Low-dose Chemotherapy, in Addition to Impacting Breast Cancer Metastasis, Establishes Immunomodulation: A Preclinical Study
by Dileep Kumar1, Luana Cristina Camargo1, Victor Carlos Mello da Silva2, Natalia Lemos Chaves1,3*, Thyago Jose Arruda Pacheco4, Luis Alexandre Muehlmann5, Joao Paulo Figueiró Longo1
1Department of Genetics and Morphology, Institutes of Biological Sciences, University of Brasilia, Brasilia 70910-900, DF, Brazil.
2Laboratory of Microscopy and Microanalysis, Department of Cell Biology, Institute of Biological Sciences, University of Brasília, Brasília 70910-900, Brazil.
3Laboratory of Bioactive Compounds and Nanobiotechnology (LBCNano), Department of Genetics and Morphology, Institutes of Biological Sciences, University of Brasilia, Brasilia 70910-900, DF, Brazil.
4Department of Physiological Sciences, Institutes of Biological Sciences, University of Brasilia, Brasilia 70910-900, DF, Brazil.
5Faculty of Ceilândia, University of Brasilia, Campus Universitário - Centro Metropolitano, Ceilândia Sul - DF. CEP: 72220-275 Brasilia, Brazil.
*Corresponding author: Chaves NL, Laboratory of Bioactive Compounds and Nanobiotechnology (LBCNano), Department of Genetics and Morphology, Institutes of Biological Sciences, University of Brasilia, Brasilia 70910-900, DF, Brazil.
Received Date: 02 March, 2026
Accepted Date: 11 March, 2026
Published Date: 16 March, 2026
Citation: Kumar D, Camargo LC, Mello da Silva VC, Chaves NL, Arruda Pacheco TJ, et al. (2026) Low-dose Chemotherapy, in Addition to Impacting Breast Cancer Metastasis, Establishes Immunomodulation: A Preclinical Study. J Oncol Res Ther 11: 10335. DOI: https://doi.org/10.29011/2574-710X.10335
Abstract
Background: Metastatic breast cancer (MBC) is a major cause of morbidity and mortality among women globally. While early-stage breast cancer is often effectively treated, advanced metastatic treatment still faces significant challenges. Myeloid-Derived Suppressor Cells (MDSCs) play a significant role in immunosuppression process, which proliferating tumor cells evade immune detection by suppressing the antitumor immune response and development of treatment resistance. This study proposes to evaluate the presence of MDSC after treatment of mice bearing tumor with two classes of drugs in the low-dose chemotherapy regimen. Methods: The effects of low-dose chemotherapy (LDC) with cyclophosphamide (CP) and 5-fluorouracil (5-FU) on the tumor microenvironment and immune response were evaluated in a preclinical model of metastatic breast cancer. Mice bearing 4T1 tumors undergoing LDC treatment and controls were assessed by analyzing body weight, survival, tumor progression, lung metastasis, spleen size, and MDSC (myeloid-derived suppressor cells) population. Results: LDC was well-tolerated with no significant impact on body weight or survival. LDC effectively inhibited primary tumor growth and reduced lung metastasis progression, particularly with 5-FU. Histopathological analysis confirmed the absence of metastases in treated animals’ lungs. LDC also reduced MDSC populations, suggesting an immunomodulatory effect. Hematological results also indicated an active immune response. Conclusion: In summary, LDC shows potential as an effective, well-tolerated MBC treatment, inhibiting tumor growth and modulating the immune response.
Keywords: Metastatic Breast Cancer; Myeloid Derived Suppressor Cells; Low-Dose Chemotherapy; Cyclophosphamide; 5-Fluorouracil.
Introduction
Breast cancer (BC) remains one of the most common and serious cancers, especially when metastases are present. A striking feature of tumor growth is immune evasion, which allows tumor cells to escape and suppress antitumor immune responses. One of the important cellular mechanisms involved in this “escape” involves the generation of certain immature immunosuppressive myeloid cells in the patient, known as myeloid-derived suppressor cells (MDSCs), which inhibit the immune response directed at antitumor defenses [1-8].
Targeting MDSCs is considered a promising strategy to prevent tumor progression and metastasis. Traditional chemotherapy, while effective in eliminating tumor cells, can also impact immune cell populations, leading to systemic immunosuppression and potential side effects that compromise therapeutic success [9-15]. Low-dose chemotherapy (LDC) offers a targeted approach, using lower drug concentrations to minimize side effects while controlling cancer. LDC has gained attention for its ability to specifically inhibit the suppressive arm of the immune response, modulate the tumor microenvironment, inhibit angiogenesis, and stimulate immune responses, thereby exerting indirect effects on tumor cells [16, 17].
Cyclophosphamide (CP) and 5-fluorouracil (5-FU) are essential chemotherapeutic agents. However, their use at high doses is associated with severe side effects like hemorrhagic cystitis or cardiac toxicity [18, 19]. Low-dose CP is known to enhance anti-tumor immune responses by reducing regulatory T cells and increasing effector T cell activity, shifting the tumor microenvironment from immunosuppressive to immunostimulatory [18, 20-23].
This study aimed to evaluate the impact of LDC with CP and 5-FU on MDSC presence in mice bearing 4T1 tumors, investigating its role in immunomodulation and metastasis inhibition.
Materials and Methods
Animals, Experimental Design, and Pharmacological Study
Female BALB/c mice (20 ± 1 g) were obtained from the Animal Resources Centre, Universidade Federal de Goiás (UFG), and housed under specific pathogen-free conditions. All animal procedures adhered to the Ethics Committee for the Use of Animals (CEUA) Guidelines (Protocol 026/2020). Mice, 8-10 weeks old, were maintained on a standard diet in controlled conditions. After a one-week quarantine, mice were randomly assigned to four experimental groups (n=5 per group):
- Healthy control group: Non-tumor-bearing mice receiving saline.
- Cyclophosphamide-treated group: Tumor-bearing mice treated with cyclophosphamide (80 mg/kg).
- 5-Fluorouracil-treated group: Tumor-bearing mice treated with 5-fluorouracil (80 mg/kg).
- Positive control group: Tumor-bearing mice receiving no treatment.
