Mapping the Burden of Type 2 Diabetes: Real-World Evidence on Management Patterns, Comorbidities, and Glycemic Outcomes in India
by Pankaj Aneja1, Rahul Medidar2, Bhasker Anand3, D.P. Chakraborty4, Sumit Bhushan5*, Sanjay Choudhari5, Rahee Borulkar5, Prajakta Bhosale5, Rujuta Gadkari5, Saiprasad Patil5
1Aneja diabetes and diagnosis centre, Delhi, India
2Medidar Diabetes Care, Pune, India
3Integrated Medicine Clinic, Chennai, India
4New Town Nursing Home, Kolkata, India
5Glenmark Pharmaceuticals ltd, India
*Corresponding author: Sumit Bhushan, Glenmark Pharmaceuticals Ltd, India
Received Date: 30 January 2026
Accepted Date: 06 February 2026
Published Date: 09 February 2026
Citation: Aneja P, Medidar R, Anand B, Chakraborty DP, Bhushan S (2025) Mapping the Burden of Type 2 Diabetes: Real-World Evidence on Management Patterns, Comorbidities, and Glycemic Outcomes in India. J Diabetes Treat 11: 10157. DOI: https://doi.org/10.29011/2574-7568.010157
Abstract
Background: The global prevalence of diabetes mellitus is rising at an alarming rate, with India frequently described as the epicentre of this growing epidemic. Type 2 diabetes mellitus (T2DM) contributes substantially to morbidity, mortality, and healthcare expenditure due to its chronic course and associated cardiometabolic complications. Although a wide range of antidiabetic therapies is available and commonly prescribed in clinical practice, real- world evidence on prescribing patterns, comorbidity burden, and glycaemic outcomes in Indian patients remains limited. Understanding real-world treatment practices is essential for identifying gaps between guideline recommendations and routine care. Objective: To comprehensively characterise real-world prescribing patterns of antidiabetic medications and to evaluate glycaemic control, comorbidities, renal and metabolic parameters, and treatment regimens among patients with T2DM across diverse clinical settings in India. Methods: This multicentre, retrospective, cross-sectional drug utilisation study analysed data from 2,444 adults diagnosed with T2DM across 430 outpatient clinics in India. Demographic characteristics, medical history, laboratory parameters, comorbidities, and antidiabetic treatment regimens were collected using structured electronic case record forms. Descriptive statistical methods were applied to summarise clinical characteristics, treatment patterns, and outcome measures. Results: The mean age of the study population was 55.55 ± 10.41 years, with males constituting 66.34% of participants. Hypertension (49.10%) and dyslipidaemia (27.95%) were the most prevalent comorbidities. Despite widespread use of oral hypoglycaemic agents and multidrug regimens, glycaemic control was suboptimal, with only 15.36% of patients achieving HbA1c levels below 7%. More than two-thirds of patients were receiving triple or higher antidiabetic therapy. Conclusion: Despite extensive use of contemporary antidiabetic therapies, the majority of Indian patients with T2DM fail to achieve recommended glycaemic targets. These findings highlight the need for patient-centred, rational, and integrated diabetes management strategies to improve long-term clinical outcomes.
Keywords: Type 2 Diabetes Mellitus; India; Drug Utilization Study; Glycaemic Control; Comorbidities; Real-World Evidence
Introduction
Type 2 diabetes mellitus (T2DM) is a major global health challenge, disproportionately affecting low- and middle- income countries. In 2024, an estimated 589 million adults aged 20-79 years were living with diabetes, causing 3.4 million deaths and global health expenditure exceeding US$1 trillion; prevalence is projected to reach 853 million by 2050 [1,2].
India, often termed the ‘diabetes capital of the world’, [3,4] exemplifies this crisis. In 2024, approximately 89.8 million Indian adults had diabetes (10.5% prevalence), with numbers projected to rise to 156.7 million by 2050 [5,6]. Nearly 43% remain undiagnosed, highlighting a substantial detection gap [7,8]. The clinical burden is compounded by frequent comorbidities like hypertension, dyslipidaemia, cardiovascular and kidney disease, and microvascular complications, all contributing to increased morbidity, premature mortality, and healthcare costs [9].
