Effectiveness and Safety of Triple Combination of Sitagliptin + Pioglitazone + Metformin In T2DM Patients: a Retrospective, Multi-Centre Study
by Alok Agarwal1, Mehul Sundesha2, Manoj Mali3, Sanjay Gupta4, Sumit Bhushan5*, Sanjay Chaudhari5, Rahee Borulkar5, Prajakta Bhosale5, Rujuta Gadkari5, Saiprasad Patil5
1Apollo Hospital, Delhi, India
2 Sundesha Healthcare, Mumbai, India
3MK Diet and Cure Clinic, Jaipur, India
4Diabetes and Liver Care, Kolkata, India
5Glenmark Pharmaceuticals Ltd, India
*Corresponding author: Sumit Bhushan, Glenmark Pharmaceuticals Ltd., India.
Received Date: 02 March, 2026
Accepted Date: 09 March, 2026
Published Date: 12 March, 2026
Citation: Agarwal A, Sundesha M, Mali M, Gupta S, Bhushan S, et al. (2026) Effectiveness and Safety of Triple Combination of Sitagliptin + Pioglitazone + Metformin In T2DM Patients: a Retrospective, Multi-Centre Study. Curr Trends Intern Med 10: 251. DOI: https://doi.org/10.29011/2638-003X.100251
Abstract
Background: Type 2 diabetes mellitus (T2DM) often requires combination therapy for adequate glycemic control. The fixeddose combination (FDC) of Sitagliptin, Pioglitazone, and Metformin offers complementary mechanisms of action that target insulin resistance, hepatic glucose output, and the incretin pathways. Objective: To evaluate the effectiveness and safety of Sitagliptin, Pioglitazone, and Metformin in T2DM patients inadequately controlled on mono or dual therapy. Methods: A retrospective, multicentre database analysis was conducted on 1380 adult patients with T2DM across 186 sites in India. Patients who had received the FDC for at least 3 months were included. The primary endpoints were changes in glycemic parameters (HbA1c, fasting plasma glucose [FPG], and postprandial plasma glucose [PPG]); secondary endpoints included lipid, hepatic, and renal parameters, body weight, and adverse events. Statistical analysis used descriptive methods and paired t-tests. Results: After 3 months of Sitagliptin, Pioglitazone, and Metformin FDC therapy, significant improvements were observed in HbA1c (8.35 ± 0.96% to 7.57 ± 0.95%; p<0.0001), FPG (164.01 ± 38.84 mg/dl to 139.28 ± 30.77 mg/dl; p<0.0001), and PPG (244.88 ± 62.69 mg/dl to 202.98 ± 43.79 mg/dl; p<0.0001). The lipid profile showed numerical reductions in total cholesterol, LDL, and triglycerides at 3 months. HDL levels decreased slightly; however, the clinical relevance of this change remains uncertain. Renal function showed a slight decline in eGFR (91.6 to 87.5; p=0.0102), but serum creatinine (1.05 to 0.92 mg/dl; p<0.0001) and blood urea nitrogen (18.54 to 14.46 mg/dl; p<0.0001) improved. Hepatic parameters remained stable. Adverse events were reported in 6.3% of patients, most commonly hypoglycemia (2.54%), breathlessness (3.62%), and pedal edema (1.16%); other mild symptoms (0.87%) were also noted. No serious adverse events occurred. Conclusion: Sitagliptin, Pioglitazone, and Metformin FDC significantly improved glycemic control and metabolic parameters over 3 months, with a favorable safety profile, supporting their role as effective treatments for T2DM patients inadequately controlled on mono- or dual-therapy.
Keywords: Type 2 Diabetes Mellitus, Sitagliptin, Pioglitazone, Metformin, Fixed-Dose Combination, Glycemic Control, Realworld Evidence, Safety Profile, Retrospective Study, Indian Population
Introduction
Type 2 diabetes mellitus (T2DM) is a major and growing publichealth concern in India. According to the International Diabetes Federation (IDF)’s 2025 Diabetes Atlas, 589 million adults (20-79 years) worldwide live with diabetes, roughly 1 in 9 adults, and this number is predicted to rise to 853 million by 2050 [1]. It is characterized by a non-autoimmune, heterogeneously progressive decline in pancreatic β-cell insulin secretion, often occurring in conjunction with insulin resistance (IR) and metabolic syndrome (MS) [2]. Impaired β-cell function reduces insulin secretion and limits the body’s ability to maintain normal glucose levels, while insulin resistance promotes excessive hepatic glucose production and diminishes glucose uptake in muscle, liver, and adipose tissue. Although both defects contribute early in disease development, β-cell dysfunction tends to be more pronounced. Several antidiabetic agents have been developed to address these underlying defects, thereby enhancing glycemic control in T2DM [3].
