Novoglan Device May Rescue Advanced Phimosis After Failed Medical Therapy
by Zhong Li Titus Lim1*, Alice Nicol1, Jason Kim2, Eric Chung3, Handoo Rhee4
1Department of Urology, Princess Alexandra Hospital 199 Ipswich Road, Woolloongabba, Queensland, Australia 4102
2Department of Urology, Gold Coast University Hospital Blvd, Southport QLD 4215 University of Queensland St Lucia, Queensland, Australia
3Department of Urology, Princess Alexandra Hospital 199 Ipswich Road, Woolloongabba, Queensland, Australia 4102
4Department of Urology, Princess Alexandra Hospital 199 Ipswich Road, Woolloongabba, Queensland, Australia 4102 Australia and New Zealand Dialysis and Transplant Registry, South Australian Health and Medical Research Institute (SAHMRI), Adelaide, South Australia, Australia
*Corresponding Author: Zhong Li Titus Lim, Department of Urology, Princess Alexandra Hospital, 199 Ipswich Road, Woolloongabba, Queensland, Australia
Received Date: 09 March 2026
Accepted Date: 13 March 2026
Published Date: 16 March 2026
Citation: Lim ZLT, Nicol A, Kim J, Chung E, Rhee H (2026) Novoglan Device May Rescue Advanced Phimosis After Failed Medical Therapy. J Surg 11: 11580 DOI: https://doi.org/10.29011/2575-9760.011580
Abstract
Background: Phimosis, whether congenital or acquired, affects a significant proportion of adult males in Australia. While manual manipulation with topical treatments remains a common strategy, a subset of patients experience persistent or progressive disease necessitating surgical intervention. Circumcision is effective, although associated with potential long-term complications including meatal stenosis, altered penile sensitivity and suboptimal cosmetic outcomes. Consequently, there is growing interest in non-surgical alternatives. This prospective study evaluates the clinical utility of the Novoglan device in patients referred for circumcision at a tertiary hospital following failed conservative management with medical therapies.
Methods: A single-arm, open-label prospective trial was conducted at the Princess Alexandra Hospital (Brisbane, Australia), enrolling 24 adult males with persistent phimosis unresponsive to standard conservative therapies. Ethics approval was obtained (HREC: PR/2023/QMS/66682). Participants were offered an 8-week trial of the Novoglan device whilst awaiting circumcision. Baseline and post-treatment assessments included phimosis grading, symptom burden, and quality of life metrics across domains such as pain, sexual function, daily activities, and psychological well-being.
Results: Of the 24 enrolled participants, 20 completed the study with 4 withdrawing due to personal reasons. The mean age of participants was 34.05 ± 12.48 years. Comorbidities included diabetes mellitus (n=6, 30%) and genital psoriasis (n=2, 10%). Balanitis Xerotica Obliterans (BXO) was clinically diagnosed in 70% of cases. Following 8 weeks of Novoglan use, 75% of participants reported improvement in foreskin retractability and symptom relief. Quality of life scores improved across all measured domains. Notably, 20% of patients avoided circumcision altogether, and 80% indicated they would recommend the device to others.
Conclusion: In a cohort of patients with refractory phimosis, including those with suspected BXO, the Novoglan device demonstrated clinically meaningful improvements in symptoms and quality of life. Importantly, it enabled a subset of patients to avoid circumcision, highlighting its potential role as a salvage therapy in public health settings where surgical resources are limited or patient preference favors conservative management.
