Microscopic Haematuria in Practice: Diagnostic Yield and Adherence to Risk-Based Guidelines in a Contemporary Australian Series
by Nicholas Gillman1*, William Layt1, Christopher Eng2, Joshua Ng2, Chelsea Powell1, Jason Kim1, Alice Mistry1
1Gold Coast University Hospital Department of Urology, 1 Hospital Blvd, Southport QLD 4215, Australia Australia
2Griffith University School of Medicine and Dentistry, 170 Kessels Rd, Nathan QLD 4111, Australia
*Corresponding Author: Nicholas Gillman, Gold Coast University Hospital Department of Urology, Australia.
Received Date: 12 March 2026
Accepted Date: 17 March 2026
Published Date: 19 March 2026
Citation: Gillman N, Layt W, Eng C, Ng J, Powell C, et al. (2026) Microscopic Haematuria in Practice: Diagnostic Yield and Adherence to Risk-Based Guidelines in a Contemporary Australian Series. J Surg 11: 11586 DOI: https://doi.org/10.29011/2575-9760.011586
Abstract
Objective: To quantify real-world adherence to risk-stratified investigation of Microscopic Haematuria (MH), focusing on guideline concordance of upper tract imaging in an Australian cohort, where laboratories report automated cell counts (cells/µL) rather than RBC/HPF.
Methods: A single-centre retrospective cohort study of adults undergoing flexible cystoscopy for MH (March 2023–March 2025) was performed. Demographics, smoking, anticoagulation, RBC counts, and imaging were recorded. Risk was stratified using AUA thresholds, applying a prespecified equivalence (1 RBC/HPF ≈ 4 cells/µL) to derive low/negligible (<40), intermediate (40–100), and high (>100 cells/µL) risk groups. Imaging concordance was defined a priori as none (low), renal ultrasound (intermediate), and CT IVP (high). Outcomes were clinically significant cystoscopic findings (biopsied lesions) and malignancy. Proportions were reported with Clopper-Pearson 95% CIs and Number Needed to Scope (NNS). Patients triaged directly to treatment after positive imaging were not captured.
Results: Among 205 cystoscopies, malignancy was detected in 4.0% (n=8; 95% CI 1.7-7.7; NNS 25). In intermediate-risk patients (n=104), imaging concordance was 28.9%, with CT used in 69.2%, yielding 71.2% misalignment; malignancy rate was 2.9% (95% CI 0.6-8.2; NNS 34.7). In high-risk patients (n=94), concordant imaging occurred in 67.0%; malignancy rate was 5.3% (95% CI 1.7-12). Only 3.5% of cystoscopies were guideline discordant.
Conclusion: In real-world practice, imaging misalignment, not cystoscopy overuse, was the primary quality gap. CT IVP was overused in intermediate risk and underused in high risk. Harmonisation of cell count reporting with RBC/HPF and locally adapted risk-based pathways are needed to support guideline-aligned MH evaluation.
Introduction
Microscopic Haematuria (MH) frequently prompts urologic referral from primary care. It is common, with an estimated prevalence in the population between 2.4% to 31.1% dependant on testing methods and populations screened [1]. There are many aetiologies for MH including infection, urolithiasis, prostatic and renal disease, and while most cases are benign or transient, MH can be an indicator of underlying urothelial malignancy [2]. While the diagnostic evaluation of visible haematuria is well established, variation exists regarding the definition of MH, criteria for referral to urologic services, and appropriate investigations for assessment [3]. Given the risk of underlying urothelial malignancy, historically, patients referred with MH have been evaluated with Computed Tomography (CT) urography and cystoscopy [1]. This broad-brushed approach does not factor individual patient risk, leading to over-investigation in certain lower risk patient population [4]. To avoid over-investigation in low-risk strata while preserving diagnostic accuracy for Urothelial Cancer (UC), the American Urological Association (AUA) published guidelines in 2020 recommending a risk-stratified approach to diagnostic workup, which were updated in 2025. Clinicians are urged to consider demographic factors known to increase malignancy risk including age, sex, smoking history, history of UC, as well as the severity of MH using Red Blood Cell Count Per High-Powered Field (RBC/HPF), to risk stratify patients as low/negligible, intermediate, and high-risk [5]. Appropriate imaging modality for investigation and recommendation to proceed to cystoscopy are based on this risk stratification.
