia in past 1 year |
|
|
Number of patients, n (%) |
1382 (55.56) |
|
Number of episodes |
0.82 ± 1.83 |
|
All data are presented as mean±SD, unless otherwise mentioned. bpm, beats per minute; T2DM, type 2 diabetes mellitus. |
|
Table 1: Demographics and Clinical Characteristics of Patients Prescribed with TEN/MET FDC
Among patients who were prescribed TEN/MET FDC, most common comorbid conditions were cardiovascular disease (32.36%), and obesity (31.60%), each prevalent in almost one third of the patients (Table 2). Other frequent comorbid conditions were dyslipidaemia (17.45%), chronic kidney disease (14.56%) and anxiety/depression (13.59%).
|
Comorbidities |
Number of patients, n (%) |
|
Cardiovascular disease |
805 (32.36) |
|
Chronic kidney disease |
362 (14.56) |
|
Dyslipidemia |
434 (17.45) |
|
Obesity |
786 (31.60) |
|
Obstructive sleep apnea |
128 (5.15) |
|
Anxiety/Depression |
338 (13.59) |
|
Osteoporosis |
104 (4.18) |
|
Others |
87 (3.5) |
Table 2: Comorbidities among Patients Prescribed with TEN/MET FDC (N = 2487)
The use of concomitant medications among the study population has been depicted in (Figure 1). Antihypertensives were the most frequent concomitant medication among the study population with nearly half of the patients using them, followed by antidyslipidaemic and antidiabetic drugs other than TEN/MET FDC. Among antihypertensives, angiotensin receptor blockers were most commonly prescribed. The use of antacids and multivitamins was also noted among the patients.
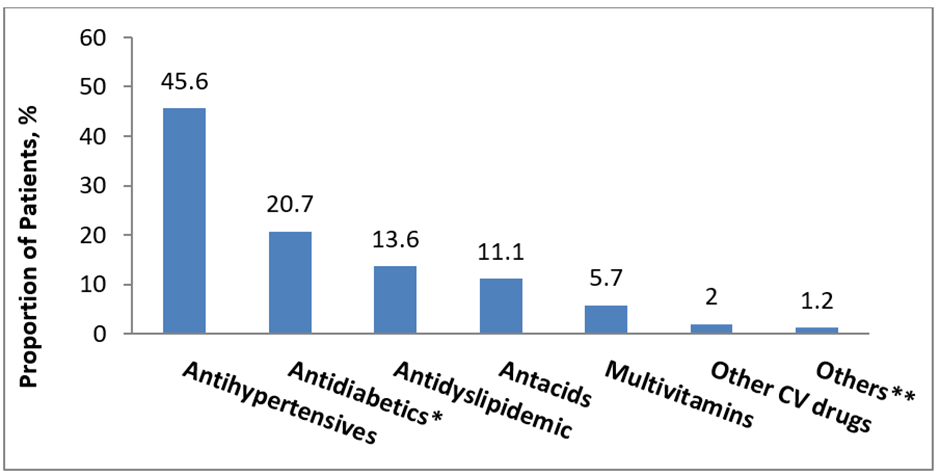
*Other than TEN/MET FDC;** antibiotics and anticonvulsants.
CV, cardiovascular; FDC, fixed dose combination.
Figure 1: Concomitant Medications of Patients Prescribed with TEN/MET FDC (N=1340)
Overall, 46.3% (n=1072/2314) of the patients were treatment naïve and the remaining 53.7% (n=1242/2314) switched to TEN/MET FDC from other antidiabetics. Of those switched, 17.7% (n=220/1242) were previously taking monotherapy, ~32% (n=397/1242) were taking dual FDC and 10.8% (n=134/1242) were on triple FDC (Figure 2).