Keywords: BXO; Circumcision; Novoglan; Phimosis
Introduction
Adult phimosis is common, whether congenital or acquired, affecting approximately 3.4% of male population[1]. Due to the inability to retract the foreskin, it may impact voiding and sexual function, with complications such as pain, irritation, splitting, infection and progression of phimosis. In rare circumstances, it can be associated with penile cancer with up to 75% of penile cancers associated with phimosis in certain parts of the world [2]. While topical corticosteroids and antimicrobial agents remain the first-line therapies, a subset of patients experience persistent or progressive disease necessitating surgical intervention. Circumcision, although effective, is associated with potential complications including meatal stenosis, altered penile sensitivity, and suboptimal cosmetic outcomes [3, 4]. It may impact quality-of-life activities such as sexual function [5] and activities of daily living. Consequently, there is growing interest in non-surgical alternatives [6]. Devices such as Novoglan, Phimostop or Vajraang Phimosis stretching rings are developing more interest and popularity. There is also growing interest in novel medical treatment such as platelet-rich plasma therapy. The Novoglan device is a novel device that incorporates a handheld system designed to gently and evenly stretch the prepuce, guided by patient’s preference, progress and need. It consists of a small self-inflatable balloon, a guide rod, a stopcock, and an inflation bulb. The balloon is inserted beneath the prepuce using the guide rod, then inflated with the bulb and left in place for a set period of time. The device is typically used twice daily with the degree of dilatation set by the patent’s comfort level, and the response is generally demonstrable between 2 and 8 weeks [7]. A pilot study in Australia has shown promising results [8] as an initial treatment, with 90% of patient reporting improvement in foreskin retraction. This prospective study evaluates the clinical utility of the Novoglan device in patients referred for circumcision following failed conservative management at a tertiary referral metropolitan hospital.
Methods
A single-arm, open-label prospective trial was conducted at the Princess Alexandra Hospital (Brisbane, Australia), enrolling 24 adult males with persistent phimosis unresponsive to standard conservative therapies. Ethics approval was obtained (HREC: PR/2023/QMS/66682). Participants were offered an 8-week trial of the Novoglan device while awaiting circumcision. Baseline and post-treatment assessments included phimosis grading, symptom burden, and quality of life metrics across domains such as pain, sexual function, daily activities, and psychological well-being. Statistical analyses, grafts and figures were performed using Excel (Microsoft Corporation, Office 365, 2026)
Results
Visit Schedule: Participants attended four scheduled visits following enrollment. Participants were initially screened for suitability for the trial, and enrolled after a week to deliberate on the consent process and to obtain sufficient training. At the first visit, participants were assessed to confirm comprehension of study requirements and correct use of the investigational product. Their second visit at week 2 was focused on evaluation of techniques, treatment progress and monitoring for adverse events. A subsequent third review was conducted at week 5 to assess ongoing efficacy and safety. The final study visit occurred between weeks 7 and 9, during which comprehensive assessments and quality of life scores were completed and study participation concluded.
Demographics: The study enrolled 24 participants. 4 of these patients withdrew throughout the trial due to personal reasons. Of the remaining 20 participants, the mean age was 34.05 years (SD ±12.48) and all participants were of Caucasian ethnicity. The mean Body Mass Index (BMI) was 30.84 kg/m² (SD ±9.03). BMI distribution and relevant medical history are summarized in (Tables 1 and 2) and (Figure 1). 70% of patients exhibited features of BXO on the foreskin with the classic signs of pale thickened scarred unretractable foreskin.
|
Characteristic |
Value |
|
Age (years) |
34.05 ± 12.48 |
|
Ethnicity |
100% Caucasian |
|
BMI (kg/m²) |
30.84 ± 9.03 |
|
BMI Categories |
>40: 4 |
|
35–39: 2 |
|
|
30–34: 3 |
|
|
25–30: 4 |
|
|
<25: 7 |
Table 1: Participant Demographics.
|
Condition |
Number of Participants |
|
Diabetes Mellitus (DM) |
6 |
|
Crohn’s Disease |
1 |
|
Psoriasis |
1 |
|
Balanitis Xerotica Obliterans (BXO) |
14 |
Table 2: Relevant Medical History.
