Immune Surgery in Pancreatic Cancer Mice Model For Prolong Their Life And the Duration of Recurrence
by Jian Zhang1, Yang Yang2, Yan Han3, Yuxuan Fu1, Dong Chen3, Xiaoyu, Li6, Baofa Yu1-5*
1Jinan Baofa Cancer hospital, Jinan, Shandong Province, China 2Immune Oncology Systems, Inc, San Diego, CA, USA
3TaiMeiBaofa Cancer hospital, Dongping, Shandong Province, China
4Beijing Baofa Cancer Hospital, Beijing, China
5South China Hospital of Shenzhen University, Shenzhen, China
6 The affiliated Hospital of Qingdao University
*Corresponding Author: Baofa Yu, TaiMeiBaofa Cancer hospital, Dongping, Shandong Province, China
Received Date: 18 February 2026
Accepted Date: 23 February 2026
Published Date: 25 February 2026
Citation: Zhang J, Yang Y, Han Y, Fu Y, Chen D, Li X, Yu B (2026) Immune Surgery in Pancreatic Cancer Mice Model for Prolong Their Life and the Duration of Recurrence. J Surg 11: 11572 DOI: https://doi.org/10.29011/2575-9760.011572
Abstract
Objective: Surgery is an major methods for rectal cancer and is significantly improved thorough not only combine with modern medical equipment also combine with immune therapy. Awaking the immune cells can be effectively eliminated residual tumor cells following the surgical of tumor. Hapten Enhanced Intratumoral Chemotherapy (HEIC) has successful applied into cancer treatment. HEIC integrated into surgery is a new way to improve outcome of surgery while the immune response is elicited during of rectal surgery.
Method: After tumor cells were inoculated, the tumor volume was measured to be within the range of 100-200 mm3. The mice were randomly divided into groups (on the 0th day of the experiment). According to 0.1 ml per 100 mm3 of tumor volume, intratumoral administration of combination drug as HEIC was performed on the next day (on the 1st day of the experiment), plus surgery. Another group was performed with only surgery, or only drugs. It was comparison with survival time and cancer recurrence between immunosurgery and surgery.
Result: In this study, it showed that that the recurrence time in the immunosurgical group was later than that of the simple surgery group over 50 days (P<0.05), and the recurrence rate in the immunosurgical group was lower than that of the simple surgery group 6 times (P < 0.05) (Table 5). It showed that the survival time in the immunosurgery group was significantly longer than that of the other groups (P < 0.001).
Conclusion: It is approved that immunosurgery is a great way to replace the general cancer surgery and improve it from physical improvement to immunological improvement for control tumor recurrence to live longer.
Keywords: Cancer Immunotherapy; Drug Delivery; Immune Rectal Cancer Surgery; Immune Rectotomy; Intracellular Drug Delivery For Surgery; Intratumoral Injection
Introduction
The early 20th century marked the beginning of the development of cancer surgery techniques. In 1908, Mills performed the first abdominoperineal resection, in 1912 the first lobectomy, and in 1906, Werthem performed the first radical hysterectomy. All these surgeries were conducted in accordance with oncological standards. [1] For more than 100 years since the emergence of cancer treatment methods, surgery has played a significant role in cancer treatment. [2] Historically, the belief of surgeons has been that increasingly aggressive surgeries would improve the cure rate. Although we have witnessed the development of new technologies over the past 50 years, surgery remains the standard method for cancer treatment and is still the dominant approach in oncology, with no signs of change [2]. Surgery is the cornerstone of current rectal cancer treatment, with tumor cure and overall survival remaining the main goals. It requires multidisciplinary collaboration, and preoperative staging is crucial in the treatment plans of these patients [3].
Pancreatic cancer is an extremely high mortality rate. Over the past few decades, the 5-year survival rate for pancreatic cancer has remained largely unchanged, and in Western countries, it is expected to become the second leading cause of cancer-related deaths in the next decade. Pancreatic cancer patients are typically classified into four categories based on the severity of their condition: resectable, marginally resectable, locally advanced, and metastatic. [4,5] The 5-year recurrence rate is over 50%, 30% for stage I, and up to 80% for stage III. Recurrence types is local recurrence (1-2 years after surgery) and distant metastasis (2-3 years after surgery), there were 75% of local recurrence in analysis of 688 resected PDAC patients, the cumulative incidence of LM at 5 years was 41%, and patients who developed LM first (n = 194) had the lowest 1-year Overall Survival (OS) (34%), compared with 322 patients who developed extra-hepatic recurrence first (61%). [6] Eighty-nine studies with 17 313 patients undergoing PDAC resection were included. the median rates of initial recurrence were 20.8 per cent for locoregional sites, 26.5 per cent for liver, 11.4 per cent for lung and 13.5 per cent for peritoneal dissemination. The weighted median overall survival times were 19.8 months for locoregional recurrence, 15 months for liver recurrence, 30.4 months for lung recurrence and 14.1 months for peritoneal dissemination. [7]