Metformin remains the cornerstone of initial pharmacological therapy for type 2 diabetes mellitus (T2DM) due to its efficacy, safety, weight neutrality, cardiovascular benefits, and costeffectiveness. However, because T2DM is a progressive disease characterized by declining β-cell function and increasing insulin resistance, metformin monotherapy often fails to sustain adequate glycemic control over time and frequently falls short of adequately correcting dyslipidemia [4]. Hyperglycemia and dyslipidemia are the major metabolic defects of focus in Type 2 diabetes mellitus patients, which cannot be addressed solely by a single drug [5,6].
The U.S. study found that approximately 42% of patients experienced secondary metformin failure over a 2- to 5-year period [7]. This rising incidence of secondary treatment failure underscores the growing need for early initiation of combination therapy, particularly with agents that have complementary mechanisms of action, such as DPP-4 inhibitors, SGLT-2 inhibitors, GLP-1 receptor agonists, thiazolidinediones, or basal insulin. Current international and Indian guidelines therefore recommend combination therapy in patients who do not achieve the target HbA1c with metformin alone, to improve long-term glycemic durability and reduce the risk of complications [4, 8].
Sitagliptin, a dipeptidyl peptidase-4 (DPP-4) inhibitor, increases the levels of active incretin hormones (GLP-1 and GIP), thereby stimulating glucose-dependent insulin secretion and suppressing glucagon release. This, in turn, reduces hepatic glucose production and improves postprandial glycemia [9,10]. Pioglitazone, a thiazolidinedione, acts as a peroxisome proliferator-activated receptor-γ (PPAR-γ) agonist, improving insulin sensitivity in adipose tissue, skeletal muscle, and the liver, while also preserving β-cell function and reducing hepatic glucose output [11, 12].Thus, pioglitazone is frequently prescribed as an add-on therapy when metformin, DPP-4 inhibitors, GLP-1 receptor agonists, or their combinations fail to achieve adequate glycemic control [13].
A 16-week randomized controlled trial in patients inadequately controlled on metformin demonstrated that both sitagliptin (100 mg) and pioglitazone (30 mg) as add-on therapies significantly lowered HbA1c from baseline, with sitagliptin showing additional benefits in body weight control compared with weight gain with pioglitazone [14]. Additionally, the recent IMPACT study conducted in India compared a fixed-dose combination of triple therapy (FDC, metformin/sitagliptin/pioglitazone) vs. dual therapy (metformin/sitagliptin) in patients with HbA1c between 8-11% who were insufficiently controlled with metformin alone; triple therapy showed a greater HbA1c reduction (−1.64% vs. −1.32%) and superior fasting and postprandial glucose improvements [15].
Although randomized controlled trials, including the Indian IMPACT study [16], have demonstrated the efficacy and tolerability of the sitagliptin, metformin, and pioglitazone combination, largescale, multicentre real-world data evaluating its effectiveness and safety in routine Indian clinical practice remain limited.
Given the substantial burden of T2DM in India and the common f inadequacy of monotherapy or dual therapy to achieve optimal glycemic control, it is essential to assess the clinical effectiveness, safety, and metabolic impact of the combination of sitagliptin, pioglitazone, and metformin. The present study seeks to fill this gap by providing real-world evidence derived from a large, multicentric cohort.
Materials and Methods Study Details
We conducted a retrospective, observational, single-arm, multicentre study to evaluate the effectiveness and safety of a triple combination of sitagliptin, pioglitazone, and metformin in patients diagnosed with T2DM across India. The study was conducted in compliance with the International Conference on HarmonizationGood Clinical Practice Guidelines (ICH-GCP), the Declaration of Helsinki, and local regulatory requirements (Indian Good Clinical Practice, the Indian Council of Medical Research (ICMR), and the New Drugs & Clinical Trials Rules, 2019).
Study Population and Sample Size
Patients aged 18 years and older diagnosed with type 2 diabetes mellitus were assessed for eligibility during their first visit. The inclusion criteria comprised adults with T2DM who had been prescribed the FDC of Sitagliptin, Pioglitazone, and Metformin for at least 3 months and who had inadequate glycemic control at initiation, defined as an HbA1c level of ≥8.0% prior to starting the triple combination therapy.