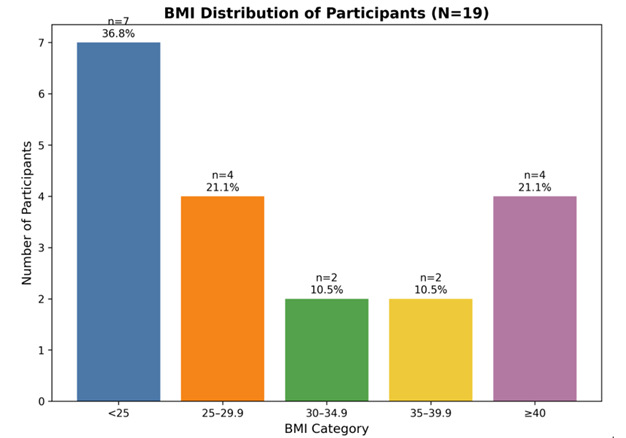
Figure 1: BMI distribution.
Phimosis Severity at Baseline: Phimosis severity was categorized into six levels. These included: (1) absolutely no retraction, (2) slight retraction leaving a gap, (3) partial retraction sufficient to see the meatus, (4) partial retraction exposing part of the glans (5) full retraction of the foreskin, but tight behind the glans, and (6) full and free retraction of the foreskin, no tightness behind the glans. At baseline, most participants fell within categories 2 (n=8), 3 (n=6), and 4 (n=6). No participants were in categories 1, 5, or 6. (Table 3) shows the distribution of phimosis severity at baseline.
|
Category |
Description |
Pre-treatment |
Post-treatment |
||
|
n |
% |
n |
% |
||
|
-1 |
Absolutely no retraction |
0 |
0 |
0 |
0 |
|
-2 |
Slight retraction leaving a gap between the tip of the prepuce and the glans |
8 |
40 |
1 |
5 |
|
-3 |
Partial retraction, just sufficient to see the glandular meatus |
6 |
30 |
3 |
15 |
|
-4 |
Partial retraction, exposing part of the glans |
6 |
30 |
9 |
45 |
|
-5 |
Full retraction of the foreskin, tight behind the glans |
0 |
0 |
6 |
30 |
|
-6 |
Full and free retraction of the foreskin, no tightness behind the glans |
0 |
0 |
1 |
5 |
Table 3: Phimosis Severity(N-20).
Treatment Progress and Adverse Events: At the second visit, pain was reported by 3/20 participants, itching by 1/20, foreskin discoloration by 2/20, discomfort by 3/20, and splitting by 1/20. Sixteen participants (80%) felt their condition was progressing well. By the third visit, pain remained at 3/20, itching resolved (0/20), discoloration decreased to 1/20, discomfort persisted (3/20), and splitting resolved (0/20). Again, 16 participants reported good progress. Finally, at the last visit, mean phimosis score improvement was 1.25 ± 0.94. Itching was described by 1/20, discoloration by 1/20, discomfort by 3/20, and splitting by 0/20. Adverse events across visits are summarized in (Figure 2). Four participants (20%) did not require circumcision after treatment, and 15 participants (75%) stated they would recommend the intervention.
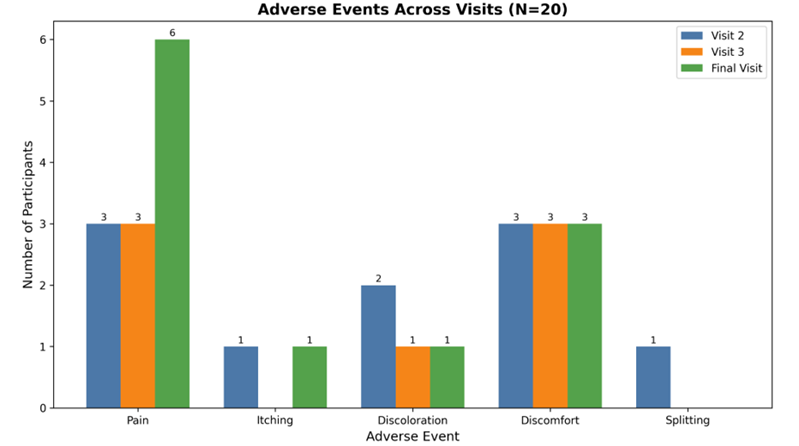
Figure 2: Adverse events noted at each visit.