In the past, the combination of chemotherapy, radiotherapy and surgery has been a major breakthrough in cancer treatment. Adjuvant chemotherapy and radiotherapy were used before surgery to shrink the tumors [8,9]. INTRAOPERATIVE RADIOTHERAPY (IORT) was also used during the surgery. Although IORT does not extend the overall survival period of patients undergoing surgery of pancreatic cancer, it shows good local regional control effects in specific stages of patients and does not increase the risk of complications [10-13]. However, due to the tendency to compress tumor remnants during surgery, tumor metastasis or residual tumor cells during the surgical process remain a clinical problem. Therefore, in many clinical cases, a successful surgery does not necessarily mean a successful treatment. To improve the longterm survival rate of surgical treatment and prevent local residual tumors and surgical residual metastasis caused by the surgery, many tools and surgical robots that improve the surgery and make it more perfect and successful have emerged. [14] The inflammatory response after colorectal cancer surgery was observed, and the proliferation, differentiation and secretion of immune cells in the intestinal area after microenvironment remodeling were discussed. The intestine is also the largest digestive organ in the human body. [15,16] These studies raised a question: How can we use the inflammatory response during the surgical process to control the regeneration of tumor cells and the recovery of tumors? Inducing the immune response triggered by semi-antigenenhanced intratumoral chemotherapy in esophagectomy provides a good example for exploring possible immunological surgery. [17] During the surgical process, it is necessary to stimulate the specific immune response of the body to residual cancer cells to simultaneously inhibit potential tumor metastasis. HaptenEnhanced Intratumoral Chemotherapy (HEIC) can trigger an immune response by stimulating the release of tumor-associated antigens modified by semi-antigens, thereby killing the tumor and promoting the presentation of new antigens [18,19]. HEIC can also enhance the functions of CD4, CD8, dendritic cells and B cells [20-23], it is also called Ultro-Minimum Incision Personalized Intratumoral Chemoimmunotherapy (UMIPIC). In this study, we aimed to determine whether Hapten-Enhanced Intratumoral Chemotherapy (HEIC) integrated into surgery for inducing an acute immune response in order to prevent tumor metastasis and tumor recurrence in animal model of pancreatic cancer. The drugs is injected into the tumor at site before remove the tumor and the cancer mass is completely removed 1 hour later after injection. HEIC was used for immune surgery rectal cancer and esophageal cancer during of surgery, before and after surgery, cancer tissue were took for sc-RNA-Seq analysis of the expression characteristics of malignant epithelial cells and immune cells, including bone marrow cells, stromal cells, T cells, plasma cells, B cells, platelets, epithelial cells, as well as the dynamic changes in cell percentage and cell subtype heterogeneity. [14,24]. It indicated that HEIC can be a tool for immune surgery to stimulate the immune systems in order to fight cancer cells and prevent the tumor metastasis. However, animal experiments are still necessary because they can provide a quicker assessment of the advantages of immunosurgery in treating tumors compared to conventional surgical treatment, in terms of survival time and the time of recurrence and metastasis.
Materials and Methods
- Cultivate Panc02 cells in vitro for animal tumor model.
- Establish tumor-bearing animal models: Collect the Panc02 cells cultivated in vitro, inoculate them subcutaneously on the right axilla of C57 mice, and select mice with tumors ranging from 5 to 7 mm in size to randomly group.
- Number of Mice and Grouping:
Experimental Method
After tumor cells were inoculated, the tumor volume was measured to be within the range of 100-200 mm3 (the longest diameter was approximately 7 mm, and the shortest diameter was approximately 5 mm). The mice were randomly divided into groups (on the 0th day of the experiment). The body weight and tumor size were measured, and the tumor volume V = 1/2ab2 was calculated (a is the longest diameter, and b is the shortest diameter). According to 0.1 ml per 100 mm3 of tumor volume, intratumoral administration was performed on the next day (on the 1st day of the experiment). The second administration was carried out on the 8th day of the experiment, with 0.1 ml per mouse, and 15 mice were used in each group (10 mice for survival time + 5 mice for efficacy). All mice were female. On the 15th day, blood was collected from the orbital area of mice numbered 11-15.
Observation Indicators
- Body Weight, Tumor Volume: Before grouping; before each administration; every 3 days after administration, weigh and measure once.
- Tumor Weight And Tumor Growth Inhibition Rate: At 15 days, dissect the tumor, weigh it, and calculate the tumor growth inhibition rate as a percentage using the following formula:Tumor growth inhibition rate (%) = × 100%.
- Evaluation Criteria: If the tumor growth inhibition rate (%) is less than 40, it is considered ineffective. If the tumor growth inhibition rate (%) is greater than or equal to 40 and the result is statistically significant with P < 0.05, it is considered effective.
The average tumor weight of the negative control group should be less than 1g, or 20% of the tumor weight should be less than 400mg, indicating poor tumor growth and the experiment should be invalidated.
- Organ Coefficient: At 15 days, dissect the thymus and spleen for weighing, and then calculate the thymus and spleen coefficients according to the following formula: Thymus coefficient: Thymus weight / Body weight × 100% Spleen coefficient: Spleen weight / Body weight × 100%
- Relative Tumor Proliferation Rate T/C (%): It is calculated according to the following formula based on the measurement of tumor size: T/C % = TRTV / CRTV*100% Efficacy evaluation criteria: If T/C is greater than 40%, it is considered ineffective; if it is less than or equal to 40% and statistically processed with P < 0.05, it is considered effective. TRTV: Treatment group RTV; CRTV: Negative control group RTV The formula for calculating Tumor Volume (TV) is: V = 1/2 × a × b². Among them a and b represent length and width respectively. Based on the measurement results, the Relative Tumor Volume (RTV) is calculated. The calculation formula is: RTV = Vt / V0. Here, V0 is the tumor volume measured when the cages were filled (i.e., d0), and Vt is the tumor volume measured each time.
