Marked Chylous Ascites in a Patient with Transforming Mantle Cell Lymphoma
by Nagle Sheetal1*, Ramakrishna Raj2, Manoharan A2
1Clinical Dietitian, Southern Cross University, Military Rd, East Lismore NSW 2480, Australia
2University of Wollongong, Northfields Ave, Wollongong NSW 2500, Australia
*Corresponding author: Sheetal Nagle, Clinical Dietitian, Southern Cross University, Military Rd, East Lismore NSW 2480, Australia
Received Date: 24 February 2026
Accepted Date: 02 March 2026
Published Date: 04 March 2026
Citation: Sheetal N, Raj R, Manoharan A (2026) Marked Chylous Ascites in a Patient with Transforming Mantle Cell Lymphoma. Ann Case Report. 11: 2546. DOI: https://doi.org/10.29011/2574-7754.102546
Abstract
Chylous ascites is a rare clinical condition resulting from disruption of abdominal lymphatics. Non-Hodgkin’s lymphoma is one of the well- known, if uncommon, causes of chylous ascites [1,2]. In this report, we describe a 74-year-old man with a 15-year history of mantle cell lymphoma (MCL) with IgM paraprotenaemia, who developed marked chylous ascites when the disease transformed to a high proliferative entity.
Case Report
A 74-year man was diagnosed with MCL in 2009 when he presented with minor inguinal lymphadenopathy; histological features were consistent with an indolent form of MCL. Cytogenetic studies showed t(11:14) translocation. Two years later, he was found to have axillary and abdominal lymphadenopathy, rectal and terminal ileal lesions, bone marrow infiltration (16%) and IgM paraproteinemia with immunoparesis. He was treated with R-COP (rituximab, cyclophosphamide, vincristine and prednisolone), followed by ibrutinib as maintenance therapy. He remained in very good partial remission for 13 years. In 2024, he developed rapid onset abdominal pain and marked ascites associated with massive para-aortic, mesenteric and pelvic lymphadenopathy and weight loss. Repeat lymph node biopsy and molecular studies showed high-risk MCL with a high Ki 67 index of 90%, p53 positivity and BIRC3 positivity. Abdominal paracentesis was performed and more than 16 litres of chylous fluid was drained (Figure 1).
Treatment comprised R-CHOP (R-COP plus doxorubicin) and a high energy, high protein, low fat diet with < 20gms fats (non MCT fats) + MCT fats (medium chain triglycerides) with nutritional supplements. This dietary modification assists in hampering chylomicrons processing, thus reduce the production and flow of chyle. MCTs can be absorbed by enterocytes and travel through the portal vein directly to the liver rather than via the lymphatic system [3]. Multidimensional approach to management of chylous ascites appears to have beneficial clinical effect.
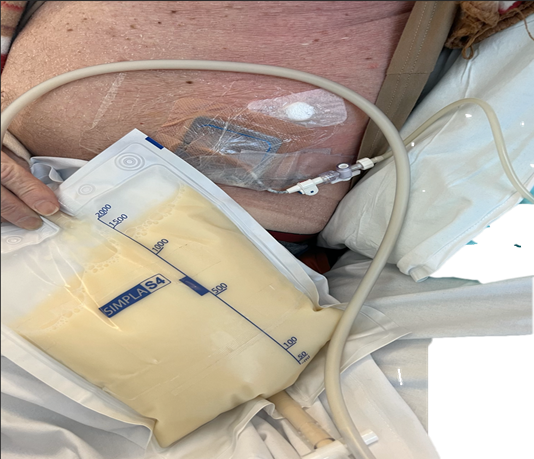
Figure 1: Drainage of Chylous fluid.
His MCL remained refractory but responded to zanirutinib therapy with an excellent clinical response, including complete resolution of his ascites. Figure 2a and Figure 2b show the pre and post treatment abdominal CT scans.