The American Diabetes Association (ADA) recommends an integrated approach combining lifestyle modification and pharmacological therapy to achieve optimal metabolic control and long-term disease management in diabetes. Obesity is a major contributor to lifestyle-related disorders, particularly type 2 diabetes mellitus (T2DM), through its role in promoting insulin resistance and metabolic dysfunction. Failure to adhere to recommended lifestyle interventions therefore contribute significantly to inadequate glycemic control despite ongoing pharmacological treatment [3].
While the multiple therapies including biguanides, sulfonylureas, DPP4 inhibitors, SGLT2 inhibitors, GLP1 receptor agonists, and insulin combinations, real-world glycemic control remains inadequate due to clinical heterogeneity, treatment inertia, and limited access [1,10,11]. Meanwhile, ongoing research into innovative therapeutic approaches is crucial to address persistent challenges in glycemic control and reduce long-term complications associated with type 2 diabetes mellitus [12,13]. Guidelines recommend early use of agents with proven cardio renal benefits alongside comprehensive cardio metabolic risk management, yet implementation in routine practice remains inconsistent [10,11].
Accordingly, this cross-sectional study of Indian adults with T2DM aims to characterise demographics, comorbidities, therapeutic patterns, and intermediate outcomes, generating actionable insights to improve the quality, equity, and effectiveness of diabetes care in India.
Materials and Methods
Study Design and Methodology
This retrospective, cross-sectional drug utilization study was conducted over six months across 430 clinics from urban and suburban areas in India, analysing medical records of 2444 adults with T2DM receiving prescribed antidiabetic medications. Records with missing key clinical or laboratory data were excluded, this approach enabled the inclusion of a population reflective of realworld outpatient practice while ensuring the completeness and reliability of the dataset. Since there is no hypothesis to proof, no formal sample size has been calculated for the study.
The primary objective was the use of several antidiabetic medications, while Secondary objective included glycaemic (HbA1c, FPG, PPG), renal (eGFR, creatinine), lipid, and hepatic parameters; patient demographics and medical history; treatment regimens (monotherapy, dual, triple, or >triple therapy); rates of target attainment; and associations between treatment regimens and outcomes.
A predesigned electronic case report form (eCRF), was used to collect and extract demographic information, vital signs, diagnoses, detailed history, therapeutic regimens, laboratory results (glycemic, renal, lipid, hepatic parameters), comorbidities, and adverse events Descriptive statistics were applied, with categorical variables expressed as frequencies and percentages, and continuous variables summarized by means and standard deviations.
Results
A total of 2444 adults with type 2 diabetes mellitus were included in the analysis. The mean age was 55.55 ± 10.41 years, in terms of gender distribution, 66.34% were male, 30.11% were female, and 3.55% had missing data.
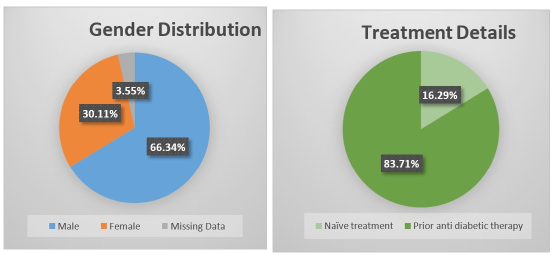
Figure 1: Gender distribution and Treatment Details.
Mean weight and height were 73.02 ± 11.57 kg and 163.13 ± 12.38 cm, respectively. Mean systolic and diastolic blood pressure were at 135.71 ± 18.42 mmHg and 88.13 ± 14.65 mmHg, respectively. The majority (83.71%) were pre-treated and 16.29% were treatment naïve, with a mean diabetes duration of 7.41 ± 6.78 years and mean therapy duration of 6.14 ± 5.25 years.
|
Parameter |
Mean ± SD |
|
Age (years) |
55.55 ± 10.41 |
|
Weight (kg) |
73.02 ± 11.57 |
|
Height (cm) |
163.13 ± 12.38 |
|
Systolic Blood Pressure (mmHg) |
135.71 ± 18.42 |
|
Diastolic Blood Pressure (mmHg) |
88.13 ± 14.65 |
|
Pulse Rate / min |
85.40 ± 9.98 |
|
Mean Duration of Diabetes Mellitus (in years) |
7.41 ± 6.78 |
|
Mean Duration of therapy for Diabetes Mellitus (in years) |
6.14 ± 5.25 |
Table 1: Baseline Demographic and Clinical Profile.