On Day 0, mice were subcutaneously injected with 4 × 105 4T1Luc cells into the left dorsal hip to establish a tumor model. Tumor growth and metastasis were monitored from Day 1-7. Body weight and tumor size were recorded every three days. Drug administration (CP and 5-FU) was initiated on Day 8 and continued for four consecutive days (Days 8-11), with daily intraperitoneal injections after tumors reached approximately 50 mm³. Bioluminescence imaging (BLI) of tumors and metastatic regions was performed on Day 7 and Day 41. Survival rates were assessed from Day 12-42. On Day 42, mice were euthanized for biological sample collection, including hematological analyses, splenic cell extractions for MDSC quantification, and histopathological evaluations of organs (lungs and liver).
Cell culture Conditions
The murine breast cancer cell line 4T1 modified to express firefly luciferase (4T1-Luc) using cell culture medium from Lucexpressing 4T1 cells cultivated in a 96-well cell culture plate to enable cell tracking and quantification in vivo [24] 4T1-Luc cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM), which is enhanced with 10% (v:v) fetal bovine serum and 1% (v:v) antibiotic solution (100 IU/mL penicillin and 100 mg/mL streptomycin). The cells were maintained at 37ºC in a humidified atmosphere with 5% CO2. Adherent cells were incubated with 0.25% trypsin-EDTA solution (Sigma-Aldrich), centrifuged, and then rinsed with sterile phosphate buffer before cell resuspension in sterile media. Cell suspensions were counted at a final concentration of 4 x 10 5 cells/ml.
Chemotherapy
The chemotherapeutics used in these studies: CP and 5-FU were purchased from Merck Sigma-Aldrich. Twenty-nine-gram insulin syringe was used to administer doses intraperitoneally (I.P) to each mouse (Figure 1). According to the manufacturer’s recommendations, we performed CP and 5-FU chemotherapy and diluted in sterile conditions using either phosphate-buffered saline (PBS). Chemotherapy concentration of 80 mg/kg of both drug CP and 5-FU separately were diluted for 20-gram mice to receive a 100 μl intraperitoneal infusion [25]. All other materials were of pharmaceutical or analytical grade (Figure 1).
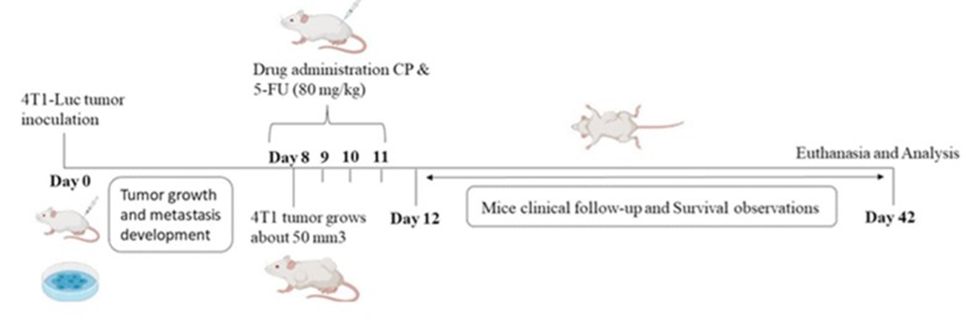
Figure 1: In-vivo 4T1-Luc breast cell lines induction. Schematic representation of treatment and analysis of tumor-bearing mice and experimental data endpoints.
Evaluations of Metastasis by Bioluminescence
To assess the development of lung tumors and metastases, we employed in vivo bioluminescence imaging. 100 μl of 15 mg/ ml concentration of D-luciferin (Sigma) was administered intraperitoneally per animal, and images were taken using the IVIS Lumina® XR Series III (Caliper Life Sciences). Animals were kept under isoflurane anesthesia after luciferin administration, and 20 bioluminescent photos were taken every 20 minutes one photo per minute. The open emission filter took pictures for 60 seconds for each image capture. The bioluminescent data for each area of interest was calculated using the highest bioluminescence measurement (tumor or thorax region) two times firstly day 7 and lastly day 41 during whole experiment. Living Image 3.0 software was used to process each picture (Caliper Life Sciences, CA, USA).
MDSCs Quantification
MSDCs populations were evaluated in spleen samples. Female balb/c mice spleens were harvested under sterile conditions, placed in a sterile culture dish and 2 mL PBS was added, then was quickly cut into small pieces of about 1 mm3 and crushed gently. The process was repeated for each treatment group of mice. Using a serological pipette (10 mL), the cell suspension was filtered using a 70 μm cell strainer (BD Falcon). Prepared and dissociated singlecell suspension was incubated with ACK (Ammonium Chloride Potassium) lysis buffer (10 mL, Thermo Fisher Scientific) at RT for 7 minutes to remove red blood cells. The lysis buffer was removed, and the reaction was stopped with the addition of 10 mL ice-cold PBS. Counting of cells was performed using trypan blue and a hemocytometer Neubauer chamber. Next, a cell suspension containing 1×106 splenocytes was prepared in 100 µl for phenotype characterization. Splenocytes cells suspension was labeled with a 20 µl aliquot of fluorescently FITC (Fluorescein isothiocyanate) conjugated anti-bodies anti-CD11b (rat monoclonal anti-mouse), and PE (Phycoerythrin) conjugated anti-Ly6C, anti-Ly6G (rat monoclonal anti-mouse) 1:100 dilution fixed and incubated under dark condition for 20 minutes at 4 °C. Cells were washed twice with PBS buffer and analyzed in a FACSCaliburTM flow cytometer (BD Biosciences). A total of 10.000 events were counted per sample, and data were analyzed using the FlowJo® software.
Histopathological Evaluation
The whole organs (lung and liver) were thoroughly rinsed in phosphate-buffered saline solution to effectively eliminate any excess blood and then subjected to fixation in 10% buffered formalin (Sigma-Aldrich) for a total of 24 hours at room temperature. Following this, the organs were treated with alcohol solutions (100%, 100%, 70%) to achieve dehydration. Then diaphanized in xylol, and ultimately included and embedded in paraffin through the utilization of a tissue processor. Manual microtome (Leica Microsystems, Nussloch, Germany) Leica RM2235, was employed to obtain 5 μm thick sections, which were subsequently stained with H&E, and prepared for light microscope analysis.
Statistical Analysis
Data are reported as the mean ± SD. The Statistical significance evaluation of data was performed using a t-test, and one way ANOVA analyses of variance, followed by XY non-linear regression using the GraphPad Prism Prism® 8.0 software. Survival curve analysis was performed using log-rank (Mental-Cox) test. The significance level (α) adopted in statistical comparisons was 0.05.
Results
Impact of Low-Dose Chemotherapy: Body Weight, Survival Rate, and Tumor Progression
As illustrated in Figure 1(a), continuous LDC of CP and 5-FU were administered intraperitoneally (i.p.) from 8 – 11 day.
