Liquid Biopsy–Detected EGFR S645C Driving a Striking and Durable Complete Response to Afatinib: A Case Report
by Letizia Padula1, Alice Ilari1*, Davide Seminati2, Diego Cortinovis1,3, Luca Sala1
1SC Medical Oncology, Fondazione IRCCS San Gerardo dei Tintori, Monza, Italy.
2Molecular Pathology Unit, Fondazione IRCCS San Gerardo Dei Tintori, Monza, Italy.
3Medical Department, Università degli Studi di Milano-Bicocca, Milano, Italy.
*Corresponding author: Ilari A, SC Medical Oncology, Fondazione IRCCS San Gerardo dei Tintori, Monza, Italy.
Received Date: 02 March, 2026
Accepted Date: 10 March, 2026
Published Date: 13 March, 2026
Citation: Padula L, Ilari A, Seminati D, Cortinovis D, Sala L (2026) Liquid Biopsy–Detected EGFR S645C Driving a Striking and Durable Complete Response to Afatinib: A Case Report. J Oncol Res Ther 11: 10334. DOI: https://doi.org/10.29011/2574-710X.10334
Abstract
Uncommon mutations in the EGFR gene currently represent both a diagnostic and therapeutic challenge in non-small cell lung cancer (NSCLC). This is a rarity in oncology, characterized by a different response to tyrosine kinase inhibitors (TKIs) due to the heterogeneity that characterizes each individual mutation, making it even more complex to understand the correct treatment to administer to this class of patients. This case report presents the case of a woman with recurrent lung adenocarcinoma in whom the EGFR S645C (exon 17) mutation detected in liquid biopsy after disease progression on anti-PD-L1 immunotherapy. The patient was therefore treated with Afatinib, a second-generation TKI, with a partial and then complete clinical and radiological response for more than nine months. After initial toxicity with G3 diarrhea related to treatment at the maximum dose, the dose was reduced with excellent tolerance to therapy (from 40 to 20mg/day). The EGFR S645C mutation was identified only through repeat molecular testing using liquid biopsy, as no indication for tissue re-biopsy was present. This finding enabled targeted treatment, improved patient survival, and supports the efficacy of afatinib in rare EGFR mutations.
Keywords: EGFR S645C Mutation; Afatinib; Uncommon EGFR Mutations; Liquid biopsy; ctDNA; Non-Small Cell Lung Cancer; NSCLC.
Introduction
Over the last decade, non-small cell lung cancer (NSCLC) has become treatable through targeted therapy thanks to the identification of driver mutations using Next Generation Sequencing (NGS) analysis. These mutations are defined as actionable, as they are amenable to treatment with targeted agents that have dramatically improved survival rates in this patient population. In particular, the percentage of patients with epidermal growth factor receptor (EGFR) mutations is estimated to represent approximately 12% of all non-Asian patients with NSCLC [1].
This group of patients is further subdivided based on the type of EGFR mutation, which can be common or uncommon [2].
Common mutations, represented by exon 19 deletion and the L858R point mutation in exon 21, account for approximately 85-90% of EGFR-mutated cases and respond to first- and thirdgeneration TKIs [3, 4]. Uncommon mutations, on the other hand, account for 10-15% of EGFR-mutated cases and are characterized by enormous heterogeneity related to a different response to TKIs. The different therapeutic response depends on the mutated protein structure. Among the uncommon mutations, the most frequent are G719X (exon 18), L861Q (exon 21), exon 20 insertion (nonT790M), and S768I (exon 20) mutations. These are mutations that respond differently to second- or third-generation TKIs, and for which there is little data in the literature [5].
One of the drugs that has proven most effective in treating some of the uncommon mutations is Afatinib. It is an irreversible inhibitor of the ErbB family with simultaneous inhibitory action on both wild-type and mutated EGFR, ErbB2 and ErbB4 [1]. As described in the analysis conducted by Yang JC et al., which collected data from the LUX-Lung 2, 3 and 6 trials, Afatinib demonstrated objective response rates (ORR) of 60% and a duration of response (DoR) of 17.1 months in patients with major uncommon mutations (G719X, L861Q, and S768I) [6]. Available evidence suggests a potential role for TKIs in the management of ultra-rare EGFR mutations; notably, the therapeutic landscape is evolving and includes novel third-generation inhibitors, such as furmonertinib, that are currently being investigated [7].