- Survival Function and Life Extension Rate Detection Time: Throughout the entire experiment period Number of cases: Mice with survival periods from 1 to 10 Analysis method: SPSS statistical analysis of the survival function, and calculate the life extension rate % according to the following formula Life extension rate (%) = × 100%
Result of Experiment
Drug Efficacy Results
Drug Efficacy on Body Weight
The results showed that there was no significant difference in the body weight changes of the mice in each group from day 4 to day 25 (P > 0.05); on day 0 of the experiment, the body weight of the surgical group was higher than that of the immunosurgical group, the Dox+Ara control group and the model group (P < 0.05), and the body weight of the HEIC group was higher than that of the Dox+Ara control group (P < 0.05) (Table 1 and Figure 1).
|
Weight (g) |
0 Day |
4 Day |
8 Day |
12 Day |
16 Day |
20 Day |
25 Day |
|
Immunosurgery |
18.28±0.48* |
18.84±0.89 |
18.92±0.69 |
19.1±0.49 |
19.28±0.84 |
19.92±0.85 |
20.14±0.59 |
|
Surgery |
19.06±0.75 |
||||||
|
HEIC (UMIPIC) |
18.92±0.33 |
19.08±0.13 |
19.24±0.43 |
19.88±0.62 |
19.6±0.75 |
20.66±0.73 |
20.86±0.79 |
|
Dox+Ara control |
17.92±0.42*# |
18.72±0.47 |
18.72±0.47 |
18.98±0.56 |
18.82±0.5 |
19.76±0.52 |
20.18±0.46 |
|
Model Control |
18.32±0.66* |
18.94±0.46 |
19.2±0.5 |
19.32±0.6 |
19.58±0.27 |
19.86±0.32 |
20.14±0.22 |
|
F |
3.752 |
0.377 |
1.065 |
2.438 |
1.661 |
2.088 |
2.026 |
|
P |
0.02 |
0.771 |
0.392 |
0.102 |
0.215 |
0.142 |
0.151 |
Table 1: Comparison of Weight Changes Based on Pharmacodynamics.
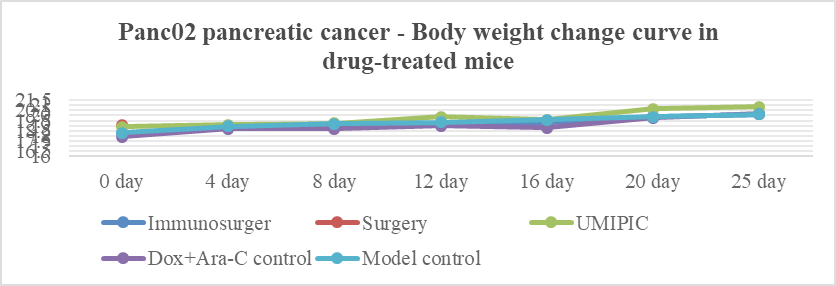
Figure 1: Drug efficacy experiment for pancreatic Cancer for weight change curve.
Therapeutic Effect and Tumor Volume
The results showed that on 0 day of the experiment, the tumor volume of mice in the HEIC group was smaller than that of the simple surgery group (P < 0.05); on days 12 and 16, the tumor volumes of mice in the model group and the Dox + Ara-C control group were significantly higher than those of the UMIPIC group (P < 0.05). On day 25 after the operation, the tumor volume of the model group was significantly higher than that of the UMIPIC group (P < 0.05) (Table 2 and Figure 2).
|
Tumor volume (mm3) |
0 day |
4 day |
8 day |
12 day |
16 day |
20 day |
25 day |
|
Immunosurgery |
117.67±15.87 |
||||||
|
Surgery |
154.85±68.11 |
||||||
|
HEIC (UMIPIC) |
102.02±16.14* |
127.47±44.3 |
174.94±52.21 |
72.73±30.47 |
71.45±22.44 |
115.34±68.42 |
81.41±67.42 |
|
Dox+Ara-C control |
121.67±11.79 |
148.4±26.14 |
178.76±47.54 |
206.29±86.75# |
203.42±94.15# |
252.91±138.21 |
306.07±196.28 |
|
Model control |
118.65±11.98 |
121.08±22.78 |
143.65±20.29 |
164.35±46.45# |
243.84±93.17# |
233.16±102.28 |
335.43±205.18# |
|
F |
1.73 |
0.968 |
1.031 |
6.595 |
6.755 |
2.424 |
3.401 |
|
P |
0.183 |
0.408 |
0.386 |
0.012 |
0.011 |
0.131 |
0.068 |
Table 2: Comparison of therapeutic effect and tumor volume changes.
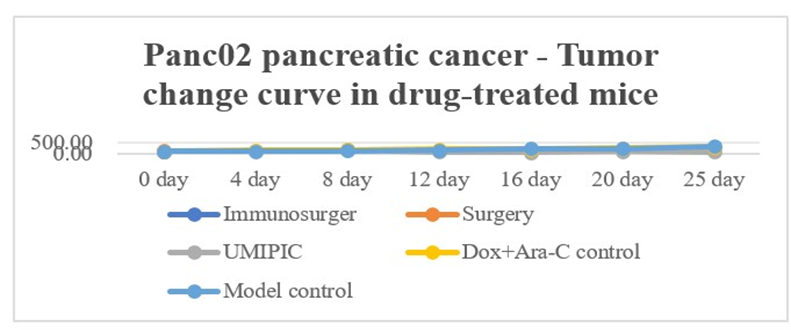
Figure 2: Changes in tumor volume due to pharmacodynamics.