Many Australian centres face a practical challenge when adopting this risk-stratified approach. The AUA guideline s use urine microscopy reported as RBC/HFP in the definition, and risk stratification of MH, while most Australian clinical laboratories use automated urine cell counts reported as cells/µL. Currently, no universally agreed conversion between RBC/HPF and cells/µL exists. Indeed, the only guideline consensus incorporating automated cell counts was published by the Japanese Urological Association by Higashihara in 2008, defining haematuria as >5 RBC/HPF or 20 cells/µL [6]. This proportion was then validated in 2014 by Ichiyanagi providing a rough estimate of 1 RBC/HPF ≈ 4 cells/µL [7]. This conversion however is not typically used in laboratory reports, referral processes or risk calculators. As a result, real-world local concordance may not align with guideline recommendations. There is a paucity of local data and limited real-world yield studies in Australian cohorts using this risk-stratified approach to investigation. Moreover, few studies have examined clinical concordance with risk-based imaging recommendations. The aim of this study is to evaluate outcomes in MH workup with a focus on cancer detection and adherence to guidelines and to determine and quantify whether local gaps in MH care exist by assessing adherence to risk-stratified imaging pathways among patients undergoing flexible cystoscopy for MH at a single Australian centre over a two year period.
Methods
Data was collected for all patients presenting for flexible cystoscopy from March 2023 to March 2025 at a single centre where the primary reason for investigation was MH. Patients were excluded if there was incomplete data, a history of visible haematuria, or previous diagnosis of urothelial malignancy. Demographic data including age at time of flexible cystoscopy, gender, smoking history, and anticoagulant/antiplatelet use was collected, as well as urinary RBC count and imaging modality employed for investigation. Automatic cell counts were used in all patients to assess haematuria severity. Patients were risk stratified following the updated AUA Microscopic Haematuria guidelines (2025). As the AUA, and other international guidelines, use number of Red Blood Cells Per High Powered Field (RBC/HPF) in guideline recommendation, a pre-specified equivalence of 1RBC/HPF ≈ cells/mL based on the Japanese Medical Association panel recommendation was used to derive risk bands (low/negligible <40, intermediate 40-100, high >100 cells/mL). Imaging concordance was determined using the 2025 AUA guidelines as framework. Concordance was defined a priori as no imaging for low/negligible risk patients, renal tract Ultrasound (US) for intermediate risk, and CT Intravenous Pyelogram (CT IVP) for high-risk patients. Cystoscopy concordance was also assessed using the same guideline recommendations. Outcomes were clinically significant cystoscopy findings, defined as any urothelial lesion proceeding to biopsy and rate of malignancy, which was based on histopathologic findings. Proportions were reported with Clopper-Pearson 95% confidence intervals. Number Needed to Scope (NNS) was calculated as n/events. Patients triaged directly to treatment, such as rigid cystoscopy + biopsy, or Transurethral Resection of Bladder Tumour (TURBT), nephroureterectomy, or nephrectomy were not captured in this cohort (Table 1, Figure 1).
|
Characteristics |
n |
% |
|
Demographic Data |
||
|
Number |
205 |
|
|
Male |
87 |
|
|
Female |
118 |
|
|
Age, median (IQR) |
65 (54.5-74) |
|
|
Haematuria Severity (cells/mL) |
||
|
<40 |
77 |
37.56 |
|
40-100 |
77 |
37.56 |
|
>100 |
51 |
24.88 |
|
Smoking History |
||
|
Never |
100 |
48.78 |
|
Former |
70 |
34.15 |
|
Current |
31 |
15.12 |
|
Unknown |
4 |
1.95 |
|
Anticoagulation Use |
||
|
Apixaban |
12 |
5.85 |
|
Rivaroxaban |
10 |
4.88 |
|
Clopidogrel |
4 |
1.95 |
|
Ticagrelor |
1 |
0.488 |
|
Aspirin |
12 |
5.85 |
|
Warfarin |
1 |
0.488 |
|
Nil |
165 |
80.49 |
|
Risk Stratification |
||
|
Not Meeting Definition |
4 |
1.95 |
|
Low/Negligible |
3 |
1.46 |
|
Intermediate |
104 |
50.73 |
|
High |
94 |
45.85 |
Table 1: Demographic Data.