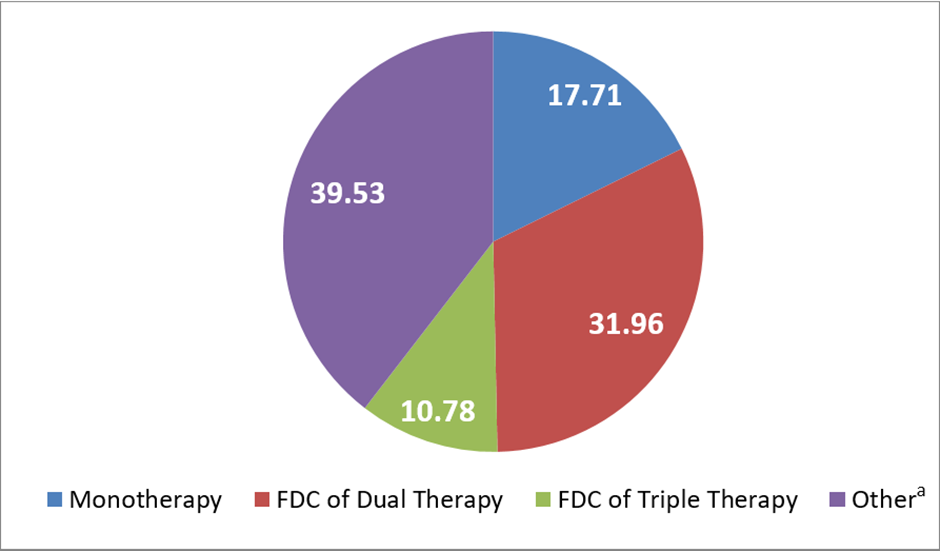
aRepresents use of multiple antidiabetics outside FDCs.
FDC, fixed dose combination.
Figure 2: Treatment Patterns among Patients Switching to TEN/MET FDC (N=1242)
Among the patients who switched to TEN/MET FDC, metformin was the most commonly prescribed antidiabetic agent, used by 78.9% of patients with an average duration of use of 3.72 ± 3.48 years (Table 3). It was most frequently administered once daily (OD) or twice daily (BID) at mean daily dose of 645.61 ± 247.74 mg. Glimepiride followed closely, prescribed to 63.0% of patients, at a mean dose of 1.69 ± 0.70 mg per day, with a similar duration of use of 3.50 ± 3.06 years. Among DPP-4 inhibitors, sitagliptin (21.9%), teneligliptin (13.1%), and linagliptin (2.7%) were commonly prescribed. Patients on sitagliptin had a mean daily dose of 76.07 ± 91.31 mg. Those on teneligliptin had shorter mean duration of use of 1.46 ± 3.09 years with a mean dose of 20.12±2.20 mg. Sodium-glucose cotransporter-2 (SGLT2) inhibitors such as dapagliflozin (16.4%), canagliflozin (0.6%), and empagliflozin (0.3%) were also utilized, with dapagliflozin being the most frequent, given at a mean dose of 9.87 ± 1.83 mg OD. Alpha-glucosidase inhibitors including voglibose (11.2%) and acarbose (3.7%) were used less frequently, mostly in OD or BID regimens. Pioglitazone was prescribed for 6.2% of patients, often in OD regimen, with a mean dose of 14.73 ± 1.42 mg. The least commonly used agents included remogliflozin (1.9%) and other antidiabetic drugs (included insulin, sitagliptin + metformin, vildagliptin, vildagliptin + metformin (1.5%).
|
Drug |
Proportion of patients, n (%) |
Dose (mg) |
Frequency, n (%) |
Duration of Use (Years), Mean (SD) |
||
|
N=1242 |
OD |
BID |
TID |
|||
|
Metformin |
980 (78.9) |
645.61±247.74 |
413 (16.61) |
329 (13.23) |
13 (0.52) |
3.72±3.48 |
|
Glimepiride |
783 (63.0) |
1.69±0.70 |
364 (14.64) |
210 (8.44) |
9 (0.36) |
3.50±3.06 |
|
Acarbose |
46 (3.7) |
37.50±17.68 |
9 (0.36) |
-- |
-- |
3.00±1.73 |
|
Voglibose |
139 (11.2) |
0.25±0.05 |
82 (3.30) |
28 (1.13) |
3 (0.12) |
2.93±2.68 |
|
Sitagliptin |
272 (21.9) |
76.07±91.31 |
164 (6.59) |
19 (0.76) |
-- |
1.79±1.52 |
|
Teneligliptin |
163 (13.1) |
20.12±2.20 |
159 (6.39) |
4 (0.16) |
-- |
1.46±3.09 |
|
Linagliptin |
33 (2.7) |
5.00±0.00 |
25 (1.01) |
-- |
-- |
4.67±2.42 |
|
Dapagliflozin |
204 (16.4) |
9.87±1.83 |
147 (5.91) |
5 (0.20) |
-- |
2.38±1.91 |
|
Canagliflozin |
8 (0.6) |
100.00±4.02 |
7 (0.28) |
1 (0.04) |
-- |
5.00±0.00 |
|
Empagliflozin |
4 (0.3) |
25.00±0.00 |
2 (0.08) |
-- |
-- |
6.00±0.00 |
|
Remogliflozin |
23 (1.9) |
100.00±0.00 |
11 (0.44) |
2 (0.08) |
-- |
2.50±1.08 |
|
Pioglitazone |
77 (6.2) |
14.73±1.42 |
54 (2.17) |
5 (0.20) |
-- |
3.14±2.92 |
|
Other Drugs* |
19 (1.5) |
-- |
-- |
-- |
-- |
-- |
|
*used by ≤0.4% of patients, include insulin, sitagliptin+metformin, vildagliptin, vildagliptin+metformin. BID, twice a day; OD, once a day; TID, thrice a day. |
||||||
Table 3: Antidiabetic Treatment Patterns among Patients Switched to TEN/MET FDC
Discussion
This study aimed to characterize the demographic and clinical profile of patients prescribed the TEN/MET FDC, as well as to describe the real-world treatment patterns observed in outpatient departments of Indian clinics. The findings demonstrate that the study population, consisting of patients who were either treatment-naive or switching from other therapies, presented with a diverse range of clinical characteristics, including a notable prevalence of specific comorbidities and history of substance or nicotine use. These data provide valuable insights into the type of patient for whom this FDC is being utilized in clinical practice.