The S645C point mutation of EGFR on exon 17 is based on the substitution of a serine with a cysteine on the tyrosine kinase domain of EGFR, resulting in a different intracellular response to TKIs. Preclinical studies have demonstrated resistance to Osimertinib in the S645C mutation [8, 9]. Furthermore, there is little data in the literature regarding the best therapeutic strategy for this class of patients. In the case report published by Shen CI et al. [10], the efficacy of using Afatinib plus Bevacizumab in the treatment of a patient with NSCLC with EGFR S645C mutation was demonstrated.
Our case report further reinforces the efficacy of Afatinib in the treatment of patients with the EGFR S645C point mutation, providing a therapeutic opportunity for this class of patients for whom the action of the target therapy typically used for common EGFR mutations is associated with resistance. Furthermore, this study highlights the importance of using liquid biopsy as a diagnostic tool in uncommon EGFR mutations [11].
Case Presentation
In March 2022, a 65-year-old female, never-smoker, with a personal history of estrogen receptor-positive (ER+) breast cancer and a family history of breast and gastric cancer, was evaluated due to an increase in carcinoembryonic antigen (CEA) levels. Imaging studies, including 18F-FDG PET/CT, revealed a solitary pulmonary nodule in the left upper lobe. The patient underwent wedge resection of the left upper lobe, and histopathological analysis identified a papillary-predominant invasive lung adenocarcinoma with a lepidic growth pattern, moderately differentiated (PL2), staged as pT2a pN0 (according to the 8th edition of the American Joint Committee on Cancer, AJCC).
At 6-month follow-up, due to persistently elevated CEA levels, radiological and PET/CT examinations demonstrated recurrence of pleural disease on the left side, which proved challenging to biopsy, even with minimally invasive approaches. In January 2023, a CT-guided biopsy of a left paravertebral pleural lesion confirmed recurrence of lung adenocarcinoma, TTF-1 positive and p40 negative. Programmed death-ligand 1 (PD-L1) expression was >50%, and next-generation sequencing (NGS) performed on pleural tissue sample, using Oncomine Comprehensive Assay (v3) NGS panel revealed pathogenic variants pathogenic variants in the BRCA2, TP53, and STK11 genes, none of which were considered actionable according to the European Society for Medical Oncology (ESMO) ESCAT level I–II classification at the time [12].
Based on these findings, first-line immunotherapy with immune checkpoint inhibitors was indicated by national and international guidelines. Baseline CT staging showed pleural effusion, multifocal pleural thickening, a newly detected round lymph node in the left mammary region, and enlargement of the ipsilateral anterior cardio-phrenic lymph node. PET/CT confirmed the presence of metabolically active disease. Target lesions as per RECIST criteria included: left apical parietal pleural thickening (11 mm), left pericardial thickening (10 mm), internal mammary lymph node (7 mm), and anterior cardio-phrenic adenopathy (13 mm).
Treatment with Atezolizumab was started in March 2023. The disease remained radiologically stable under close CT surveillance for 10 months. In May 2024, CT imaging indicated possible disease progression, which was not confirmed on PET. The case was then reviewed by a multidisciplinary team and radiological progression of the disease at the lymph node level was confirmed. To further assess disease status, endobronchial ultrasound (EBUS)-guided biopsy was performed on left hilar lymph node (10L). Cytological analysis confirmed malignant epithelial cells consistent with carcinoma of no special type (NST), although the sample was inadequate for molecular profiling.
Treatment with Atezolizumab was discontinued and the patient was subsequently enrolled in the pre-screening of the B-FAST study in July 2024, a phase 2 trial based on ctDNA analysis on peripheral blood via liquid biopsy with the aim of identifying targetable mutations [13]. Liquid biopsy performed with FoundationOne Liquid CDx confirmed mutations in BRCA2 and TP53 genes and identified also EGFR exon 17 S645C mutation.