TRTV: RTV of the treatment group; CRTV: RTV of the negative control group The formula for calculating tumor volume (tumor volume, TV) is: V = 1/2 × a × b². Among them A and B represent length and width respectively. Based on the measurement results, the Relative Tumor Volume (RTV) is calculated. The calculation formula is: RTV = Vt / V0. Here, V0 is the tumor volume measured when the drug is administered in separate compartments (i.e., d0), and Vt is the tumor volume measured in each individual measurement.
Survival Period Experiment Results
Survival Period Weight
The results showed that there was no significant difference in body weight between the UMIPIC group and the model group at the baseline (on the day of surgery, P > 0.05); on the 8th day after surgery, the body weight of the HEIC(UMIPIC(group was lower than that of the model group (P < 0.05); on the 16th day after surgery, the body weight of the Dox + Ara-C control group was lower than that of the model group (P < 0.05), and there was no significant difference in body weight among the groups at other time points (all P > 0.05). All the mice in the model group died on the 72nd day after surgery. See (Table 3 and Figure 3). The mice in the simple surgery group were used for supplementary experiments and had different measurement times and frequencies, so they were not included in the overall experimental statistical analysis. Specific data are shown in Table 3 and Figure 3.
Body weight (g) | 0 day | 4 day | 8 day | 12 day | 16day | 20 day | 25 day | 29 day | 33 day | 37 day | 41 day | 46 day | 51 day | 55 day | 60 day | 67 day |
Immunosurgery Group | 18.17±0.85 | 18.71±0.87 | 18.79±0.87 | 19.35±0.98 | 19.25±0.92 | 20.25±1.16 | 20.29±0.88 | 20.69±0.99 | 21.06±0.87 | 21.19±0.88 | 21.15±0.97 | 21.71±1.28 | 20.69±0.51 | 20.47±2.03 | 22.17±1.1 | 23.4±1.89 |
HEIC (UMIPIC) | 18.17±0.83 | 18.1±0.79 | 18.32±0.74* | 18.92±0.85 | 19.08±0.8 | 20.18±0.67 | 20.41±0.83 | 20.53±0.95 | 20.47±1.03 | 21.32±1.01 | 21.44±1.34 | 21.03±1.08 | 21.79±1.27 | 21.59±0.95 | 21.66±1.33 | 22.26±1.47 |
Dox+Ara-C Control | 18±0.82 | 18.64±1.28 | 18.65±1.24 | 18.84±1.05 | 18.58±1.07* | 19.93±1.21 | 20.26±1.3 | 20.51±1.22 | 20.16±1.19 | 21.74±1.22 | 21.4±1.36 | 21.59±1.54 | 22.14±1.78 | 22.08±2.27 | 22.15±3.19 | 23.57±2.37 |
Model | 18.28±0.49 | 18.92±0.51 | 19.26±0.39 | 19.54±0.49 | 19.66±0.41 | 20.39±0.4 | 20.98±0.68 | 20.78±0.93 | 20.98±0.97 | 21.36±1.09 | 21.4±1.14 | 21.96±1.85 | 22.23±2.22 | 21.22±2.39 | 22.9±0.96 | 22.85±0.92 |
F | 0.23 | 1.4 | 1.92 | 1.47 | 2.841 | 0.4 | 1.24 | 0 | 1.74 | 0.496 | 0.117 | 0.6 | 1.88 | 0.9 | 0.42 | 0.714 |
P | 0.88 | 0.3 | 0.14 | 0.24 | 0.052 | 0.7 | 0.31 | 1 | 0.18 | 0.688 | 0.949 | 0.6 | 0.16 | 0.5 | 0.74 | 0.559 |
Table 5: Changes in Body Weight of Mice during Survival Period.
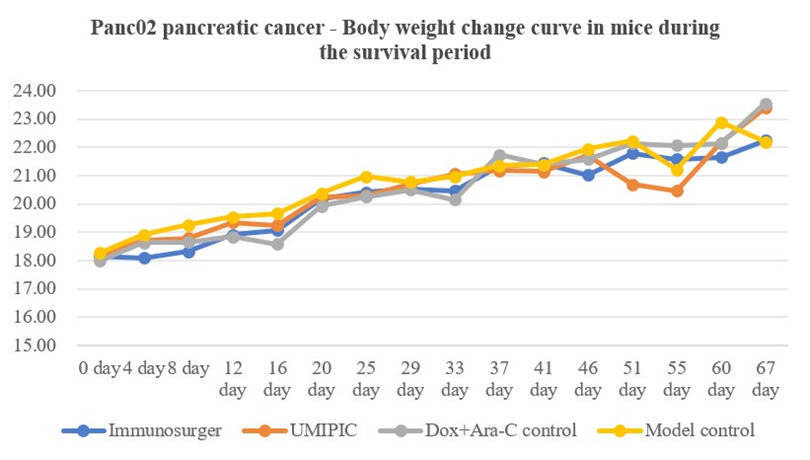
Figure 3: Changes in the body weight of mice during the survival period
Tumor Volume At the end of the Survival Period
The results showed that at the baseline (on the day of the surgery), the tumor volume of the mice in the immunosurgery group was larger than that of the other three groups (all P values < 0.05); on the 20th day and the 37th day after the surgery, the tumor volume of the mice in the HEIC (UMIPIC) group was significantly lower than that of the model group (P < 0.05), from the 25th day to the 33rd day after the surgery, the tumor volume of the UMIPIC group and the Dox+Ara-C control group was significantly lower than that of the model group (all P values < 0.05), on the 41st day after the surgery, the tumor volume of the UMIPIC group was significantly lower than that of the model group and the Dox+Ara-C control group (all P values < 0.05), on the 60th day after the surgery, the tumor volume of the HEIC group was significantly lower than that of the Dox+Ara-C control group (P < 0.05). See (Table 4 and Figure 4). On the 72nd day after the surgery, all mice in the model group died. The mice in the simple surgery group were used for supplementary experiments and had different measurement times and frequencies, so they were not included in the overall experimental statistical analysis. On the 10th day after the surgery, there was a recurrence in the simple surgery group, and the specific data are shown in (Table 4 and Figure 4).