During the first 24 hours of observation and at the conclusion of the treatment period, no signs of skin ulceration or behavioral changes were noted in any of the animals. Additionally, body weights remained stable across all groups throughout the study.
As shown in Figure 2(c), both CP and 5-FU treatments independently exhibited primary volume tumor inhibition compared to the untreated positive control group. Notably, 5-FU displayed superior therapeutic efficacy during the final three days of the study (days 38 to 41) compared to CP.
Figure 2(d) indicates that the tumor weight was significantly lower in the 5-FU treated group compared to the CP group and the non-treated control. Further analysis using bioluminescence imaging (Figure 2e) confirmed a reduction in tumor growth in 5-FU treated mice relative to the CP-treated and untreated groups. In vivo bioluminescence imaging on days 7 and 41 (Figure 2 f) highlighted a noticeable gap in the 5-FU group due to the death of one animal. There were no statistically significant differences between the treated groups.
The 5-FU group showed minimal weight fluctuation and effectively improved the survival time in comparison to the CP-treated and tumor-bearing groups. As depicted in Figure 2(a), no significant differences in body weight were observed between treated and untreated mice, as well as the healthy control group throughout the experimental period.
Overall survival analysis (Figure 2 b) revealed an 80% survival rate in the 5-FU treated group, with a 20% mortality rate. This difference was not statistically significant according to the MantelCox test. These results suggest that while both CP and 5-FU have tumor-inhibitory effects, 5-FU demonstrates a more pronounced therapeutic benefit with minimal adverse effects on body weight and survival.
Evaluation of low dose therapy on distant lung metastasis progression
All treated groups had similar lungs weight with no significant difference between the treated and positive control groups. The lungs of CP or 5-FU-treated mice presented a similar morphology to that observed in healthy mice. Bioluminescence imaging of thorax region (Figure 2F) demonstrates a trend of reduction in metastasis among CP or 5-FU groups compared to the positive control, the observation is the treatment LDC impair the secondary metastasis in the thorax breast region. Despite, these data were not statistically significant, the histopathological H&E staining analysis present many tumor metastasis loci (black square presentation) in lungs of positive control group, while there was no metastasis in the lungs of CP or 5-FU treated group, as shown in Figure 3 (c). These results suggest that CP as well as 5-FU can inhibit distant tumor metastasis to lungs.
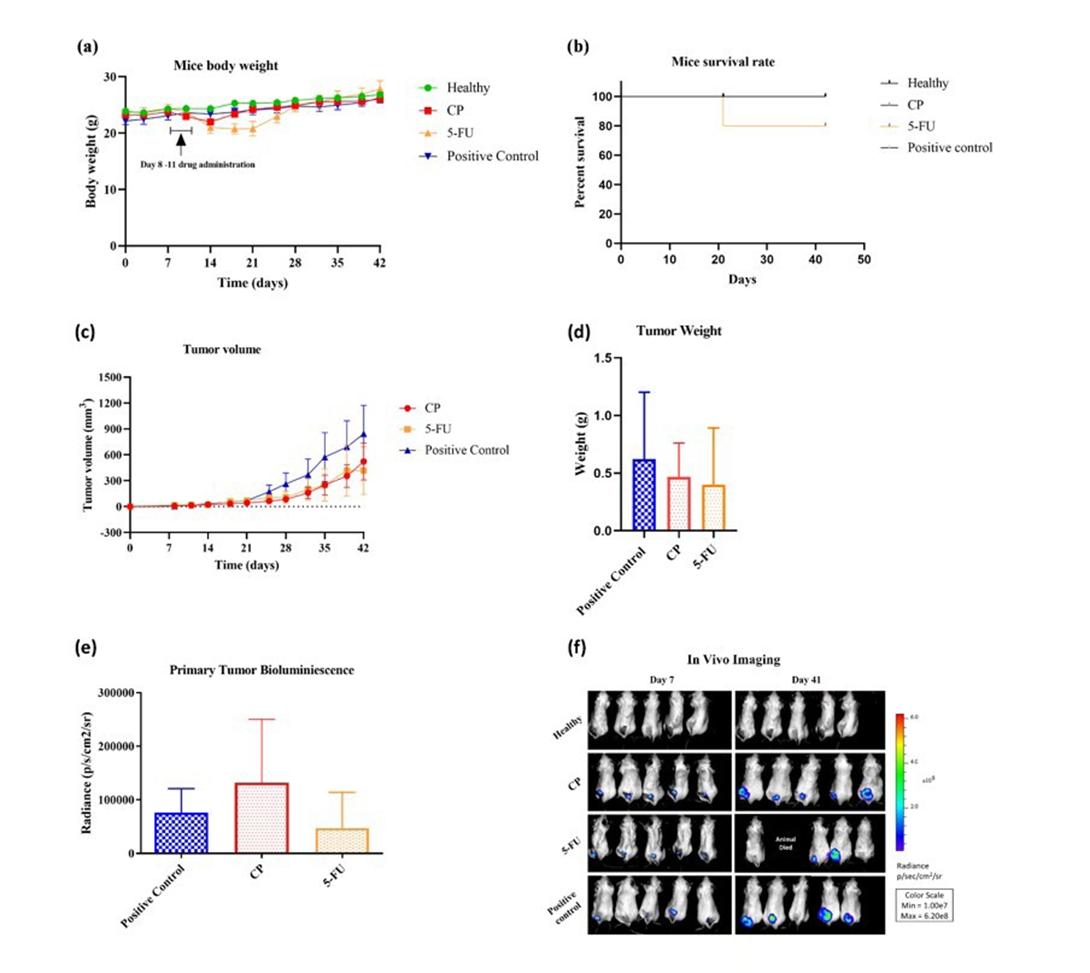
Figure 2: Treatment Effects on Body Weight and Animal Survival (a) Body weight measurements were recorded from day 0 to day 42. Low-dose drug administration occurred between days 8 and 11. Throughout the experimental period, no significant differences in overall body weight variations were observed among the treatment groups. (b) The percentage survival curve indicated an 80% survival rate in the 5-FU group, with one animal death recorded. However, this survival rate did not show statistical significance according to the Mantel-Cox test (P = 0.3916, n = 5). Evaluation of In Vivo Tumor Progression and Therapeutic Effects c) Tumor volumes were measured at 3-day intervals using a caliper across all administration groups. (d) The excised tumors from all treated groups were quantified for weight after 42 days of tumor induction. (e) Region of Interest (ROI) analysis was conducted to thoroughly quantify tumor load across treatment groups compared to untreated controls after 42 days of tumor induction. (f) In vivo bioluminescence imaging was performed on days 7 and 41. The mean bioluminescence radiance (expressed as photons/sec/cm²/sr) ± SD was presented in a bar graph format. All data are presented as mean ± SD. No statistically significant differences were observed in tumor volume, tumor weight, or primary tumor bioluminescence among the treatment groups.