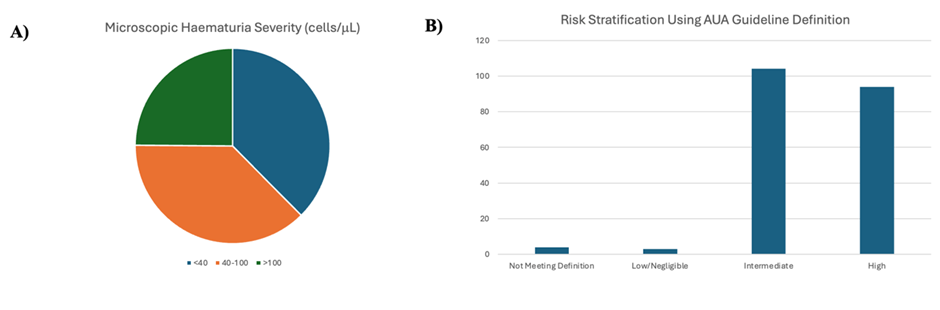
Figure 1: A) Microscopic Haematuria Severity (cells/uL) Derived from AUA Risk Strata. B) Risk Stratification of Patients Presenting with MH for Cystoscopy Derived from AUA Risk Strata.
Results
Between March 2023 and March 2025, 205 patients met inclusion criteria. The cohort included 87 males (42.44%) and 118 females (57.56%). The median age of the cohort was 65 years (IQR 54.24-74.0). Regarding haematuria severity, 77 patients (37.56%) had <40 cells/mL, 77 patients (37.56%) had 40-100 cells/mL and 51 (24.88%) had >100 cells/mL. Based on AUA risk stratification, 4 patients (1.95%) did not meet the definition of MH, 3 (1.46%) were classified as low/negligible risk, 104 patients (50.73%) intermediate risk and 94 (45.85%) high risk. Upper tract imaging selection varied across risk groups. Among the 104 intermediate-risk patients, 65 (62.5%) underwent CT IVP and 30 (28.85) received renal tract ultrasound, representing only a 28.9% concordance with guideline-recommended imaging. Adherence was higher in the high-risk cohort, where 63 of 94 (67.0%) underwent CT IVP and 26 (27.7%) received renal tract ultrasound, with an overall concordance rate of 67.0%. No patients in the low/negligible risk group appropriately met criteria for imaging, although numbers were small. Overall, the data demonstrates over imaging in intermediate-risk patients, and greater alignment with guideline-directed imaging among high-risk patients. As demonstrated in Figure 3, in total, 13 lesions were identified and proceeded to biopsy (overall yield 6.5%, 95% CI 3.5-10.8; NNS 15.5). The intermediate- and high-risk groups demonstrated a yield of 5.8% (6/104; 95% CI 2.1-12.1) and 7.4% (7/94; 95% CI 3.0-14.7) respectively, correcting to NNS values of 17.3 and 13.4. There were no positive cystoscopy findings in the low/negligible risk group. As shown in in Figure 2, a total of eight urothelial malignancies were identified among 201 patients meeting the definition of MH, corresponding to an overall cancer detection rate of 4% (CI 1.7-7.7), and a number needed to scope (NNS) of 25.1 (Table 3). No malignancies were detected in the low/negligible-risk group although numbers were small. The intermediate-risk group demonstrated a malignancy yield of 2.9% (3/104, 95% CI 0.6-8.2) with a NNS 34.7, while the high-risk cohort had the highest detection rate at 5.3% (5/94, 95% CI 1.7-12.0) with a NNS of 18.8. Among the eight urothelial malignancies detected, three were low grade Ta (LgTa) in the intermediate-risk patients. Of the five malignancies detected in the high-risk group, three additional LgTa tumours were identified, as well as one high grade T1 and one case of Carcinoma In Situ (CIS). This data demonstrates that higher risk cancers were confined to the high-risk group, although numbers were small.