Literature review and discussion within an internal molecular tumor board supported initiation of Afatinib at a dose of 40 mg/day in August 2024. However, treatment was poorly tolerated, with Grade 3 diarrhea, Grade 2 cutaneous rash, G2 scalp lesions, Grade 1 onychopathy, and electrolyte imbalances (requiring intravenous supplementation for hypo-/hypercalcemia). The dose was then reduced to 30 mg/day.
In October 2024, PET/CT imaging demonstrated a partial metabolic and dimensional response. Due to persistent grade 2 gastrointestinal adverse events, the dosage was further reduced to 20 mg/day from the fourth treatment cycle onwards, achieving excellent tolerability to treatment.
By January 2025, follow-up CT and PET/CT scans revealed a complete radiological and metabolic response, which was also confirmed at the May 2025 follow-up. In October 2025, after TC-PET scan, stable disease was observed in accordance with RECIST (Response Evaluation Criteria in Solid Tumors) morphological criteria, with increased metabolic activity in two mediastinal lymph nodes and two abdominal lesions. The regions with increased metabolic activity underwent radiation treatment in 10 fractions, as per decision of the multidisciplinary oncology group. The patient continues Afatinib at a maintenance dose of 20 mg/day. The patient’s clinical timeline is best described in Figure 1 and Table 1.
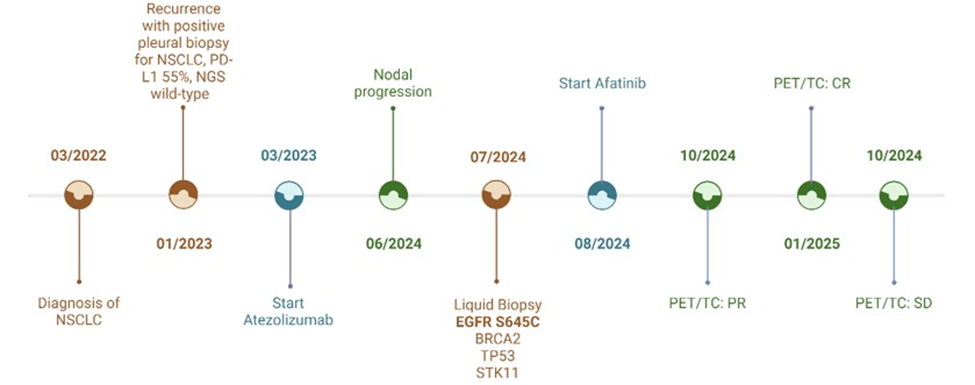
Figure 1: Figure 1 shows the main events in the patient’s clinical history. The diagnostic phases are shown in red, while the treatments initiated are shown in blue, first immunotherapy with Atezolizumab and then the use of TKI with Afatinib. Radiological reassessments using PET/CT are highlighted in green.