Quality of Life Benefits: Quality of life parameters, including pain and discomfort during sexual activity, impact on daily activities, and anxiety, were assessed in this study. Prior to use of the device, 9 of 20 participants reported no pain or discomfort during sexual activity. Mild pain or discomfort was reported by 2/20 participants, moderate pain by 4/20, and severe pain by 1/20. Four participants did not provide a response. One participant reported a mild impact of phimosis on daily activities, and 2/20 participants reported an impact on anxiety related to their condition. Following use of the device, overall pain and discomfort during sexual activity improved. Fourteen of 20 participants reported no pain or discomfort, 2/20 reported mild pain, and 2/20 reported moderate pain. One participant reported increased pain following device use; notably, this individual also experienced worsening pain during device application due to overstretching of the foreskin. There was no change in the impact on daily activities, and 2/20 participants continued to report a mild impact of phimosis on their anxiety post-intervention.
With respect to overall satisfaction, 15/20 participants reported that they would recommend the device, 1/20 would not recommend it, 3/20 provided no response, and 1/20 expressed uncertainty.
Discussion
In this prospective study, a significant subset of patients demonstrated improvement with the use of the Novoglan device over the study period. Importantly, in a public hospital setting where surgical procedure associated resources are limited, 20% of patients were able to avoid circumcision with associated known complications. In most patients, the phimosis score improved, allowing for some relief. 30% reported ongoing pain and discomfort, with some cases worsened by the overuse of Novoglan device. Ultimately, 15 out of the 20 participants recommended the product. Adverse events were generally mild and transient. Itching and splitting were mainly resolved by the third visit. The observed improvement on quality-of-life scores and patient symptom scores suggest that the intervention may be an effective conservative option for patients with moderate phimosis, particularly where surgical management is not preferred due to medical comorbidities or personal reasons or carries additional risks such as in those with diabetes or on treatment with anticoagulation. Another example would be a patient deemed high risk for anaesthesia. Avoidance of circumcision in one in five participants is clinically relevant, potentially reducing operative morbidity, anaesthetic exposure, and resource utilization. The stable or resolving nature of most adverse events, alongside high patient-reported progress at the second and third visit, supports acceptable tolerability. Some patients reported persistence in discomfort and increased pain at the final visit (6/20), reflecting poor technique such as hyperintensity of balloon dilatation, transient irritation from the device, mild reaction, or concurrent conditions (e.g., BXO). It is also likely that the self-reported dilatation of the foreskin was more aggressive than the initial. This should be taken into consideration during patient counselling and follow‑up. For patients presenting with moderate phimosis, particularly those with comorbidities or preferences to avoid surgery, these findings support offering conservative treatment with clear expectations regarding symptom trajectories and the possibility of avoiding circumcision.
It should be noted that BXO in itself is not a predictor of treatment failure with Novoglan. In this study, of 13 patients with clinical diagnosis of BXO (pale white thickened or sclerotic glans and foreskin), three were able to avoid circumcision. With regards to quality of life, the participants reported overall improvement in pain or discomfort with sexual activity after using the Novoglan device. The device did not significantly impact activities of daily living or change in anxiety levels. While circumcision remains definitive for pathologic phimosis, conservative strategies-such as topical therapies, behavioural stretching regimens, or device-assisted approaches-are frequently considered for selected cases [9]. These strategies include topical corticosteroids [10], platelet-rich plasma therapy [11] and Phimostop [12]. According to the Cochrane review, topical corticosteroids significantly improved complete or partial clinical resolution of phimosis compared to placebo or no treatment (12 studies, 1,395 participants: RR 2.45, 95% CI 1.84 to 3.26). These findings suggest corticosteroids are an effective non-surgical option for managing phimosis. However, the review noted that adverse effects were poorly reported across studies, limiting the ability to fully assess the safety profile of this intervention. In contrast, the use of platelet-rich plasma therapy and phimosis stretching rings are relatively new. Platelet-rich plasma therapy for treating phimosis is a relatively recent development, and current evidence is limited by small sample sizes and the absence of randomized controlled trials. Similarly, in the prospective trial for Phimostop, there were low sample sizes and 50% of the study’s participants still required surgical management post treatment. Lastly, no journal articles are available for the Vajraang phimosis ring which is heavily advertised on popular online shopping platforms such as Amazon. Despite these short-comings, non-surgical techniques may play a role in patients who have considerable co-morbidities or avoidance. The strengths of this study include the ability to research a subset of patients with phimosis who failed conservative therapy in a prospective manner. Over 8 weeks on average, we were able to closely monitor the techniques and progress whilst the patients were awaiting for circumcision, and thereby ensuring that all patients received optimal and standard care whilst participating in the clinical trial. Detailed patient reported outcomes and quality of scores allowed for accurate capture of relevant information.