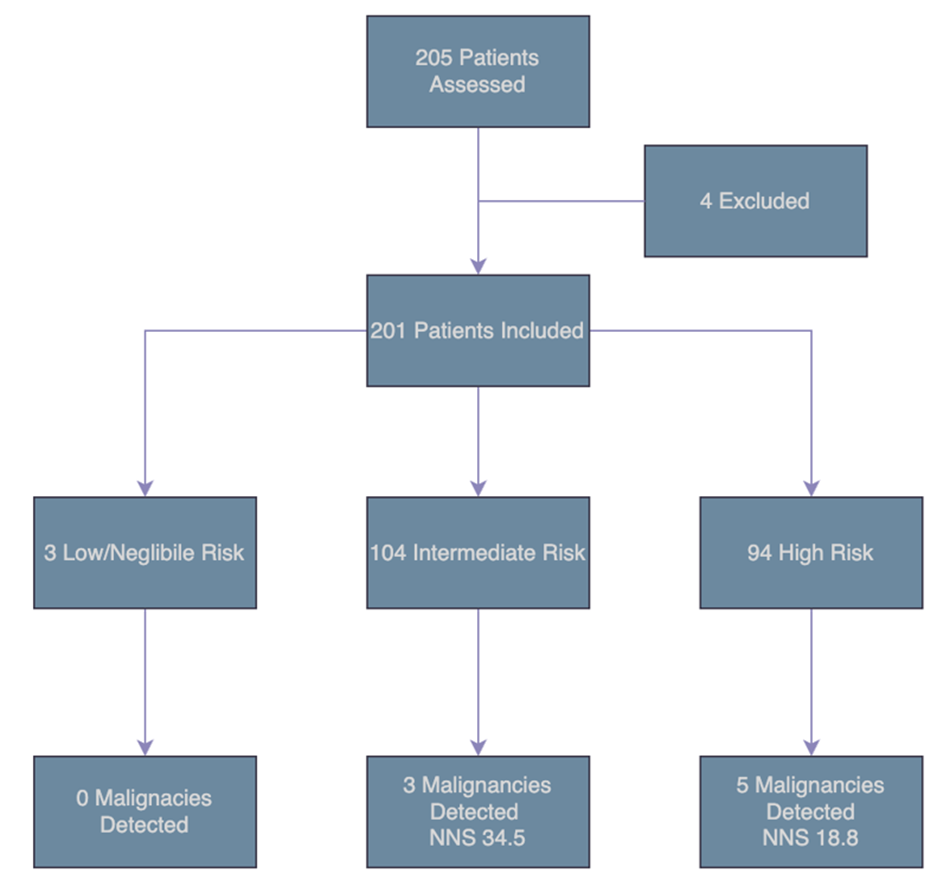
Figure 2: Malignancy Detection and Number Needed to Scope (NNS) in the MH Cohort Stratified by Risk Group.