|
Date |
Clinical Event |
Procedure / Treatment |
Response / Outcome |
Toxicity (CTCAE) |
|
Mar-22 |
Diagnosis of left upper lobe lung adenocarcinoma, pT2aN0 (moderately
differentiated) |
Surgical wedge resection |
Post-operative remission |
– |
|
Sep-22 |
Suspected left pleural recurrence |
PET/CT for re-evaluation |
Suspicion of progression |
– |
|
Jan-23 |
Pleural biopsy positive for adenocarcinoma (TTF1+, p40–); PD-L1 55%;
NGS BRCA2, STK11 and TP53 variants |
CT-guided pleural biopsy |
Recurrence confirmed |
– |
|
Mar-23 |
Start of immunotherapy within FORTBEAT trial (Arm A) |
Atezolizumab monotherapy |
Stable disease for ~10 months |
– |
|
Jun-24 |
Thoracic–abdominal nodal progression |
CT and PET/CT imaging |
Documented progression |
– |
|
Jul-24 |
Liquid biopsy (B-FAST) → EGFR S645C mutation + BRCA2 and
TP53 variants |
ctDNA NGS analysis |
Actionable molecular target identified |
– |
|
07-Aug-24 |
Start of Afatinib 40 mg daily |
2nd-generation EGFR-TKI |
Targeted therapy initiated |
Diarrhea G3, rash G2, onychopathy G1 |
|
Sep-24 |
Treatment optimization |
Afatinib dose reduction to 30 mg daily |
Toxicities improving |
GI AEs G1 |
|
Oct-24 |
PET/CT re-evaluation |
Imaging follow-up |
Partial response (PR) |
– |
|
Nov-24 |
Further dose reduction for maintenance |
Afatinib 20 mg daily |
Good tolerability |
Rash G1 |
|
Jan-25 |
PET/CT + whole-body CT: complete radiologic/metabolic response |
Restaging |
Complete response (CR) |
– |
|
May-25 |
Follow-up assessment |
Continuing Afatinib 20 mg daily |
CR maintained; PS 1 |
No significant toxicity |
|
Oct-25 |
PET/CT + whole-body CT: stable disease in accordance with RECIST
criteria |
Continuing Afatinib 20 mg daily and loco-regional radiotherapy in
lymph nodes |
SD recist |
|
Table 1: Table 1 describes the patient’s clinical timeline, treatments performed, therapeutic response and associated toxicity.
Discussion
Uncommon EGFR mutations, including the S645C mutation, pose a challenge for clinicians in choosing the best treatment option. The patient in this study is an example of how Afatinib proves effective in this class of patients. Cases have been described in the literature in which the use of Osimertinib in patients with cooccurring EGFR L858R and S645C mutations led to the acquisition of resistance to third-generation TKIs [14]. This demonstrates the extreme heterogeneity of patients with NSCLC with target mutations, who therefore show different sensitivity to TKIs.
According to the literature, afatinib is the leading TKI used in the treatment of uncommon EGFR mutations. As demonstrated in the randomized ACHILLES/TORG1834 study, the first study to compare afatinib with chemotherapy in treatment-naïve NSCLC patients with uncommon sensitizing EGFR mutations (G719X, L861Q and S768I), afatinib provided a significant benefit in terms of progression-free survival, with a median PFS of 10.6 months compared to 5.7 months in the chemotherapy arm (HR 0.42) [15].
In this context, the clinical experience described in this article further supports the efficacy of afatinib in the case of ultra-rare EGFR mutations, such as S645C, where the use of a secondgeneration TKI led to a complete pathological response followed by prolonged disease stability exceeding 14 months, despite a dose reduction to 20 mg/day. The EGFR S645C mutation is defined as ultra-rare also because of its scarce mention in reviews on uncommon mutations in the literature. For this reason, as well as for the costs associated with more extensive NGS panels, many of the genomic analyses performed at patient diagnosis do not include this mutation within the standard panels.
Alongside afatinib, numerous TKIs are currently being studied for the treatment of NSCLC patients with uncommon EGFR mutations. These agents include novel and next-generation EGFR tyrosine kinase inhibitors, such as zipalertinib (CLN-081), lazertinib and sunvozertinib, which have shown signs of clinical and preclinical efficacy. Although many of these studies are still in the early stages, the available results appear promising for this class of patients and demonstrate how the therapeutic landscape for uncommon EGFR mutations is rapidly evolving [16-18].
The turning point in this patient’s disease history was the performance of the liquid biopsy. The NGS panel used in 2023 for molecular analysis of the biopsy sample did not include EGFR exon 17, where S645C is located. This generated a technical false negative, attributable to the inability to detect this mutation and prevent the use of a targeted therapy such as Afatinib from the outset. In this scenario, the B-FAST study was the diagnostic trigger that allowed the ultra-rare EGFR mutation to be detected. This highlights the indispensability of using broad NGS panels from the outset of diagnosis, without which there is a risk of missing targetable mutations [13].
A speculative hypothesis is that additional factors may have caused this mutation to appear at a later stage. In fact, it is possible that the immunotherapy administered for 10 months acted with selective pressure on the tumor clones, leading to greater proliferation of the clones that developed resistance to Atezolizumab and already expressed the EGFR S645C mutation [19].