The patients with T2DM included in our study had high prevalence of cardiovascular comorbidities, obesity, hypertension, dyslipidemia, and frequent hypoglycaemia that aligns with known profiles of patients with T2DM in India, reinforcing the real-world applicability of this study. For instance, recent Indian data showed even newly diagnosed patients are often already at “high” or “very high” risk for ASCVD, with hypertension and smoking being common risk factors [9]. Similarly, a rural South Indian study reported a hypoglycemia prevalence of ~57.4%, with ~10.7% experiencing severe episodes [10]. These align closely with our observations of ~55.6% patients with hypoglycemia experience in past one year. The prevalence of these comorbidities aligns with the frequent use of concomitant medications including cardiovascular, antihypertensive and antidyslipidemic drugs.
The clinical usage of antidiabetic agents observed in this study reflected real-world prescribing practices across the T2DM spectrum. A high proportion of treatment-naïve patients taking TEN/MET FDC is reflective of a shift towards early combination therapy and aligns with clinical findings demonstrating clinical benefits of early combination therapy in T2DM [11]. Predominant reliance on metformin and glimepiride, alongside growing use of DPP-4 inhibitors like sitagliptin and teneligliptin corroborates with clinical results reported in literature. Our observation that majority of patients who were prescribed TEN/MET FDC were previously taking metformin aligns with the evidence supporting the efficacy and tolerability of add-on teneligliptin in patients inadequately controlled on metformin monotherapy [12-14]. The noted widespread use of glimepiride only second to metformin may be reflective of its cost-effectiveness in Indian practice. While metformin-glimepiride combination demonstrated greater cost-effectiveness, evidence suggests that teneligliptin offers comparable glycemic efficacy, particularly in post-prandial glycemic control, along with additional benefit of weight neutrality and a lower risk of hypoglycaemia [15]. These attributes make teneligliptin a valuable and promising option in patients having concerns of sulphonylurea-related side effects. The analysis of prescription patterns revealed that the FDC is being prescribed to both patients initiating therapy and those switching from existing treatments. The observation that a significant percentage of patients were treatment-naive suggests that clinicians may be considering this FDC as a first-line option. The data on patients switching to the FDC is also particularly insightful, as it may indicate a need to address possible issues such as inadequate efficacy, poor tolerability, or treatment complexity with prior regimens. This finding underscores the clinical utility of the FDC in optimizing treatment for patients at all stages of diabetes treatment continuum who have not achieved their goals on other medications. This study has its limitations which should be considered. First, the data are subject to the inherent limitations of medical record abstraction. Second, the study did not assess the glycemic parameters which limit the assessment of treatment effectiveness, appropriateness and clinical rationale behind prescription patterns in patients with T2DM.
Study Limitations: The cross-sectional design precludes assessment of temporal relationships and clinical outcomes. The retrospective data collection may have introduced selection bias. The study was sponsored by the manufacturer of the studied medication, which could potentially influence site selection and data interpretation. The absence of a control group limits comparative effectiveness assessments. Mention about short duration, less number of participant.
Conclusions
This study provides a detailed characterization of the patient population receiving the TEN/MET FDC and sheds light on its real-world usage patterns. The findings suggest that the FDC is a relevant therapeutic option for a diverse patient population, including those with significant comorbidities and at different stages of their T2DM treatment journey, including both treatment naïve and those who are transitioning from other treatments. These insights contribute to a better understanding of the role of TEN/MET FDC in clinical practice.