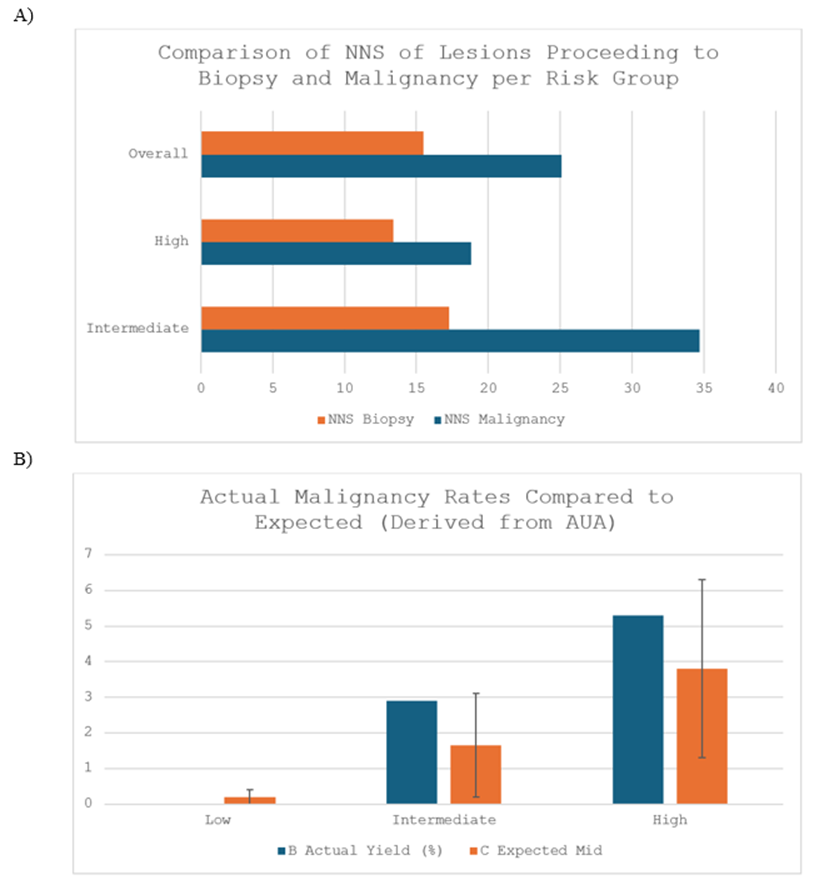
Figure 3: A) Comparison of NNS of Lesions Proceeding to Biopsy and NNS for Detection of Malignancy per Risk Group. B) Comparison of Actual vs Expected Malignancy Rates. Expected Malignancy Rates
|
Risk group |
n |
Malignancies (n) |
Yield % (95% CI) |
NNS |
NNS 95% CI |
|
Low/Negligible |
3 |
0 |
0.0 (0.0–70.8) |
- |
- |
|
Intermediate |
104 |
3 |
2.9 (0.6–8.2) |
34.7 |
12.20–166.98 |
|
High |
94 |
5 |
5.3 (1.7–12.0) |
18.8 |
8.35–57.16 |
|
Overall |
201 |
8 |
4.0 (1.7–7.7) |
25.1 |
13.00–57.68 |
Table 3: Malignancy Yield and NNS Stratified by Risk Group.
Discussion
This study aimed to investigate real world adherence to a risk-stratified approach to diagnostic workup in MH at a single centre in Australia, with an emphasis on AUA guideline alignment. In this audit, we observed a moderate overall diagnostic yield, with malignancy detected in 4.0% of patients. Of note, no cancers were identified in the low/negligible risk group, although numbers in this subgroup were small. Yield increased progressively across risk strata, demonstrating a gradient of disease prevalence, supporting the adoption of risk-based frameworks recommended in the AUA guidelines. Despite this, our findings revealed variation between guideline recommendations and local practice patterns, regarding imaging modality employed for investigation (Table 2). A high proportion of intermediate-risk patients underwent CT IVP, representing over-investigation in this group where US is typically recommended. Meanwhile, CT IVP was underutilised in the high-risk cohort, where guideline concordant practice recommends cross-sectional imaging to account for the higher probability of upper tract urothelial carcinoma. This pattern suggests locally adapted diagnostic pathways are necessary to ensure adherence to risk-stratified decision making. Haematuria is commonly encountered in urologic practice, accounting for up to 20% of evaluations [8]. Despite its high prevalence and association with urothelial malignancy, significant deficiencies exist in the evaluation of patients with haematuria. Johnson found that haematuria prompted urologic evaluation from primary care in less than 50% of patients, with an adjusted hazard ratio 1.65 for male referral compared to female referral [9]. Another study demonstrated that of 15,161 patients with MH, only 8.4% underwent upper tract imaging and cystoscopy [10]. This disparity is unsurprising when one considers the variation in major organisational guideline recommendations on the evaluation and management of patients with MH. Linder et al reviewed international guidelines for asymptomatic microscopic haematuria and demonstrated substantial heterogeneity in both diagnostic thresholds and recommended evaluation pathways [3]. While most organisation support investigation in the absence of benign causes, definitions of MH vary (≥3-5 RBCs/HPF vs dipstick-based criteria), as do age cut-offs for evaluation (35-60 years). While there is consensus that cystoscopy should be performed as part of evaluation, variation exists regarding upper tract imaging. Several guidelines do not state recommendation for upper tract imaging including American College of Physicians [11] and NICE [12] or do not specify method (British Association of Urological Surgeons [13]). Ultrasonography is recommended as first line by the Canadian Consensus Document [14], Japanese Urological Association [15] and Dutch Association of Urology [16]. Previous large retrospective MH cohorts have demonstrated lower diagnostic yields than those observed in the present study. Gonzalez et al demonstrated a 1.2% yield of urothelial carcinoma amongst 2118 patients with a median age of 61 undergoing cystoscopy [17].