|
tumor volume (mm3) |
0 day |
4 day |
8 day |
12 day |
16 day |
20 day |
25 day |
29 day |
33 day |
37 day |
41 day |
46 day |
51 day |
55 day |
60 day |
67 day |
|
Immunosurgery |
117.88±20.74 |
4 |
4 |
|||||||||||||
|
HEIC (UMIPIC) |
100.11±18.32* |
119.45±23.15 |
152.71±37.27 |
142.26±98.05 |
133.5±91.58 |
94.54±62.4# |
120.36±95.43# |
190.97±153.17# |
225.44±188.03# |
380.88±431.93# |
517.52±439.1#& |
757.09±675.25 |
1060.12±1017.6 |
1293.12±863.6 |
1333.86±918.25& |
1843.95±1145.26 |
|
Dox+Ara-C Control |
116.61±10.1* |
117.84±16.38 |
164.64±31.66 |
147.9±26.71 |
182.25±46.91 |
172.11±66.99 |
162.07±65.61# |
249.58±103# |
327.1±159.77# |
619.22±263.66 |
1072.29±503.14 |
1251.87±791.38 |
1344.98±552.85 |
2106.33±387.88 |
2523.8±739.92 |
2281.3±1078.67 |
|
Model |
116.06±18.73* |
120.3±26.23 |
145.08±46.79 |
203.81±122.98 |
222.77±179.67 |
274.68±179.74 |
316.26±197.22 |
433.95±262.4 |
571.02±287.77 |
757.4±422.45 |
1042.3±576.36 |
1320.32±1041.96 |
1911.15±1381.57 |
2205.39±1544.4 |
2336.03±424.5 |
2824.73±799.68 |
|
F |
2.318 |
0 |
0.636 |
1.37 |
1.4 |
6.02 |
6.11 |
4.686 |
6.59 |
2.51 |
3.7 |
1.186 |
1.27 |
1.55 |
3.2 |
0.608 |
|
P |
0.092 |
1 |
0.537 |
0.27 |
0.26 |
0.007 |
0.01 |
0.018 |
0.01 |
0.1 |
0 |
0.324 |
0.3 |
0.24 |
0.1 |
0.571 |
Note: Compared with the immunosurgery group, *P < 0.05. Compared with the model group, #P < 0.05; Compared with the Dox + Ara control group, &P < 0.05;
Table 4: Changes in tumor volume of mice at the end of the survival period.
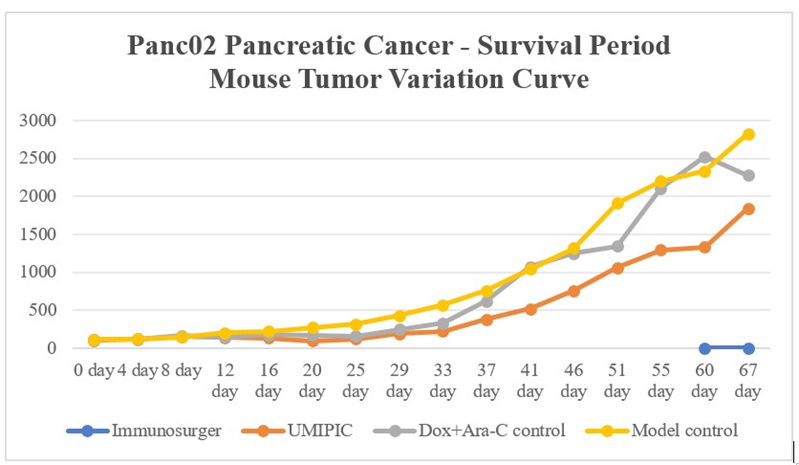
Figure 4: Changes in tumor volume of mice with shortened lifespan.