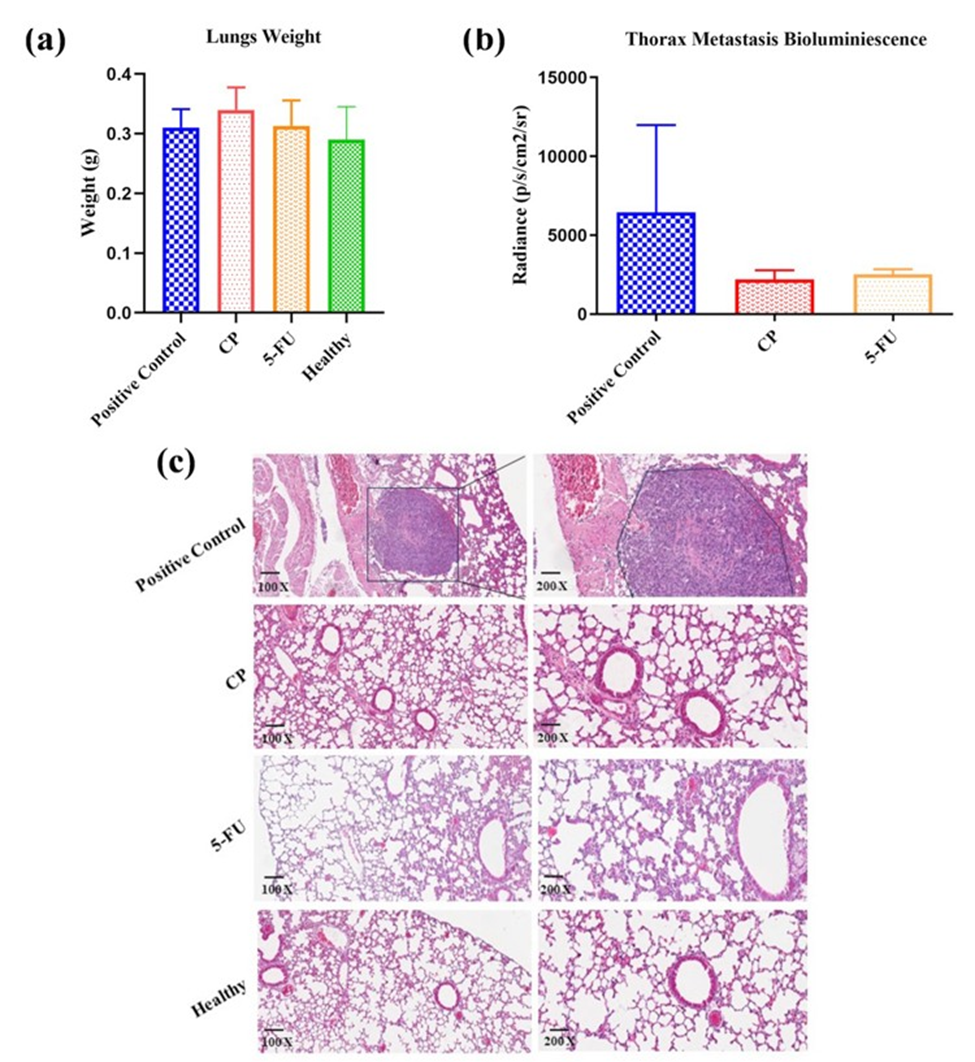
Figure 3: Effective lung metastasis prevention (a) Average lung weight after animal euthanasia on day 42 after tumor induction of all treated groups (n = 5). (b) Quantification of bioluminescence with the mean fluorescent intensity of the thorax regions, observed inhibition of metastasis in CP and 5-FU groups. (c) Histopathological examination of the lungs: representative images of H&E staining (100x and 200x) of isolated lung of different administration groups to evaluate the tissue histological conditions. No significant difference has been found in lung weight and in thorax metastasis bioluminescence.
Treatment effects on spleen and MDSCs population quantifications
It was observed in figure 4a variation between the spleen weight in all treated groups compared to the healthy. The spleens of untreated tumor-bearing mice positive control presented a three spleen from five, enlarged in size (Figure 4b) having weight increase compared with the spleens of mice healthy without tumors. Likewise, difference was observed between animal healthy with CP or 5-FU treated mice.
According to the findings shown in Figure 4 (c), the tumor-bearing mice without treatment had a higher number of MSDCs compared with healthy and CP/5-FU treated mice with no significant difference. Although, this finding suggests that the low-dose treatment with CP or 5-FU reduces the Ly6C/Ly6G and CD11b+ populations.
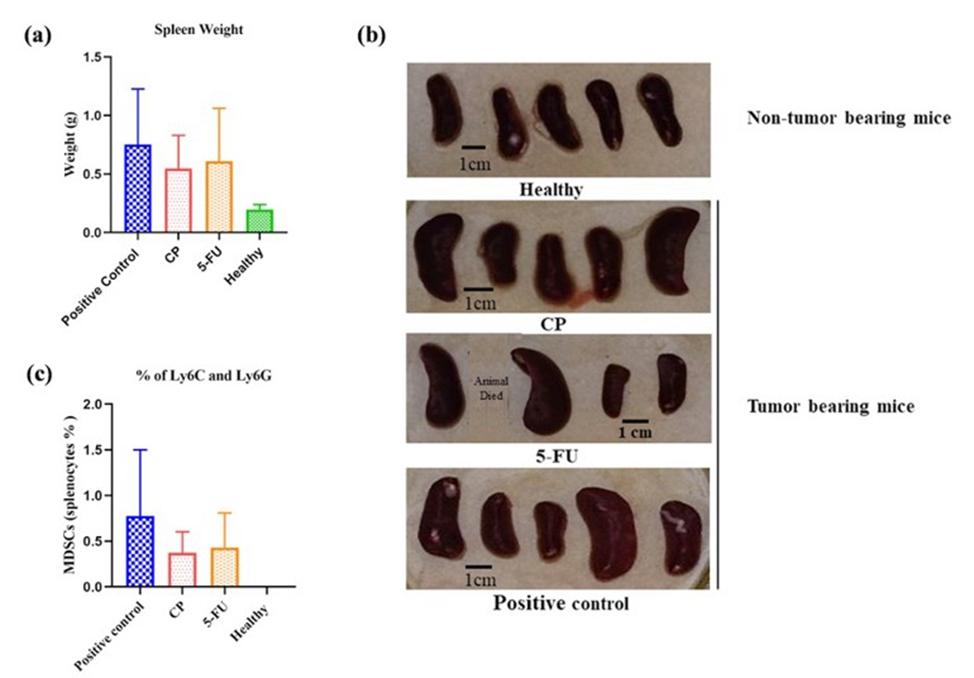
Figure 4: Overall treatment effects on spleen weights, morphology and splenocytes population quantifications using flow-cytometry. (a) Average spleen weights variation of experimental mice. (b) Macroscopic aspects of spleen excised from non-tumor bearing mice and tumor bearing group of animals. (c) MDSCs splenocytes populations percentage among all animal group. Each group included n = 5 mice, expect the 5-FU group n=4 mice. Mean and SEM is shown. Spleen weight and MDSCs % population which comparatively having no statistically significant difference with positive control.