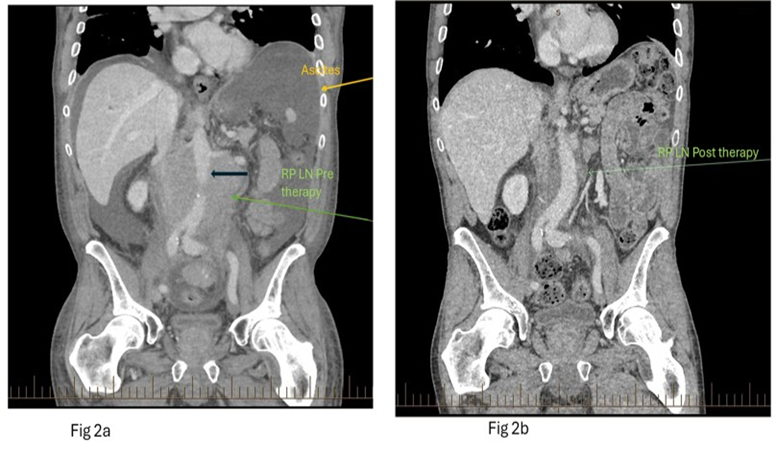
Figure 2a and 2b: Coronal contrast enhanced CT of the abdomen pretreatment (Figure 2a, left), demonstrates moderate ascites (yellow arrow) and marked soft tissue lymph nodal retroperitoneal mass (green arrow) encircling the aorta (bold black arrow). Post treatment coronal image (Figure 2b; right image) demonstrates significant reduction in the retroperitoneal mass (green arrow).
Unfortunately, six months later his disease recurred with marked lymphadenopathy, without ascites. Treatment with salvage chemotherapy followed by CAR-T therapy was ineffective. However, he showed minor response to a combination of rituximab, bendamustine and cytarabine (R-BAC) therapy and is currently receiving pirtobrutinib showing sustained partial remission status.
Discussion
MCL is an uncommon subtype of non-Hodgkin’s lymphoma, which initially runs an indolent clinical course, akin to follicular lymphoma – the most reported subtype of lymphoma associated with chylous ascites [4,5].
Reports of MCL associated chylous ascites are rare and described in indolent phase of disease [1]. The rapid onset of chylous ascites in our patient occurred when the disease transformed to a high proliferative state (highlighting the need for repeat biopsy), which required aggressive chemoimmunotherapy to resolve the ascites.
The dietary changes used as part of the management in our patient were a short-term measure and was supervised by a qualified Dietitian to reduce chyle production (which is triggered by dietary long chain triglycerides); short and medium chain triglycerides are absorbed directly by the portal venous system [6].
Prognosis depends on controlling underlying disease activity with appropriate therapy.
Conflict of interest: None to declare.
References
- Fernandes R, Leite M, Cochicho J, Veríssimo R, Oliveira A. (2018). Chylous ascites due to mantle cell lymphoma. Eur J Case Rep Intern Med. 5: 000871.
- Tlili Y, Hadrich Z, Hafsi M, Sahir O, Rached B, Mestiri H. (2024). Refractory chylous ascites revealing follicular lymphoma: A case report. International Journal of Surgery Case Reports. 116: 109414.
- Duletzke NT, Kiraly LN, Martindale RG. (2023). Chylothorax and chylous ascites: Overview, management and nutrition. Nutr Clin Pract. 38: 557-563.
- Weber B, Luke ND. (2022). Rapid Onset Chylous Ascites Presenting as the Initial Manifestation of Follicular Lymphoma: A Case Report. Cureus. 14: e27199.
- Ohe M, Baba M, Shidal H. (2019). A case of follicular lymphoma accompanied with chylous ascites. Blood Res. 54: 163-163.
- Al-Busafi SA, Ghali P, Deschenes M, Wong P. (2014). Chylous Ascites: Evolution and Management. ISRN Hepatol. 3: 240473.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.