Recurrence Time of Surgical Mice
The results showed that the recurrence time of mice in the immunosurgical group was later than that of the simple surgery group over 50 days (P<0.05), and the recurrence rate of mice in the immunosurgical group was lower than that of the simple surgery group 6 times, with statistically significant differences (P < 0.05) (Table 5).
|
Group |
Recurrent time (days) |
Recurrent rate (%) |
|
Immuosurgery |
60 |
10.00 |
|
Surgery |
11.33±2.07 |
66.67 |
|
Z / Chi-square |
21.813 |
6.537 |
|
P |
0.000 |
0.011 |
Table 5: Recurrence Time of Surgical Mice
Survival Analysis
The results showed that the survival time of mice in the immunosurgery group was significantly longer than that of the other groups (P < 0.001), as shown in (Table 6 and Figure 5a). The survival time of mice in the immunosurgery group was significantly longer than that of the simple surgery group (P < 0.001), as shown in (Table 7 and Figure 5b).
|
Group |
Survival time (days) |
Median survival time (days) |
|
Immunosurgery |
108.40±11.01 |
no |
|
Surgery |
47.11±11.27 |
30.00±1.49 |
|
HEIC (UMIPI) |
69.70±6.32 |
65.00±11.86 |
|
Dox+Ara-C control |
60.20±3.69 |
57.00±3.87 |
|
Model |
68.53±4.63 |
57.00±7.11 |
|
Log Rank (Mantel-Cox) |
22.895 |
|
|
P |
0 |
Note: In the immunosurgery group, 9 out of 120 animals survived at 120 days, with a survival rate of 90%. Therefore, the median survival time could not be determined.
Table 6: Survival Time Analysis.
|
Group |
Survival time (days) |
Median survival time (days) |
|
Immunosurgery |
108.40±11.01 |
No conclusion |
|
Surgery |
47.11±11.27 |
30.00±1.49 |
|
Log Rank (Mantel-Cox) |
10.608 |
|
|
P |
0.001 |
Table 7: Survival Time Analysis of the Immunosurgery Group vs the Simple Surgery Group
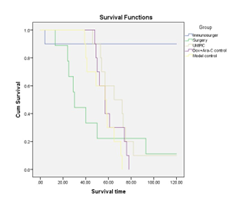
Figure 5a: Survival curves of each group of mice

Figure 5b: Survival curves of each group of mice.
Discussion
Pancreatic surgery is quite complex and has a high incidence of complications, so its development process has been relatively slow. After the initial anatomical research in the 16th century and the exploration of its physiology in the 3rd century, the development of pancreatic surgery began to accelerate. Pancreatic surgery started in the middle of the 19th century, when the surgical methods were relatively simple, such as external and internal drainage. The earliest pancreatic body and tail resection was performed in the late 19th century, while the more complex pancreatic head resection was gradually completed through multiple steps from 1898 to 1935. [25] However, the high complication rate associated with pancreatic resection surgery (mainly related to postoperative pancreatic fistula) has limited the development of pancreatic surgery for several decades. Since the 1980s, the concept of high-capacity centers emerged, which reduced the mortality rate and expanded the indications for surgery, especially for benign or inflammatory diseases. The management of postoperative complications has improved with the application of intensive care and interventional radiology techniques. At the same time, since the 1990s, pancreatic adenocarcinoma surgery has been completed through adjuvant chemotherapy, and then through neoadjuvant therapy, which has enabled more tumors to be removed and improved the prognosis of patients. In recent years, islet cell transplantation (as an alternative therapy for pancreatic transplantation) and minimally invasive surgery have expanded the possibility of non-invasive pancreatic surgery, thereby reducing the intensity of the surgery. [26] The application of robots in surgeries is continuously increasing, and it has great potential in pancreatic surgeries. Currently, the most researched robotic pancreatic surgeries are pancreaticoduodenectomy and distal pancreatectomy. Most of these studies are still in the early stages, but they report that robotic pancreatic surgeries are safe and feasible. Compared with open surgeries and laparoscopic surgeries, robotic pancreatic surgeries have several advantages. Data comparing the costs of robotic surgeries with traditional techniques are still lacking. Robotic pancreatic surgeries are still in the early stages, and there is no conclusion on whether they can reduce metastasis and recurrence. [27]. The inflammatory in cancer with the proliferation, differentiation and secretion of immune cells were observed within the intestinal region, and mucosal membranes of intestinal is a large immune organs.[13,14] How to use intraoperatively induced inflammatory responses to control tumor cells left over surgery is very important, HEIC has been successfully integrated into esophagectomy and rectal cancer and it has shown to effectively immune responses following surgery. [15,24] In earlier study, it was found a lot of 10 cell types, including Epithelial cells(EpithelialCells), Endothelial Cells (ECs), Fibroblasts, B cells(BCells), Plasma cells (PlasmaCells), TandNK cells (TandNK), Neutrophils, Mast cells (MastCells), Mononuclear phagocytes (MPs), Platelets at HEIC treatment 1 hour later after injection. The MHC-I molecular immune score of epithelial cells was significantly improved after treatment. [22] Under normal circumstances, epithelial cells would also express appropriate MHC-I molecules, which is used to present endogenous antigens to CD8+ T cells (i.e., cytotoxic T cells) to check for the presence of viral infection or other abnormal proteins. The significant increase in the expression of MHC-I class molecules may be due to the interaction of tumor antigens and the need to enhance the immune system’s recognition and clearance of these abnormal cells. [23].
Immune response in tumor almost immediate impact the peripheral blood samples, T&NK cells accounted for a large proportion up. GO pathway enrichment analysis showed the gene set enriched into tumor-related signaling pathways such as apoptosis, IL-7 and TNF. IL-7 is an important growth factor for T cell development and survival. TNF has the dual function of inducing apoptosis in certain cell types (including possibly T cells) [27] and inhibiting apoptosis by activating the expression of anti-apoptotic genes such as Bcl-2 via the NF-κB pathway. TEFF cytotoxicity scores in both of tumor tissues and blood samples were significantly improved after treatment, it indicated the tumor is cut out of body, then the immune response is set up in the body. TEFF killed tumor cells by releasing cytotoxic substances such as perforin and cytokines (IFN-γ, TNF-α), and cytotoxic T cells would release effector such as perforin and granase. Perforin forms holes in the target cell membrane, resulting in ion imbalance and changes in intracellular osmotic pressure, while granulozyme enters the target cell and induces programmed cell death [27]. HEIC therapy has showed the significant change in the frequency of clonotype between the two groups, the clonal diversity was significantly enhanced and indicated that the immune pool capacity of T cells including effector T cells was increased, it indicates the ability of effector T cells to recognize tumors was enhanced after drug administration, and patients may lack effective cloning and expansion of effector T cells before treatment [28].