Characterization of blood cell population
Hematological analysis showed significant alterations in leukogram of the treated animals compared with untreated positive control group. Total white blood cell populations in figure 5a are higher in untreated positive control group, while the value of CP or 5-FU have reduction in the cell count comparing to positive control. Production of lymphocytes (SCR-small cell ratio) showed in Figure 5b were significantly lower between healthy and in treated group along with untreated. White Blood cell - Middle cell ratio (W-MCR - basophils, eosinophils and neutrophil) were significantly higher (Figure 5c) in CP treated group with healthy, likewise 5-FU with healthy along with untreated group, indicating the activation of immune cells to fight with immunosuppressant cells. Large cell ratio (LCR) – Monocyte population in (Figure 5d) showed comparatively equal in treated groups and higher in untreated group. These findings suggest that the treatment might be affecting the immune system, potentially by reducing inflammation or suppressing overall activity, however this parameter do not have significance difference. Platelets count (Figure 5e) 5-FU group were significantly higher count among group CP along with healthy and untreated positive control.
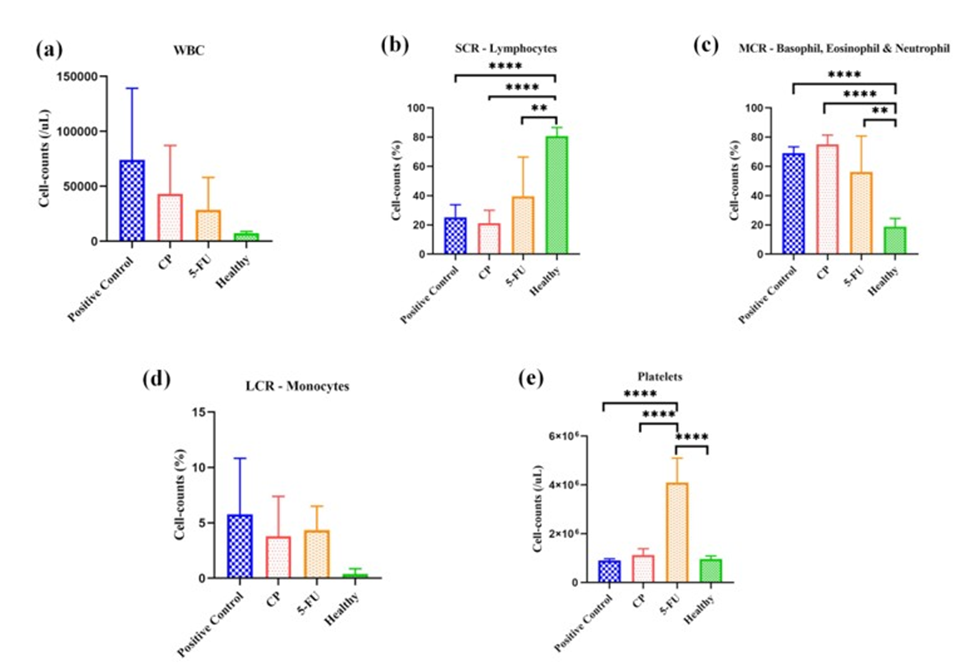
Figure 5: Leucocytes and platelets cell counts (cells/mL). (a) White blood cells - parameter has enhanced production of white blood cell count compared to the healthy group because of inflammation. (b) SCR-lymphocyte production in treated groups CP, 5-FU, and positive control have lower counts compared to the healthy group, (****p < 0.0001, **p < 0.0025, ****p < 0.0001) (n=5, 5-FU: n=4). (c) MCR -basophil, eosinophil, neutrophil production in treated groups CP, 5-FU, and positive control has higher counts compared to the healthy group, (****p < 0.0001, **p < 0.0018, ****p < 0.0001) (n=5, 5-FU: n=4) (d) LCR-monocyte production in treated groups CP, 5-FU, and positive control has higher counts compared to the healthy group having no significance difference. (e) Platelets production in treated groups CP have a little variation comparatively to healthy and positive control but group 5-FU has enhanced higher counts compared to the healthy group, (****p < 0.0001, ****p < 0.0001, ****p < 0.0001) (n=5, 5-FU: n=4).
Addtionally, the hematological evaluation (Figure 6) revealed alterations in the erythrogram among all treated groups in comparison to the positive control group of animals. The red blood cell (RBC) count, hemoglobin (HGB) level, and hematocrit (HCT) values exhibited similar variations among the treated and untreated groups with no significant treatment-related differences in total cell numbers. However, the mean corpuscular volume (MCV) values significantly have the consistent population across all groups. Both the mean corpuscular hemoglobin (MCH) and mean corpuscular hemoglobin concentration (MCHC) demonstrated consistent population counts, with significant differences observed among all treated groups compared to both the healthy and untreated groups. The RBC-distribution width variations coefficient (RDW-CV) and RBC-distribution width standard deviation (RDW-SD) count values were statistically lower across all groups when compared to the 5-FU treated group.