In the past year, 1326 (79.64%) patients reported no episodes of hypoglycemia, 330 (19.82%) experienced 1-5 episodes, and 9 (0.54%) reported 6-10 episodes. A family history of type 2 diabetes mellitus was present in 737 (30.91%) patients, with a mean duration of 15.40 (SD 11.47) years. Diabetic neuropathy was present in 454 (19.36%) patients, and retinopathy in 338 (14.41%). Blurring of vision was reported by 288 (12.08%) patients, and symptoms of fatigue by 620 (25.80%). Hospitalisation within the past year was reported by 137 (6.31%) patients. Tobacco use was documented in 593 (24.59%), with a mean smoking duration of 14.65 (SD 9.40) years.
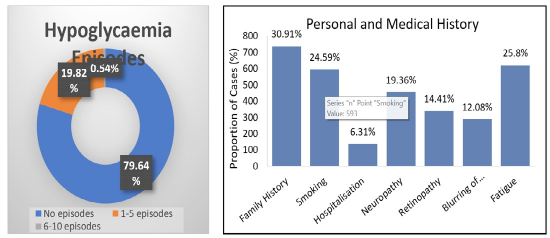
Figure 2: Hypoglycemia Episodes and Personal and Medical History.
Hypertension was the most common comorbidity, affecting 49.10% of patients, followed by dyslipidaemia (27.95%), cardiovascular disease (26.39%), obesity (25.86%), overweight (15.51%) and Anxiety/Depression was reported in 13.18%. Chronic kidney disease and metabolic dysfunction associated steatotic liver disease (MASLD) were in 3.76% and 5.16% patients, respectively. Obstructive sleep apnea affected 3.03%, and osteoporosis was reported in 5.93%. Other conditions accounted for 4.75%.
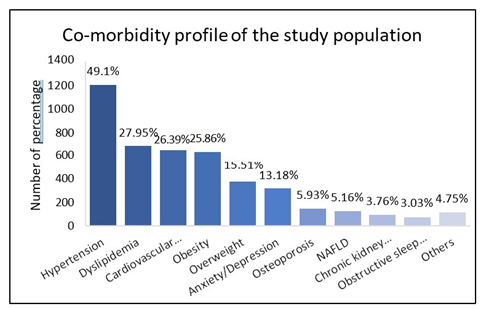
Figure 3: Co-morbidity profile.
The most frequently prescribed concomitant drugs were antihypertensives including Angiotensin receptor blockers (17.93%) and calcium channel blockers (5.06%), while statins (19.27%) predominated in dyslipidaemia management, and antihistamines (0.49%) were commonly used for respiratory conditions. Anticonvulsants (1.51%) were the most used for anxiety, and proton pump inhibitors (5.83%) were widely prescribed in other categories.
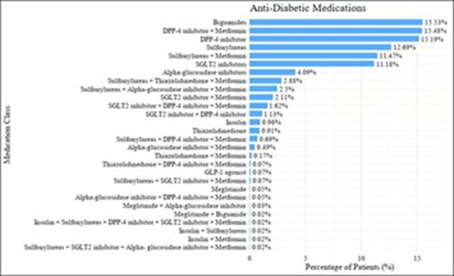
Figure 4: Distribution Anti- diabetic Medications.
The most frequently prescribed Anti-Diabetic Medications medication classes were biguanides (15.53%), DPP-4 inhibitors (15.19%), and the fixed-dose combination of DPP-4 inhibitors with metformin (15.48%). Sulfonylureas (12.69%) and SGLT2 inhibitors (11.18%) were also commonly used. Insulin was prescribed in only 0.96% of patients.