After being stimulated by antigen, it is rapidly activated, proliferated, migrated to the site of inflammation, and releases immunoactive substances for immune response. These results suggest that the increased proportion and activity of effector T cells, as well as the efficient clonal expansion, may be the reason why immunotherapy can target tumors more effectively. HEIC treatment also strengthened the interaction between Fibroblasts, B cells(BCells), Plasma cells (PlasmaCells), TandNK cells (TandNK), Neutrophils, Mast cells(MastCells), Mononuclear phagocytes(MPs) through in the expression of MHC-I class molecules. Studies have shown that these genes have a negative impact on the prognosis of patients in the tumor microenvironment, [29] while CEACAM5 the interaction strength with CEACAM6 is up-regulated. According to reports, CEACM can transmit signals that produce multiple effects, including the activation of neutrophils and lymphocytes [30,31]. In this study, it showed that the body weight of the HEIC group was higher than that of the Dox+Ara-C control group (P < 0.05) (Table 1 and Figure 1). It indicated that mice lives well in health condition. It has showed that on 0 day of the experiment to 25 day, the tumor volume of mice in the HEIC group was smaller than that of the simple surgery group (P < 0.05); than the Dox + Ara-C control group (P < 0.05), the model group (P < 0.05) (Table 2 and Figure 2). It indicated that HEIC works well for control tumor growth. In this study, it showed that that the recurrence time of mice in the immunosurgical group was later than that of the simple surgery group over 50 days (P<0.05), and the recurrence rate of mice in the immunosurgical group was lower than that of the simple surgery group 6 times (P < 0.05) (Table 5). It showed that the survival time of mice in the immunosurgery group was significantly longer than that of the other groups (P < 0.001) (Tables 6,7 and Figure 5). It is approved that immunosurgery is a great way to improve of cancer surgery from physical improvement to immunological improvement for control tumor recurrence to live longer. In summary, our present study has demonstrated that the immunosurgery can induces the interaction among TandNK cells, MPs, and BCells, DC, Mast cells (MastCells) and (PlasmaCells), and immediate promotes the expression of a large number of genes contributing to the upregulation of immune response during rectal cancer surgery. Our study has provided an evidence that immunosurgry can replace general cancer surgery and make the general cancer surgery to be a great improvement with a systemic immune response during the process of cancer surgery, thus achieving an desirable clinical results.
Translational relevance to the manuscript
This research is a useful practical and valuable for medical relevance. The study presented in the manuscript provides evidence to support precision immune surgery during rectal surgery that can potentially be applied to pancreatic cancer and all other solid tumors.
Highlights
Single-cell RNA sequencing (scRNA-seq) is used to identify the immune reaction induced by hapten enhanced local chemotherapy during of cancer surgery. Immunosurgery can replace the general cancer surgery to be great improvement from physical to immunolgical improvment,which is overcome 200 years surgry limitation of metastasis and recurrence.
Authors statement:
Baofa, Yu, MD conceived the concept and provided overall supervision of all experiments and write.
Jian Zhang, MD did a ll of animal experiment for this research
Yang Yang, did write in English for the second version
Yan Han, MS, did most of analysis for experiment
Dong Chen, MD did assistant for daily care of mice and experiment
Conflict of Interest Statement:
All of authors do not have any conflict interest for the research.
Ethical Statement:
All procedures and protocols in the study have been reviewed and approved by the Ethical Committee of the Beijing Baofa Cancer Hospital (TMBF 0010, 2015). All informed consent forms from patients have been signed prior to the start of the study. Ethical Approval: approved by the Ethical Committee of the Beijing Baofa Cancer Hospital (TMBF 0010, 2015). Funding Sources:
Data Availability Statement
The data that support the findings of this study are available from [third party name] but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of [third party name].
There is not outside funding for this research.
Financial support
This study was sponsored in part by Tai Mei Baofa Cancer Hospital, Dongping, Shandong Province, China 271500.
References
- Arruebo M, Vilaboa N, Berta Sáez-Gutierrez B (2011) Assessment of the evolution of cancer treatment therapies. Cancers (Basel) 3: 32793330.
- Wyld L, Audisio RA and Poston GJ (2015) The evolution of cancer surgery and future perspectives. Nature Reviews Clinical Oncology 12: 115-124.
- M McCourt, J Armitage, JRT Monson (2009) Rectal cancer. Surgeon 7: 162-169.
- Mizrahi JD, Surana R, Valle JV (2020) Pancreatic cancer. Lancet 395: 2008-2020.
- Neoptolemos JP, JKleeff J, Patrick Michl P (2018) Therapeutic developments in pancreatic cancer: current and future perspectives. Nat Rev Gastroenterol Hepatol 15: 333-348.