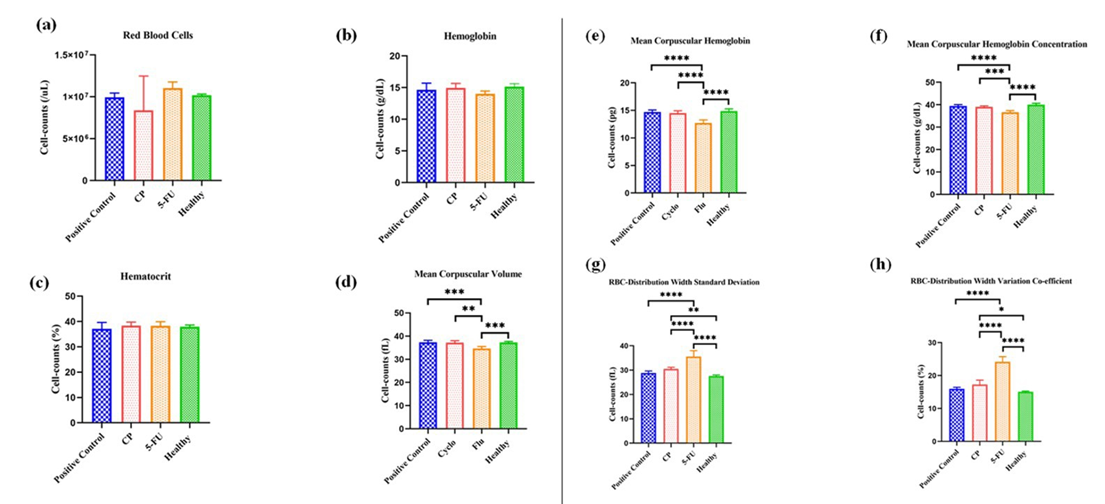
Figure 6: Absolute erythrocytes count (cells/mL). (a) Red blood cell - counts have almost similar high ratios in all groups except CP, respectively. (b) Hemoglobin counts have a similar cell counts ratio in all treated groups (c) Hematocrit counts have a similar number of cell productions in all treated groups (Fig. a-c) and found no significant difference (d) Mean corpuscular volume in group Flu has a lesser count having a significant difference in other groups, (***p < 0.0010, **p < 0.0013, ***p < 0.0008) (n=5), respectively. (e) Mean corpuscular hemoglobin in treated group Flu has a low number of counts comparatively to other groups, (****p < 0.0001, ****p < 0.0001, ****p < 0.0001) (n=5). (f) Mean corpuscular hemoglobin concentration in treated group Flu has also a low number of counts comparatively to other groups, (****p < 0.0001, ***p < 0.0003, ****p < 0.0001) (n=5) (g) RBC-distribution width standard deviation number is higher also in the group Flu comparing to other treated groups, (**p < 0.0100, ****p < 0.0001, ****p < 0.0001, ****p < 0.0001) (n=5). (h) RBC-distribution width variation co-efficient number is higher in the group Flu comparing to other treated groups, (*p < 0.0172, ****p < 0.0001, ****p < 0.0001, ****p < 0.0001) (n=5).
There is a positive correlation between % MDSC and the lung metastasis (p=0.0001), spleen weight (p=0.0005), and white blood cells quantification (p=0.0009). This positive correlation was not observed when comparing the % MDSC and primary tumor quantification (p=0.12), evidencing a systemic immune effect of the treatment (Figure S1).
Supplementary Materials
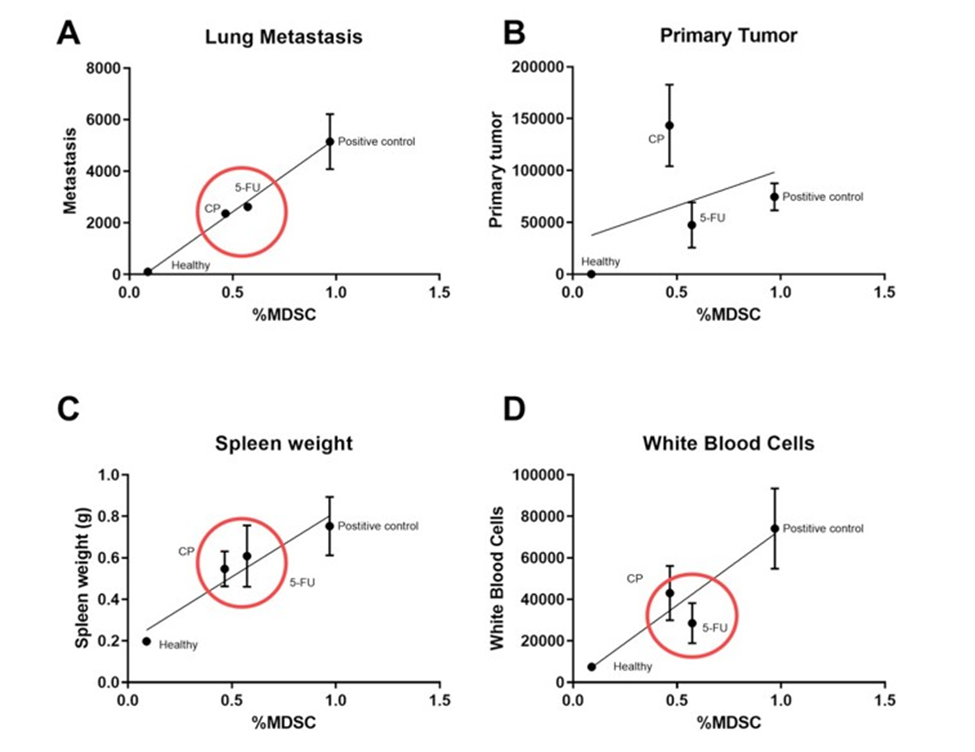
Figure S1: Linear correlation between (a) metastasis and % MDSC (p=0.0001). (b) Primary tumor and % MDSC (p=0.12) (c) Spleen weight and % MDSC (p=0.0005) (d) White Blood Cell and % MDSC (p=0.0009). Values are mean ± SEM (n=5 animals per group and n=4 animal for 5-FU group). P-values (p>0.05) were generated by Linear regression.
Discussion
The results highlight major findings after LDC application against experimental BC tumor model (4T1 Luc): We first observed that LDC with CP-80 mg/kg or 5-FU-80 mg/kg did not significantly impact the body weight of the mice over the course of the experiment, except a transitory reduction in treated group with the 5-FU or in CP group reduction occurred for one day and subsequently returned to baseline level. Furthermore, the survival rates of the treated groups were comparable to the control groups. These findings suggest that the selected LDC regimens were well-tolerated and did not induce acute toxicity or significant changes in body weight or survival rate.