|
Risk Category |
n |
US KUB |
CT IVP |
CT AP +C |
CT KUB |
None |
Concordance (%) |
|
Not Meeting Definition |
4 |
2 |
2 |
0 |
0 |
0 |
- |
|
Low/negligible |
3 |
2 |
1 |
0 |
0 |
0 |
- |
|
Intermediate Risk |
104 |
30 |
65 |
3 |
4 |
2 |
28.85 |
|
High Risk |
94 |
26 |
63 |
2 |
2 |
1 |
67.02 |
Table 2: Imaging Modality Employed for Investigation Stratified by Risk Group.
Age and smoking history were found to be significantly associated with risk of malignancy [17]. The group diagnosed with malignancy were found to have a larger number of RBCs/HFP (15.8±28.2) compared to the non-malignancy cohort (7.5±3.2), however did not reach statistical significance. Jung et al demonstrated a similarly low incidence of urothelial carcinoma of 0.68% in a large population-based cohort study of 156,691 patients with MH over a three year follow up period [18]. A 2021 meta-analysis by Waisbrod inlcuding a total of 30 studies comprising ~25,000 patients with MH demonstrated a pooled bladder cancer detection rate of 2.00%. In a subgroup analysis of high-risk patients, defined as groups in which the median age was 60 years or older, more than 50% of patients being male, and/or more than 50% of patients with a smoking history, pooled incidence increased to 4.61%, more closely mirroring the risk-gradient observed in this study [19]. These findings highlight the benefit of a risk straitified approach to management in patients presenting with MH. Cystoscopy remains the gold standard investigation for bladder cancer detection, with superior sensitivity and specificity when compared to other modalities including imaging [20] and urinary biomarkers [21], supporting its use in intermediate- and high-risk MH groups. The role of upper tract imaging as an adjunct to cystoscopy is to identify renal and upper tract urothelial malignancies, as well as identify alternative causes of MH. The overall incidence of upper tract urothelial or renal malignancy in patients with MH is low, estimated to be between 0.1-0.7% [22-24]. CT IVP offers a more sensitive detection of upper tract malignancy compared to US KUB (94% vs 33%) [1,5,24], however confers more risk to the patient secondary to ionising radiation and intravenous contrast reactions, as well as increased health system costs. Notably, in a decision-analysis model by Halpern et al of asymptomatic MH patients, renal ultrasound and cystoscopy detected nearly as many urinary tract cancers as CT IVP and cystoscopy at 1/3 the cost to the healthcare system [25].