Another factor that increases the rarity of the molecular profile of the patient described in this article is the finding of a germinal pathogenic BRCA2 variant. The mutation of a gene essential for DNA repair and homologous recombination leads to an increase in replication errors with a higher mutation index and an increase in clonality [20]. Some data in the literature suggest that genomic instability in tumors may facilitate the emergence of new drivers under the action of immunotherapy, which acts by increasing tumor plasticity and the adaptability of the cells that make-up the tumor mass.
Therefore, liquid biopsy allows indirect molecular analysis of the tumor mass when tissue is not obtainable, with the possibility of analyzing a broad tumor landscape if combined with the use of NGS panels that analyze a wide range of exons, especially those affected by rarer mutations, [21, 22] supporting the possibility of personalized therapy [23]. The patient underwent targeted treatment with Afatinib, which she would never have received if the EGFR S645C mutation had not been identified through ctDNA but, above all, thanks to the use of a broad NGS panel such as FoundationOne Liquid CDx, which analyses 324 genes [24].
Conclusion
It is essential to implement the use of NGS panels that are comprehensive and allow the identification of target mutations to ensure that patients can receive the best available treatment strategy. The case described here demonstrates this need, as comprehensive NGS analysis has allowed access to a treatment that has ensured a complete response and subsequent disease stability from the time the mutation was detected until now.
Declarations
Contributors: L.S. and D.C. were responsible for the clinical management of the patient and conceived the case report. L.P. and A.I. collected the clinical data, drafted the manuscript, and coordinated the integration of clinical and molecular findings. D.S. performed, curated, and interpreted the molecular analyses. D.C. and L.S. contributed to the interpretation of clinical data, supervised the clinical aspects of the work, and critically revised the manuscript for important intellectual content. All authors reviewed and approved the final version of the manuscript.
Competing interests: None
Patient consent for publication: Not applicable
Ethics approval and consent to participate: Not applicable
Availability of data and materials: Not applicable
Funding: No Funding
References
- Harvey RD, Adams VR, Beardslee T, Medina P (2020) Afatinib for the treatment of EGFR mutation-positive NSCLC: A review of clinical findings. J Oncol Pharm Pract 26:1461-1474.
- Harrison PT, Vyse S, Huang PH (2019) Rare epidermal growth factor receptor (EGFR) mutations in non-small cell lung cancer. Semin Cancer Biol 61: 167-179.
- Reck M, Rabe KF (2017) Precision diagnosis and treatment for advanced non–small-cell lung cancer. N Engl J Med 377: 849-861.
- Yang JC-H, Schuler M, Popat S, Miura S, Heeke S, et al. (2020) Afatinib for uncommon EGFR mutations in NSCLC: pooled analysis of LUX-Lung 2, 3, and 6. J Thorac Oncol 15: 803-815.
- Robichaux JP, Le X, Vijayan RSK, Hicks JK, Heeke S, et al. (2021) Structure-based classification predicts drug response in EGFR-mutant NSCLC. Nature 597: 732-737.
- Yang JC, Schuler M, Popat S, Miura S, Heeke S, et al. (2020) Afatinib for the Treatment of NSCLC Harboring Uncommon EGFR Mutations: A Database of 693 Cases. J Thorac Oncol 15: 803-815.
- Wang S, Shi G, Liu Q, Liu G, Liu Y, et al. (2025) Treatment of advanced lung adenocarcinoma with EGFR L833V/H835L compound mutations using furmonertinib: two case reports and literature review. Front Med (Lausanne) 12: 1658583.
- Ikeuchi, H, Hirose T, Ikegami M, Takamochi K, Suzuki K, et al. (2022) Preclinical assessment of combination therapy of EGFR tyrosine kinase inhibitors in a highly heterogeneous tumor model. Oncogene 41: 2470-2479.
- Berger AH, Brooks AN, Wu X, Shrestha Y, Chouinard C, et al. (2016) High-throughput Phenotyping of Lung Cancer Somatic Mutations. Cancer Cell 30: 214-228.