Both CP or 5-FU treatment inhibited and reduction in primary tumor growth compared to the untreated control group. Previous results have observed that LDC provides a twofold advantage by diminishing adverse effects while also offering benefits as reducing of metastasis [27]. 5-FU exhibited better therapeutic activity in the later stages of the thorax bioluminescence experiment. Additionally, both CP or 5-FU treatment led to a reduction in tumor weight compared to the control group. Bioluminescence imaging further confirmed the reduction in tumor growth in the treated groups relative to the control group. These findings highlight the efficacy of LDC in inhibiting tumor progression.
CP or 5-FU treatment impaired distant lung metastasis progression compared to the untreated control group. Histopathological analysis also confirmed the absence of metastasis in the lungs of treated mice. Low-dose protocols, have a better ability to control distant metastasis to the lungs by developing systemic immunological protection. This result is quite important since one of the goals of contemporary treatments is to reduce or impair the distant lung metastasis.
This study corroborated findings on the pathological functions of MDSCs in promoting cancerous tumors [22, 28-30] and that LDC therapy has an effect on these cells. Previous studies have shown that low doses of CP increase the antitumor immune response through targeted depletion of regulatory T cells and increased activity of effector T cells, and that the induction of MDSCs likely prevents the optimal possible immune response [18, 31].
5-FU rapidly reduces the number of circulating MDSCs, contributing to their depletion in systemic circulation and to the reduction of M-MDSCs and PMN-MDSCs in the spleen [32]. This result, also observed in the present study, highlights the effectiveness of 5-FU in reducing the presence of these MDSC subsets in this immunological organ. CP, on the other hand, has a slight effect on the growth of MDSCs in tumor sites or in the spleen of mice with tumors and showed minimal impact on the accumulation of MDSCs in tumor sites [33].
Variations in spleen weight between treated groups compared to healthy controls have been reported in studies of untreated tumor-bearing mice, due to the infiltration of neoplastic cells into the spleen. As reported, spleen enlargement is indicative of complications and inflammation caused by immune cell suppression and is also related to the overproduction of MDSCs [34]. Furthermore, treatment with LDC and CP or 5-FU reduced the population of MDSCs in the spleen of metastasis-associated tumor-bearing mice, suggesting a potential immunomodulatory effect of the treatment.
Our finding reinforces an association between MDSCs and lung metastasis formation trough the establishment of an immunosuppressive microtumor environment as a pivotal in facilitating the development these metastasis [35, 36].
Hematological analysis revealed significant alterations in leukogram parameters among the treated groups compared to the untreated control group. Notably, LDC with CP or 5-FU led to reductions in total white blood cell populations and lymphocyte percentages, indicating modulation of the immune response. The platelet counts were significantly higher in the 5-FU group, potentially due to the stimulation of bone marrow response, leading to the production of megakaryocyte precursors [34, 37].
Further analysis of the erythrogram revealed notable alterations in red blood cell parameters among the treated groups compared to the control group. While no significant differences were observed in total cell numbers, variations in mean corpuscular volume and hemoglobin concentration suggest potential treatment-related effects on erythropoiesis.
Conclusion
Taken together, the results of this study confirm the multifaceted effects of LDC on tumor progression, distant pulmonary metastasis, immune modulation, and hematological parameters in a murine model of breast cancer. These results corroborate the potential usefulness of LDC as a therapeutic strategy with reduced systemic toxicity with the chosen drugs, particularly with 5-FU, which effectively inhibited the growth of the primary tumor and reduced the progression of pulmonary metastasis, in addition to modulating the immune response thought MDSC cells, and highlight avenues for future research aimed at optimizing its efficacy and safety profile.
Funding: Not Applicable
Acknowledgments: This article is part of two research projects supported by the Brazilian agency FAP-DF/Brazil (00193.00001053/2021-24)/(00193-00000734/2021-75), CNPq (403536/2021-9)/(303182/2019-9), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brazil (CAPES) Finance Code 001.
Author Contributions: Dileep Kumar conceptualization, study design, data accumulation, writing, original draft preparation, editing and figure preparation; Luana Cristina Camargo: data accumulation; Victor Carlos Mello da Silva: data accumulation; Natalia Lemos Chaves: writing and editing; Thyago Jose Arruda Pacheco: content formation; Joao Paulo Figueiró Longo: conceptualization, study design, results analysis and project administration; Luiz Alexandre Muehlmann: conceptualization, results analysis. All authors have read and approved the submitted version of the manuscript.
Data Availability: Data is provided within the manuscript or supplementary information files. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval: The authors state that this study was conducted following the principles of the Basel Declaration and the recommendations of the Animal Use Ethics Committee of the University of Brasilia, which approved protocol CEUA (Protocol number 026/2020).
Patient consent for publication: Not applicable.
Conflict of interest: All authors declare no conflicts of interest.
References
- Łukasiewicz, Czeczelewski M, Forma A, Baj J, Sitarz R, Stanisławek A (2021) Breast cancer-epidemiology, risk factors, classification, prognostic markers, and current treatment strategies-an updated review. Cancers 13: 4287.
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, et al. (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians 71: 209-249.
- Lei S, Zheng R, Zhang S, Wang S, Chen R, et al. (2021) Global patterns of breast cancer incidence and mortality: A population‐based cancer registry data analysis from 2000 to 2020. Cancer Communications 41: 1183-1194.
- Mahvi DA, Liu R, Grinstaff MW, Colson YL, Raut CP (2018) Local cancer recurrence: the realities, challenges, and opportunities for new therapies. CA: a cancer journal for clinicians 68: 488-505.
- Kazawa N, Ishii K, Shimazu H, Tanigawa N, Sawada B (2021) A case of the pulmonary nodular amyloidosis: Multiple calcified nodules exhibiting interval growth. International Journal of Radiology and Diagnostic Imaging 4: 35-42.
- Gould Rothberg BE, Quest TE, Yeung SCJ, Pelosof LC, Gerber DE, et al. (2022) Oncologic emergencies and urgencies: A comprehensive review. CA: A Cancer Journal for Clinicians 72: 570-593.
- Mortezaee K (2020) Immune escape: A critical hallmark in solid tumors. Life sciences 258: 118110.
- Almeida CF, Correia-da-Silva G, Teixeira N, Amaral C (2024) Influence of tumor microenvironment on the different breast cancer subtypes and applied therapies. Biochemical Pharmacology 223: 116178.
- Dong P, Yan Y, Fan Y, Wang H, Wu D, et al. (2022) The role of myeloidderived suppressor cells in the treatment of pancreatic cancer. Technology in Cancer Research & Treatment 21: 15330338221142472.
- Cheng X, Wang H, Wang Z, Zhu B, Long H (2023) Tumor-associated myeloid cells in cancer immunotherapy. Journal of Hematology & Oncology 16: 71.