An important contextual challenge in applying international guidelines to the Australian settings is variation in methods used to quantify haematuria. The AUA framework relies on RBC/HPF, whereas Australian practice typically employs automated urine microscopy reporting in units of cells/mL. The absence of an established universally accepted conversion between RBC/HPF and automated cell counts introduces uncertainty when mapping patients into guideline-defined risk categories, which require severity of haematuria as part of stratification. The Japanese Urologic Association provided an estimated conversion of 1 RBC/HPF to 4 cells/mL in a guideline publication in 2008 [6]. Applying this estimate to our cohort allowed for meaningful risk stratification, with stepwise enrichment of malignancy across risk strata. These findings suggest the conversion is clinically functional to assist in stratification, however, underscore the need for future local guideline adaptations or equivalency tables that incorporate cell-based thresholds. This would enable more accurate and consistent risk stratifications in regions where automated cell counts predominate. There are several limitations to this study. Primarily, the analysis reflects practice patterns at a single institution, with data collected retrospectively which may limit generalisability. The absence of malignancy in the low/negligible-risk patient group should be interpreted cautiously, as number were small and larger cohort analyses may reveal clinically meaningful cancers. Reliance on electronic medical records introduces vulnerability to incomplete or inaccurate documentation and missing demographic data which may influence risk stratification. Third, although the conversion between automatic cell counts and urine microscopy was clinically functional, this approximation may have misclassified patients across risk groups. Future research should focus on the development of local MH guidelines that directly incorporates automated cell counts rather than relying on conversions derived from international frameworks. Such a guideline could define cell-count-based thresholds that map onto low/negligible, intermediate and high-risk categories, calibrated against Australian laboratory practices. Embedding these thresholds into referral pathways and diagnostic workup would support consistent risk stratification, reduce ambiguity and facilitate guideline-concordance use of cystoscopy and upper tract imaging. The study is subject to selection bias, specifically verification bias, as most patients with positive imaging findings would be fast-tracked to cystoscopy and biopsy and excluded from analysis, leading to under-representation of higher-risk cases.
Conclusion
In this single-centre audit of MH, overall diagnostic yield was modest but clinically meaningful with an overall malignancy detection rate of 4%. Yield increased across risk strata, supporting a risk-stratified approach consistent with AUA guidance. At this centre, there was reasonable concordance with guideline-recommended imaging in the high-risk group, however variation persisted overall, with overuse of CT IVP in inter-mediate risk patients. These findings also highlight the need to standardise urinary red cell reporting in Australia. Variation between cells/µL and RBC/HPF complicates risk classification and may contribute to inconsistent investigation pathways. An equivalent of 4cells/µL to 1 cell per HPF as used in this study can be useful in assisting risk stratification, however local guidelines are required incorporating cells/µL to allow for more accurate risk stratification. Improved adherence to risk-stratified frameworks, optimisation of imaging selection, and harmonised laboratory reporting may enhance the value and efficiency of haematuria in the Australian setting.
References
- Davis R, Jones JS, Barocas DA, Castle EP, Lang EK, Leveillee RJ et al. (2012) Diagnosis, evaluation and follow-up of Asymptomatic Microhematuria (AMH) in adults: AUA guideline. J Urol 188: 2473-81.
- Leslie SW, Hamawy K, Saleem MO (2024) Gross and Microscopic Hematuria. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
- Linder BJ, Bass EJ, Mostafid H, Boorjian SA (2018) Guideline of guidelines: asymptomatic microscopic haematuria. BJU Int 121: 176-183.
- Georgieva MV, Wheeler SB, Erim D, Smith-Bindman R, Loo R, et al. (2019) Comparison of the Harms, Advantages, and Costs Associated With Alternative Guidelines for the Evaluation of Hematuria. JAMA Intern Med 179: 1352-1362.
- Barocas DA, Lotan Y, Matulewicz RS, Raman JD, Westerman ME, et al. (2025) Updates to Microhematuria: AUA/SUFU Guideline (2025). J Urol 213: 547-557.
- Higashihara E, Nishiyama T, Horie S, Marumo K, Mitarai T, et al. (2008) Hematuria: Definition and screening test methods. International Journal of Urology 15: 281-284.
- Ichiyanagi Y (2014) Field Volume of Urine Sediment Test - Comparison of Theoretical Volume with Practical Volume -. Sysmex Journal Internation 24.
- Mariani AJ, Mariani MC, Macchioni C, Stams UK, Hariharan A, et al. (1989) The Significance of Adult Hematuria: 1,000 Hematuria Evaluations Including a Risk-Benefit and Cost-Effectiveness Analysis. The Journal of Urology 141: 350-355.
- Johnson EK, Daignault S, Zhang Y, Lee CT (2008) Patterns of Hematuria Referral to Urologists: Does a Gender Disparity Exist? Urology 72: 498-502.