Monotherapy was the least common regimen, prescribed in only 5.81% of patients. Dual therapy was used in 25.33%, while triple therapy accounted for 33.76%. The most frequent approach involved the use of more than three medications was 33.84%, reflecting the predominance of multidrug treatment strategies. This pattern likely reflects the clinical complexity and comorbidity burden of the study population. Treatment data was not available for 1.26% of patients.
|
S. No |
Treatment Regimen |
n |
% |
|
1 |
Mono Therapy |
142 |
5.81 |
|
2 |
Dual Therapy |
619 |
25.33 |
|
3 |
Triple Therapy |
825 |
33.76 |
|
4 |
> Triple Therapy |
827 |
33.84 |
|
5 |
Data not available |
31 |
1.26 |
Note: n = number of subjects; Total subjects (N) = 2444; Percentages are calculated as (n/N × 100)
Table 2: Treatment Regimen.
Controlled HbA1c (<7%) was achieved in 375 patients (15.36%), while 831 (34.03%) and 747 (30.59%) had HbA1c levels of 7–8% and 8–9%, respectively. HbA1c exceeded 9% in 489 patients (20.02%). Fasting plasma glucose (FPG ≤110 mg/dL) was controlled in 160 patients (6.66%), whereas postprandial glucose (PPG ≤140 mg/dL) was controlled in only 19 patients (0.77%).
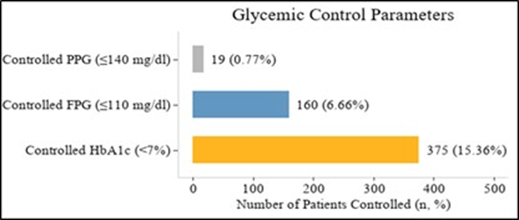
Note: n = number of subjects; Total subjects (N) = 2444; Percentages are calculated as (n/N × 100)
Figure 5: Glycemic Control Parameters.
The mean fasting blood glucose (FBG) was 162.13 mg/dL (SD
42.46; n=2415) and postprandial glucose (PPG) 248.42 mg/dL (SD 70.23; n=2417), with HbA1c averaging 8.04% (SD 1.32; n= 2442) with 15.36% of patients achieving <7%, 34.03% between 7-8%, 30.59% between 8–9%, and 20.02% >9%, indicating suboptimal glycaemic control in the majority.
Lipid profiles revealed total cholesterol 196.47 mg/dL (SD 84.49; n=1642), LDL cholesterol 116.17 mg/dL (SD 43.51; n=1284), HDL cholesterol 58.33 mg/dL (SD 38.86; n=1240), and triglycerides 170.45 mg/dL (SD 72.71; n=1312). Liver function parameters showed mean serum bilirubin 10.97 mg/dL (SD 43.83; n=969), alanine aminotransferase (ALT) 36.13 U/L (SD 16.22; n=954), and aspartate aminotransferase (AST) 36.52 U/L (SD 18.00; n=952).
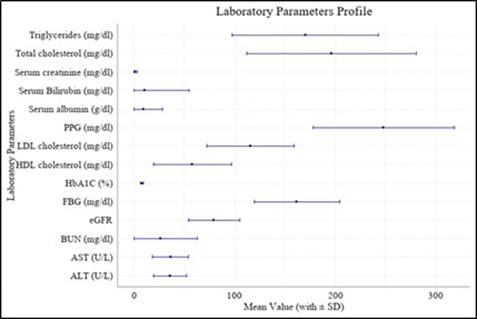
Figure 6: Laboratory Parameters.
Renal parameters showed a mean eGFR of 79.74 mL/min/1.73 m² (25.72; n=938) and serum creatinine of 1.47 mg/dL (SD 1.99; n=1471), while serum albumin was 9.34 g/dL (SD 19.07; n= 854) and blood urea nitrogen (BUN) 26.83 mg/dL (SD 36.21; n= 948). The distribution of estimated glomerular filtration rate (eGFR) indicated that most participants had normal or mildly reduced renal function. Overall, 377 (40.19%) participants had eGFR values of 90–120 mL/min per 1.73 m², and 410 (43.71%) had values of 60–90 mL/min per 1.73 m². Moderate renal impairment was observed in 53 (5.65%) participants (eGFR 45–60) and 39 (4.16%) participants (eGFR 30– 45). Severe impairment was present in 11 (1.17%) participants (eGFR 15–30), and 35 (3.73%) had eGFR <15. Hyperfiltration (eGFR >120) was identified in 13 (1.39%) participants.