- Zambirinis CP, Midya A, Chakraborty J (2022) Recurrence After Resection of Pancreatic Cancer: Can Radiomics Predict Patients at Greatest Risk of Liver Metastasis? Surg Oncol 29: 4962-4974.
- Tanaka M, Mihaljevic AL, Probst P (2019) Meta-analysis of recurrence pattern after resection for pancreatic cancer. Br J Surg 106: 15901601.
- Mo DC, Liang Z, Chen L (2022) Efficacy and safety of adjuvant therapy with PD-1/PD-L1 inhibitors in cancer. Exp Ther Med 24: 749.
- Coccolini F, Nardi M, Montori G (2018) Neoadjuvant chemotherapy in advanced gastric and esophago-gastric cancer. Meta-analysis of randomized trials. Int J Surg 51: 120-127.
- Gao P (2017) Intraoperative radiotherapy in gastric and rectal cancer cancer: meta-analysis of long-term outcomes and complications. Minerva Med 108: 74-83.
- Wang Q, Lang J, Li T (2020) Postoperative adjuvant chemotherapy versus chemoradiotherapy for node-positive rectal cancer squamous cell carcinoma: a propensity score-matched analysis. Radiat Oncol 15: 119.
- Marcos-Pablos S, José García-Peñalvo F (2022) More than surgical tools: a systematic review of robots as didactic tools for the education of professionals in health sciences. Adv Health Sci Educ Theory Pract 27: 1139-1176.
- Kampman SL, Smalbroek BP, L M Dijksman LM (2023) Postoperative inflammatory response in colorectal cancer surgery: a meta-analysis. Int J Colorectal Dis 38: 233.
- Zhou B, Yuan Y, Zhang S (2020) Intestinal Flora and Disease Mutually Shape the Regional Immune System in the Intestinal Tract. Front Immunol 11: 575.
- Yu BF, Zhang J, Gao Z (2023) Immune Reaction Induced with Hapten Enhanced Intratumoral Chemotherapy during Esophagectomy: Explore Possible Immunosurgery. Journal of Cancer Science and Clinical Therapeutics 7: 259-270.
- Adair K, Meng X, and Naisbitt DJ (2021) Drug hapten-specific T-cell activation: Current status and unanswered questions. PROTEOMICS 21: e2000267.
- Yu B, Lu Y, Gao F (2015) Hapten-enhanced therapeutic effect in advanced stages of lung ancer by ultra-minimum incision personalized intratumoral chemoimmunotherapy therapy. Lung Cancer (Auckl) 6: 1-11.
- Yu B, Fu Q, Han Y Zhang J Chen D (2022) An Acute Inflammation with Special Expression of CD11 & CD4 Produces Abscopal Effect by Intratumoral Injection Chemotherapy Drug with Hapten in Animal Model. J Immunological Sci 6: 1-9.
- Yu B, Fu Q (2020) Drug Mixed by H2O2 Injection Intratumoral to turning an Extracellular Matrix into Autologous Coagulum as Drug Depot. Novel Research in Sciences 2020 4(2).
- Yu B, Han Y, Fu Q (2023) Awaken Immune Cells by Hapten Enhanced Intratumoral Chemotherapy with Penicillin Prolong Pancreatic Cancer Survival. Journal of Cancer 14: 1282-1292.
- Yu B, Fu Q, Han Y, Zhang J, Chen D (2022) An Acute Inflammation with Special Expression of CD11 & CD4 Produces Abscopal Effect by Intratumoral Injection Chemotherapy Drug with Hapten in Animal Model. J Immunological Sci 6: 1-9.
- Kechin A, Boyarskikh U, Kel A (2017) cutPrimers: A New Tool for Accurate Cutting of Primers from Reads of Targeted Next Generation Sequencing. J Comput Biol 24: 1138-1143.
- Yu B, Gao F, Jing P (2023) Cancer Immunotherapy Preparation and Immune Cells Activation through Hapten-Enhanced Chemotherapy in Primary Lung Cancer. J Basic Clin Pharma 14: 10-16.
- Dobin A, Davis CA, Schlesinger F (2013) STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29: 15-21.
- Yu BF, Zhang J, Gao F (2024) Using Scrna-Seq to Deffne the Immune Response after Hapten Enhanced Intratumoral Chemotherapy on Cases of Rectal cancer: Exploring Immune Surgery. Journal of Surgery. J Surg 9: 11057
- Belghiti J, Sauvanet A (2026) Ancient and Modern History of Pancreatic Surgery. Pancreas 55: e21-e29.
- Khachfe HH, Habib JR, Harthi Sal (2022) Robotic pancreas surgery: an overview of history and update on technique, outcomes, and financials. J Robot Surg 16: 483-494.
- Guo W, Zhang C, Xia Wang X (2022) Resolving the difference between left-sided and right-sided colorectal cancer by single-cell sequencing. JCI Insight 7: e152616.
- Wang S, Qu Y, Xia P (2020) Transdifferentiation of tumor infiltrating innate lymphoid cells during progression of colorectal cancer. Cell Res 30: 610-622.
- Li E, Yang X, Du Y (2021) CXCL8 Associated Dendritic Cell Activation Marker Expression and Recruitment as Indicators of Favorable Outcomes in Colorectal Cancer. Front Immunol 12: 667177.
- Schrand B, Clark E, Levay A (2018) Hapten-mediated recruitment of polyclonal antibodies to tumors engenders antitumor immunity. Nature Communications 9: 3348.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.