Several limitations should be acknowledged. The sample size was modest (n = 20), which limits statistical precision and precludes meaningful subgroup analyses, such as comparisons between BXO and non‑BXO or diabetic and non‑diabetic patients. The single‑centre design and ethnically homogeneous cohort (all participants were Caucasian) restrict generalisability to more diverse populations. In addition, the absence of a control group and non‑blinded outcome assessments prevent causal inference and may introduce expectation bias. The relatively short follow‑up period, with the final visit occurring at Weeks 7–9, may not capture longer‑term durability of response, recurrence rates, or late adverse events. As a result, a larger randomized controlled trial with diverse populations in a multi-centre setting comparing Novoglan and other non-surgical managements should be considered. Longitudinal follow-up to evaluate durability, recurrence and late adverse events should be included in this trial.
Conclusion
In a cohort of patients with refractory phimosis, including those with suspected BXO, the Novoglan device demonstrated clinically meaningful improvements in symptoms and quality of life. Importantly, it enabled a subset of patients to avoid circumcision, highlighting its potential role as an alternate therapy in public health settings where surgical resources are limited or patient preference favors conservative management.
References
- Morris BJ, Matthews JG, Krieger JN (2020) Prevalence of Phimosis in Males of All Ages: Systematic Review. Urology 135: 124-132.
- Douglawi A, Masterson TA (2017) Updates on the epidemiology and risk factors for penile cancer. Transl Androl Urol 6: 785-790.
- Williams N, Kapila L (1993) Complications of circumcision. Br J Surg 80: 1231-1236.
- Holman JR, Stuessi KA (1999) Adult circumcision. Am Fam Physician 59: 1514-1518.
- Tian Y (2013) Effects of circumcision on male sexual functions: a systematic review and meta-analysis. Asian J Androl 15: 662-666.
- George M (2025) Prepuce-Preserving Management of Phimosis: Therapeutic Options, Evidence and Clinical Relevance for Urologists. Trends in Urology & Men's Health 16: e70003.
- ( 2025) Novoglan: Treatment Instructions,” Novoglan 2025.
- Chung E (2023) Novoglan device for treatment of adult phimosis: Novoglan-01 open-label clinical trial on safety, efficacy and tolerability. Transl Androl Urol 12: 1050-1061.
- Rosato E (2024) Phimosis in Adults: Narrative Review of the New Available Devices and the Standard Treatments. Clin Pract 14: 361-376.
- Moreno G (2014) Topical corticosteroids for treating phimosis in boys. Cochrane Database Syst Rev 2014: Cd008973.
- Casabona F (2017) Autologous Platelet-Rich Plasma (PRP) in chronic penile lichen sclerosus: the impact on tissue repair and patient quality of life. Int Urol Nephrol 49: 573-580.
- Carilli M (2021) Can circumcision be avoided in adult male with phimosis? Results of the PhimoStop(TM) prospective trial. Transl Androl Urol 10: 4152-4160.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.