- Shen CI, Chang JC, Jain S, Olsen S, Wu CE (2024) Afatinib Plus Bevacizumab Treatment for a Patient With EGFR S645C-Mutant Non-Small Cell Lung Cancer: A Case Report. JCO Precis Oncol 8: e2400007.
- Bazan Russo TD, Pepe F, Gristina V, Gottardo A, Russo G, et al. (2025) Recent advances in liquid biopsy for precision oncology: emerging biomarkers and clinical applications in lung cancer. Future Oncol 21: 2803-2821.
- Mosele F, Remon J, Mateo J, Westphalen CB, Barlesi F, et al. (2020) Recommendations for the use of next-generation sequencing (NGS) for patients with metastatic cancers: a report from the ESMO Precision Medicine Working Group. Ann Oncol 31: 1491-1505.
- Dziadziuszko R, Mok T, Peters S, Han JY, Alatorre-Alexander J, et al. (2021) Blood First Assay Screening Trial (BFAST) in Treatment-Naive Advanced or Metastatic NSCLC: Initial Results of the Phase 2 ALKPositive Cohort. J Thorac Oncol 16: 2040-2050.
- Wang L, Quan F, Guo Z, Lu Z, Yang D, et al. (2023) Co-Occurring EGFR S645C and EGFR L858R in a Patient with Lung Adenocarcinoma Induced Primary Resistance to Osimertinib. Lung Cancer (Auckl) 14: 79-84.
- Miura S, Tanaka H, Misumi T, Yoshioka H, Tokito T, et al. (2025) Pragmatic Randomized Study of Afatinib Versus Chemotherapy for Patients With Non-Small Cell Lung Cancer With Uncommon Epidermal Growth Factor Receptor Mutations: ACHILLES/TORG1834. J Clin Oncol 43: 2049-2058.
- Dempke WCM, Fenchel K, Reinmuth N (2026) Zipalertinib-A Novel Treatment Opportunity for Non-Small Cell Lung Cancers with Exon 20 Insertions and Uncommon EGFR Mutations. Cancers (Basel) 18: 323.
- Park S, Ahn HK, Lee S, Min YJ, Kim J, et al. (2025) Lazertinib for Patients with NSCLC Harboring Uncommon EGFR Mutations: A Phase II Multicenter Trial. J Thorac Oncol 20: 1279-1288.
- Mitsudomi T (2025) Sunvozertinib: shining light on lung cancer‘s exon 20 fight. Transl Lung Cancer Res 14: 334-340.
- Wang H, Niu X, Jin Z, Zhang S, Fan R, et al. (2025) Immunotherapy resistance in non-small cell lung cancer: from mechanisms to therapeutic opportunities. J Exp Clin Cancer Res 44: 250.
- Haider S, Brough R, Madera S, Iacovacci J, Gulati A, et al. (2025) The transcriptomic architecture of common cancers reflects synthetic lethal interactions. Nat Genet 57: 522-529.
- Rolfo C, Mack P, Scagliotti GV, Aggarwal C, Arcila ME, et al. (2021) Liquid Biopsy for Advanced NSCLC: A Consensus Statement From the International Association for the Study of Lung Cancer. J Thorac Oncol 16: 1647-1662.
- Fernández-Galán E, Puig-Butillé JA (2025) Integrating ctDNA testing for EGFR analysis in advanced non-small cell lung cancer: strategies for clinical laboratories. Adv Lab Med 6: 233-244.
- Wang FY, Yeh YC, Lin SY, Wang SY, Chen PC, et al. (2025) Real-world application of targeted next-generation sequencing for identifying molecular variants in Asian non-small-cell lung cancer. BMC Cancer 25: 715.
- Woodhouse R, Li M, Hughes J, Delfosse D, Skoletsky J, et al. (2020) Clinical and analytical validation of FoundationOne Liquid CDx, a novel 324-Gene cfDNA-based comprehensive genomic profiling assay for cancers of solid tumor origin. PLoS One 15: e0237802.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.