Discussion
The real-world cross sectional, Drug utilization data from this comprehensive Indian cross-sectional study highlights significant clinical challenges faced in managing Type 2 Diabetes Mellitus (T2DM) within this population. Consistent with previous findings in similar studies, the predominance of biguanides and DPP-4 inhibitors primarily in combination with metformin reflects current therapeutic standards [14].
However, this study documents a relatively low proportion of patients achieving glycaemic control targets, with only 15.36% maintaining HbA1c levels below 7%, and even fewer achieving controlled fasting and postprandial glucose levels (6.66% and 0.77% respectively). This gap emphasizes the persistent difficulty of diabetes management despite widespread use of recommended pharmacotherapies [14].
The high prevalence of comorbidities such as hypertension (49.10%), dyslipidemia (27.95%), and obesity (25.86%) parallels the patterns observed globally but marks an especially vulnerable patient subset at increased cardiovascular and renal risk [9]. The frequent use of multi-drug regimens, with more than one-third of patients on triple or greater combinations, signals the complexity of disease progression and the possibility of therapeutic inertia or barriers including medication adherence issues, socioeconomic factors, and health system limitations [11].
Renal function data showed that most patients had mild impairment (43.71%; eGFR 60–90 mL/min/1.73 m²), with smaller but clinically important proportions exhibiting moderate impairment (9.81%; eGFR 30–60) or severe dysfunction (4.90%; eGFR <30); notably, 40.19% had normal function (eGFR 90–120) and 1.39% had eGFR >120.
These findings reinforce the need for careful choice of antihyperglycemic agents with renal safety in mind [10,11].
Compared internationally, medication selection in Indian patients aligns with global practice [10,15]; however, diabetes control rates lag behind those documented in other studies, where higher percentages achieve glycaemic targets [16,10]. This discrepancy highlights the urgent need for focused patient education, individualized treatment strategies, enhanced monitoring, and the leveraging of diabetes registries as quality improvement tools to bridge this therapeutic gap [14,17-19]. Collectively, these results advocate for intensified multidisciplinary approaches and resource allocation tailored to the Indian healthcare context to improve long-term outcomes in T2DM management [15].
Strengths of this study include its large sample size and nationwide coverage. Limitations include retrospective data collection and absence of longitudinal follow up.
In summary, the results highlight persistent gaps in glycemic control, a high burden of comorbidities, and underutilization of cardio-renal protective agents among Indian patients with T2DM. These findings emphasize on the urgent need for a multipronged strategy that combines patient education, system-level strengthening, wider adoption of evidence-based therapies, and policy reforms to subsidize innovative treatments. Registry-based evidence can serve as the cornerstone for such efforts, ensuring that advances in diabetes research translate into tangible improvements in health outcomes for millions of Indians.
Conclusion
Current anti-diabetic regimens in India are safe and largely guideline-consistent, yet glycaemic and metabolic targets remain unsatisfactory [14]. High comorbidity rates compound management complexity, reinforcing the need for early, individualized therapeutic strategies and systematic registry-based monitoring [15,18,20]. This study provides a framework for ongoing clinical evaluation, quality improvement, and evidence-based policy formulation [10]. Targeted, data-driven interventions have the potential to reduce diabetes-related complications and improve long-term outcomes in Indian patients with T2DM [15,10].
Author Contribution Statement
Designed research: S.B.; performed research: P.A; analyzed data: R.M. & B.A; data acquisition: D.P.C.; Writing original draft: S.B., S.C, R.B & P. B; review & editing: R.G., S.P All. All authors have read and agreed to the published version of the manuscript.
Ethics Approval Statement
The work presented in this study was in accordance with the study protocol, the New Drugs and Clinical Trials Rules 2019 issued by the Government of India, the ethical principles that have their origin in the Declaration of Helsinki, International Council for Harmonisation (ICH) Good Clinical Practice (GCP), and all applicable local regulatory requirements. Independent Ethics committee approval was obtained prior to study initiation and data collection.
Acknowledgments
We would like to extend our thanks to all the institutes and respective investigators and team members for their support. We also appreciate the CRO support by Auriga Research Pvt Ltd., India for clinical trial management. We also thank Sclintech for medical writing support in the study.