Ethical standards
The work presented in this study was in accordance with the study protocol, the New Drugs and Clinical Trials Rules 2019 issued by the Government of India, the ethical principles that have their origin in the Declaration of Helsinki, International Council for Harmonisation (ICH) Good Clinical Practice (GCP), and all applicable local regulatory requirements. Independent Ethics committee approval was obtained prior to study initiation and data collection.
Acknowledgments
We would like to extend our thanks to all the institutes and respective investigators and team members for their support. We also appreciate the CRO support by IR Innovate Research Pvt Ltd, India for clinical trial management. We also thank Lifecom Scientia for medical writing support in the study.
Funding: This study was funded by Glenmark Pharmaceuticals Limited. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interest: Sumit Bhushan, Rahee Borulkar, Amey Kamble, Sanjay Choudhari and Saiprasad Patil are employees of Glenmark. All other investigators/authors have no conflicts of interest that are directly relevant to the content of this article
References
- Borgharkar SS, Das SS (2019) Real-world evidence of glycemic control among patients with type 2 diabetes mellitus in India: the TIGHT study. BMJ Open Diabetes Res Care. 7:e000654.
- International Diabetes Federation (2025) IDF Diabetes Atlas. 11th ed. Brussels, Belgium: International Diabetes Federation; 2025.
- Anjana RM, Unnikrishnan R, Deepa M, Pradeepa R, Tandon N, et al. (2023) Metabolic non-communicable disease health report of India: the ICMR-INDIAB national cross-sectional study (ICMR-INDIAB-17). Lancet Diabetes Endocrinol. 11: 474-489.
- American Diabetes Association Professional Practice Committee (2024) 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2024. Diabetes Care, 47:S158–S178.
- Zheng J, Woo SL, Hu X, Botchlett R, Chen L, et al. (2015) Metformin and metabolic diseases: a focus on hepatic aspects. Front Med. 9:173-186.
- Ghosh S, Trivedi S, Sanyal D, Modi KD, Kharb S (2016) Teneligliptin real-world efficacy assessment of type 2 diabetes mellitus patients in India (TREAT-INDIA study). Diabetes Metab Syndr Obes. 9:347-353.
- Ji L, Li L, Ma J, Li X, Li D, et al. (2021) Efficacy and safety of teneligliptin added to metformin in Chinese patients with type 2 diabetes mellitus inadequately controlled with metformin: A phase 3, randomized, double-blind, placebo-controlled study. Endocrinol Diabetes Metab. 4:e00222.
- Shaik AR, Kohli S, Vohora D (2024) Bone effects of metformin monotherapy and its combination with teneligliptin: A 12-week follow-up study in patients with type 2 diabetes mellitus. Diabetes Res Clin Pract. 213:111744.
- Unnikrishnan AG, Sahay RK, Phadke U, Sharma SK, Shah P, et al. (2022) Cardiovascular risk in newly diagnosed type 2 diabetes patients in India. PLoS One. 17:e0263619.
- Samya V, Shriraam V, Jasmine A, Akila GV, Anitha Rani M, et al. (2019) Prevalence of Hypoglycemia Among Patients With Type 2 Diabetes Mellitus in a Rural Health Center in South India. J Prim Care Community Health. 10:1-7.
- Chudasama DB, Saboo BD, Panchal D, Patil FP, Saiyed M, et al. (2018) The efficacy of teneligliptin with metformin in Drug‑Naïve type 2 subjects. Diabetes. 67-1205.
- Kim MK, Rhee EJ, Han KA, Woo AC, Lee MK, et al. (2015) Efficacy and safety of teneligliptin, a dipeptidyl peptidase-4 inhibitor, combined with metformin in Korean patients with type 2 diabetes mellitus: a 16-week, randomized, double- blind, placebo-controlled phase III trial. Diabetes Obes Metab. 17:309-12.
- Bryson A, Jennings PE, Deak L, Paveliu FS, Lawson M (2016) The efficacy and safety of teneligliptin added to ongoing metformin monotherapy in patients with type 2 diabetes: a randomized study with open label extension. Expert Opin Pharmacother. 17:1309-16.
- Das AK, Gandhi P, Saboo B, Reddy S, Chawla R, et al. (2021) Optimizing the treatment of newly diagnosed type 2 diabetes mellitus with combination of dipeptidyl peptidase‑4 inhibitors and metformin: An expert opinion. J Family Med Prim Care. 10:4398-409.
- Tandon T, Dubey AK, Srivastava S, Manocha S, Arora E, et al. (2019) A pharmacoeconomic analysis to compare cost-effectiveness of metformin plus teneligliptin with metformin plus glimepiride in patients of type-2 diabetes mellitus. J Family Med Prim Care. 8:955-959.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.