- Zhao Y, Du J, Shen X (2023) Targeting myeloid-derived suppressor cells in tumor immunotherapy: Current, future and beyond. Frontiers in Immunology 14: 1157537.
- Wang S, Zhao X, Wu S, Cui D, Xu Z (2023) Myeloid-derived suppressor cells: Key immunosuppressive regulators and therapeutic targets in cancer. Pathology-Research and Practice 248: 154711.
- De Cicco P, Ercolan G, Ianaro A (2020) The new era of cancer immunotherapy: targeting myeloid-derived suppressor cells to overcome immune evasion. Frontiers in immunology 11: 1680.
- Mukherjee O, Rakshit S, Shanmugam G, Sarkar K (2023) Role of chemotherapeutic drugs in immunomodulation of cancer. Current Research in Immunology 4: 100068.
- Anand, U, Dey A, Singh Chandel AK, Sanyal R, Mishra A, et al. (2023) Cancer chemotherapy and beyond: Current status, drug candidates, associated risks and progress in targeted therapeutics. Genes & Diseases 10: 1367-1401.
- Xie X, Wu Y, Luo S, Yang H, Li L, et al. (2017) Efficacy and toxicity of low-dose versus conventional-dose chemotherapy for malignant tumors: a meta-analysis of 6 randomized controlled trials. Asian Pacific journal of cancer prevention: APJCP 18: 479.
- Krajnak S, Schnatz C, Almstedt K, Brenner W, Haertner F, et al. (2020) Low-dose metronomic chemotherapy as an efficient treatment option in metastatic breast cancer-results of an exploratory case-control study. Breast cancer research and treatment 182: 389-399.
- Madondo MT, Seok-Joo C, Sun S, Cho H, Tae-Jin K, et al. (2016) Low dose cyclophosphamide: mechanisms of T cell modulation. Cancer treatment reviews 42: 3-9.
- Ali M, Manjula SN, Mohiuddin I, Mruthunjaya K, Shakeel F, et al. (2024) Noni enhances the anticancer activity of cyclophosphamide and suppresses myelotoxicity and hepatotoxicity in tumor-bearing mice. Journal of Cancer Research and Clinical Oncology 150: 1-16.
- Kikuchi H, Maishi N, Yu L, Jia Z, Li C, et al. (2024) Low-dose metronomic cisplatin as an antiangiogenic and anti-inflammatory strategy for cancer. British Journal of Cancer 130: 336-345.
- Mafi A, Rezaee M, Hedayati N, Hogan SD, Reiter RJ, et al. (2023) Melatonin and 5-fluorouracil combination chemotherapy: opportunities and efficacy in cancer therapy. Cell Communication and Signaling 21: 33.
- Yang Y, Zhang M, Zhang Y, Liu K, Lu C (2023) 5-Fluorouracil suppresses colon tumor through activating the p53-fas pathway to sensitize myeloid-derived suppressor cells to FasL+ cytotoxic t lymphocyte cytotoxicity. Cancers 15: 1563.
- Mathew AA, Zakkariya ZT, Ashokan A, Manohar M, Keechilat P, Nair SV, et al. (2023) 5-FU mediated depletion of myeloid suppressor cells enhances T-cell infiltration and anti-tumor response in immunotherapy– resistant lung tumor. International Immunopharmacology 120: 110129.
- Tao K, Fang M, Alroy J, Sahagian GG (2008) Imagable 4T1 model for the study of late stage breast cancer. BMC cancer 8: 1-19.
- Aston WJ, Subedi L,Jha SK, Kweon S, Kang SH, et al. (2017) A systematic investigation of the maximum tolerated dose of cytotoxic chemotherapy with and without supportive care in mice. BMC cancer 17: 1-10.
- Xiong X, Zhao J, Su R, Liu C, Guo X, et al. (2021) Double enhancement of immunogenic cell death and antigen presentation for cancer immunotherapy. Nano Today 39: 101225.
- Lin F, Chen H, Jiang T, Zheng J, Liu Q, et al. (2022) The effect of lowdose chemotherapy on the tumor microenvironment and its antitumor activity combined with anti-PD-1 antibody. Immunotherapy 14: 283294.
- Ren R, Xiong C, Ma R, Wang Y, Yue T, et al. (2023) The recent progress of myeloid‐derived suppressor cell and its targeted therapies in cancers. MedComm 4: e323.
- Li K, Shi H, Zhang B, Ou X, Ma Q, et al. (2021) Myeloid-derived suppressor cells as immunosuppressive regulators and therapeutic targets in cancer. Signal Transduction and Targeted Therapy 6: 362.
- Liao AH, Lee YA, Lin DL, Chuang HC, Wang JK, et al. (2023) Treatment efficacy of low-dose 5-fluorouracil with ultrasound in mediating 5-fluorouracil-loaded microbubble cavitation in head and neck cancer. Drug Delivery 30: 1-13.
- Leong WI, Ames RY, Haverkamp JM, Torres L, Kline J, et al. (2019) Low-dose metronomic cyclophosphamide complements the actions of an intratumoral C-class CpG TLR9 agonist to potentiate innate immunity and drive potent T cell-mediated anti-tumor responses. Oncotarget (68): 7220.
- Tang F, Tie Y, Hong W, Wei Y, Tu C, et al. (2021) Targeting myeloidderived suppressor cells for premetastatic niche disruption after tumor resection. Annals of Surgical Oncology 28: 4030-4048.
- Wang S, Wang J, Chen Z, Luo J, Guo W, et al. (2024) Targeting M2-like tumor-associated macrophages is a potential therapeutic approach to overcome antitumor drug resistance. NPJ Precision Oncology 8: 31.
- Wu C, Hua Q, Zheng L (2020) Generation of myeloid cells in cancer: the spleen matters. Frontiers in Immunology 11: 1126.
- Cui W, Wang Z, Lv J, Qin Y, Shi H (2023) MDSCs: The Key Players in the Formation of Pre-Metastatic Niche. Frontiers in BioscienceLandmark 28: 58.
- Zhang Z, Yao Z, Zhang Z, Cui L, Zhang L, et al. (2023) Local radiotherapy for murine breast cancer increases risk of metastasis by promoting the recruitment of M-MDSCs in lung. Cancer Cell International 23: 107.
- Li X, Slayton WB (2013) Molecular mechanisms of platelet and stem cell rebound after 5-fluorouracil treatment. Experimental hematology 41: 635-645. e3.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.