- Matulewicz RS, Demzik AL, DeLancey JO, Popescu O, Makarov DV, et al. (2019) Disparities in the diagnostic evaluation of microhematuriaand implications for the detection of urologic malignancy. Urologic Oncology: Seminars and Original Investigations 37: 300.e1-.e7.
- Nielsen M, Qaseem A (2016) Hematuria as a Marker of Occult Urinary Tract Cancer: Advice for High-Value Care From the American College of Physicians. Ann Intern Med 164: 488-497.
- Jefferies ER, Brewster SF (2016) Urological recommendations from the National Institute for Health and Care Excellence (NICE) Guideline, June 2015: Suspected cancer: recognition and referral. BJU Int 117: 857-860.
- Anderson J, Goldberg L, Kelly J, MacTier R (2008) Joint consensus statement on the initial assessment of haematuria. Prepared on behalf of the Renal Association and British Association of Urological Surgeons.
- Kassouf W, Aprikian A, Black P, Kulkarni G, Izawa J, et al. (2016) Recommendations for the improvement of bladder cancer quality of care in Canada: A consensus document reviewed and endorsed by Bladder Cancer Canada (BCC), Canadian Urologic Oncology Group (CUOG), and Canadian Urological Association (CUA), December 2015. Can Urol Assoc J 10: E46-80.
- Horie S, Ito S, Okada H, Kikuchi H, Narita I, et al. (2014) Japanese guidelines of the management of hematuria 2013. Clin Exp Nephrol 18: 679-689.
- van der Molen AJ, Hovius MC (2012) Hematuria: a problem-based imaging algorithm illustrating the recent Dutch guidelines on hematuria. AJR Am J Roentgenol 198: 1256-1265.
- Gonzalez AN, Lipsky MJ, Li G, Rutman MP, Cooper KL, et al. (2019) The Prevalence of Bladder Cancer During Cystoscopy for Asymptomatic Microscopic Hematuria. Urology 126: 34-38.
- Jung H, Gleason JM, Loo RK, Patel HS, Slezak JM, et al. (2011) Association of hematuria on microscopic urinalysis and risk of urinary tract cancer. J Urol 185: 1698-1703.
- Waisbrod S, Natsos A, Wettstein MS, Saba K, Hermanns T, et al. (2021) Assessment of Diagnostic Yield of Cystoscopy and Computed Tomographic Urography for Urinary Tract Cancers in Patients Evaluated for Microhematuria: A Systematic Review and Meta-analysis. JAMA Network Open 4: e218409-e.
- Devlies W, de Jong JJ, Hofmann F, Bruins HM, Zuiverloon TCM, et al. (2024) The Diagnostic Accuracy of Cystoscopy for Detecting Bladder Cancer in Adults Presenting with Haematuria: A Systematic Review from the European Association of Urology Guidelines Office. Eur Urol Focus 10: 115-122.
- Sathianathen NJ, Butaney M, Weight CJ, Kumar R, Konety BR (2018) Urinary Biomarkers in the Evaluation of Primary Hematuria: A Systematic Review and Meta-Analysis. Bladder Cancer 4: 353-363.
- Kang M, Lee S, Jeong SJ, Hong SK, Byun S-S, et al. (2015) Characteristics and significant predictors of detecting underlying diseases in adults with asymptomatic microscopic hematuria: A large case series of a Korean population. International Journal of Urology 22: 389-393.
- Samson P, Waingankar N, Shah P, Friedman D, Kavoussi L, et al. (2018) Predictors of genitourinary malignancy in patients with asymptomatic microscopic hematuria. Urol Oncol 36: 10.e1-.e6.
- Fankhauser CD, Waisbrod S, Fierz C, Becker AS, Kranzbühler B, et al. (2021) Diagnostic accuracy of ultrasonography, computed tomography, cystoscopy and cytology to detect urinary tract malignancies in patients with asymptomatic hematuria. World J Urol 39: 97-103.
- Halpern JA, Chughtai B, Ghomrawi H (2017) Cost-effectiveness of Common Diagnostic Approaches for Evaluation of Asymptomatic Microscopic Hematuria. JAMA Intern Med 177: 800-807.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.