Funding
This study was funded by Glenmark Pharmaceuticals Limited. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data Availability Statement
Data generated during this study are available from the corresponding author on reasonable request.
Conflict of Interest Statement
Dr. Sumit Bhushan, Dr. Rahee Borulkar, Ms. Prajakta Bhosale, Ms. Rujuta Gadkari and Dr. Saiprasad Patil are employees of Glenmark. All other investigators/authors have no conflicts of interest that are directly relevant to the content of this article.
References
- International Diabetes Federation. IDF Diabetes Atlas, 11th ed. Global factsheet (2025 update). 2025.
- International Diabetes Federation. IDF Diabetes Atlas (accessed 2025). Key global statistics. 2025.
- Joshi SR, Parikh RM, Sharma S, et al. (2023) A 3-Year, Pan-India, Prospective, Longitudinal Study on the Management and Real-World Outcomes of Type 2 Diabetes Mellitus. Endocrinol Diabetes Metab 6: e316.
- Mishra S, Pareek N (2023) The “diabetic capital” of the world: Cumulative burden of diabetes in India. Int J Recent Sci Res.
- International Diabetes Federation. India country profile (IDF Diabetes Atlas data portal, accessed 2025). 2025.
- ICMR-INDIAB Study Collaborators. Metabolic non-communicable disease health report of India. Lancet Diabetes Endocrinol. 2023.
- ICMR-INDIAB Study Collaborators. Metabolic non-communicable disease health report of India. PubMed record. 2023.
- Sekher TV, et al. (2023) Prevalence, Awareness, Treatment, and Control of Diabetes in India: A Nationally Representative Survey of Adults Aged 45 Years and Older. Lancet Glob Health 13: e1543-e1552.
- Gupta R, et al. Risk factors and complication burden in Indian T2DM: narrative review. Front Endocrinol/PMC record. 2025.
- Bak JCG, Serné EH, Kramer MHH, Nieuwdorp M, Verheugt CL (2020) National diabetes registries: Do they make a difference? Acta Diabetologica 58: 267–278.
- Khunti K, Wolden ML, Thorsted BL, Andersen M, Davies MJ (2018) Therapeutic Inertia in Type 2 Diabetes: Prevalence, Causes, and Consequences. Diabetes Obes Metab 20: 427–437.
- The Lancet Regional Health – Europe. Diabetes registries-key enablers of high-quality care (Commentary). 2025.
- Gieroba B, Kryska A, Sroka-Bartnicka A (2025) Type 2 diabetes mellitus - conventional therapies and future perspectives in innovative treatment. Biochem Biophys Rep 42: 102037.
- Borgharkar SS, Das SS (2019) Real-world evidence of glycemic control among patients with type 2 diabetes mellitus in India: The TIGHT study. BMJ Open Diabetes Res Care 7: e000654.
- Traina F, et al. (2022) The EUBIROD review of diabetes registries and data sources in Europe. Acta Diabetol 59.
- Eeg-Olofsson K, Svensson A-M, Franzén S, Ismail HA, Törnblom M, Levrat-Guillen F (2023) Real-world study of flash glucose monitoring among adults with type 2 diabetes within the Swedish National Diabetes Register. Diabetes Vasc Dis Res 20: 14791641211067418.
- Chamany S, Silver LD, Bassett MT, Driver CR, Berger DK, Neuhaus CE, Kumar N, Frieden TR (2009) Tracking diabetes: New York City’s A1C Registry. Milbank Q 87: 547–70.
- Russell KG, Rosenzweig J (2007) Improving outcomes for patients with diabetes using Joslin Diabetes Center’s 4 and Risk Stratification system. J Healthc Inf Manag 21: 26–33.
- Rawdaree P, Ngarmukos C, Deerochanawong C, Suwanwalaikorn S, Chetthakul T, et al. (2006) Thailand diabetes registry (TDR) project: clinical status and long-term vascular complications in diabetic patients. J Med Assoc Thai 89: S1-9.
- Toh MP, Leong HS, Lim BK (2009) Development of a diabetes registry to improve quality of care in the National Healthcare Group in Singapore. Ann Acad Med Singapore 38: 546-6.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.