Patient case records that were incomplete and deemed unsuitable for inclusion in the study by the investigator were excluded from the trial. Since there was no hypothesis to be proven, no formal sample size calculation was made for the study.
Study Conduct
The FDC used was Sitazit-Trio (Sitagliptin 100 mg, Pioglitazone 15 mg, and Metformin 1000 mg), administered as a single tablet once daily. This retrospective, observational, single-arm, multicentre study aimed to evaluate the effectiveness and safety of the triple combination of sitagliptin, pioglitazone, and metformin in patients diagnosed with T2DM.
After the eligibility criteria were confirmed, patient-level data were analysed. Data on demographic characteristics, medical history, and switch patterns from mono- or dual-therapy to the FDC were evaluated against predefined efficacy and safety endpoints.
Data were collected using an Electronic Case Report Form (eCRF) within a secure Electronic Data Capture (EDC) system. The system provided role-based access to authorized users, including Principal Investigators (PIs) and study coordinators. Each investigator was responsible for completing a predefined number of patient cases at their respective study sites.
Efficacy analysis
The primary endpoint was the mean change in glycemic parameters, including Glycosylated hemoglobin (HbA1C), Fasting plasma glucose (FPG), and Postprandial plasma glucose (PPG), at 3 months from the first visit (baseline). Secondary endpoints were mean change in lipid parameters like LDL, HDL, Triglycerides (TG) and Total cholesterol (TCH) at 3 months from 1st visit (baseline), mean change in hepatic parameters like Alanine transaminase (ALT), Aspartate transaminase (AST), Alkaline phosphate (ALP), Gamma-glutamyl transferase (GGT), Albumin and Total bilirubin at 3 months from 1st visit (baseline) and mean change in renal parameters like eGFR, serum creatinine, serum albumin, Creatinine clearance, Blood urea nitrogen at 3 months from 1st visit (baseline).
Safety Analysis
The safety assessment included analyses of treatment-emergent adverse events, serious adverse events, and laboratory parameters. It also consisted of the percentage of patients experiencing serious adverse events (SAE) leading to discontinuation of the treatment.
Statistical Analysis
All data will be summarized using descriptive statistics, including the number of subjects, mean, standard deviation (SD), minimum, median, and maximum for continuous variables, and frequency and percentage for categorical variables. Descriptive statistics were used to summarize the data. Categorical variables were presented as absolute numbers and percentages, while continuous variables were expressed as mean ± standard deviation (SD). A t-test for two samples assuming unequal variances (Welch’s t-test) was performed to compare pre- and post-treatment values for continuous variables, including Glycaemic Parameters, renal and hepatic parameters, and lipid profile markers. Statistical significance was determined using a two-tailed test, with a p-value of <0.05 considered significant.
Results Demographic and Baseline Characteristics
Data from 1380 patients were collected across 186 clinical sites across India. The mean age was 55.81 ± 10.59 years, and the average weight was 73.80 ± 19.60 kg. The gender distribution comprised 1031 males (74.71%) and 349 females (25.29%). The mean duration of T2DM was 7.71 ± 5.59 years, indicating that most patients had a long-standing history of diabetes. Baseline vitals showed a mean systolic blood pressure (SBP) of 135.48 ± 19.26 mmHg, a mean diastolic blood pressure (DBP) of 91.36 ± 16.61 mmHg, and a mean pulse rate of 86.04 ± 30.02 bpm. Baseline demographic and vital characteristics of the study population are summarized in (Table 1 and 2).
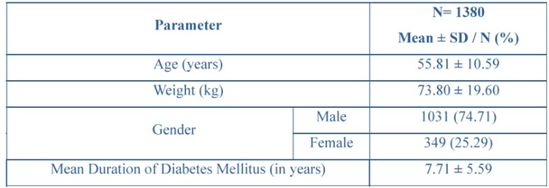
Table 1: Demographic characteristics of the study participants
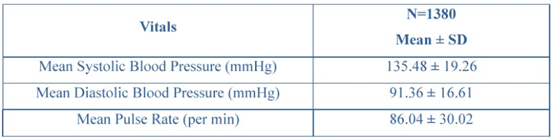
Table 2: Vital characteristics of the study participants
Treatment History
Among the study population, 711 (51.52%) patients were on dual therapy, and 632 (45.80%) were on monotherapy before switching to the triple fixed-dose combination (FDC) of Sitagliptin, Pioglitazone, and Metformin. Treatment adjustments were common, with 788 (57.10%) patients having switched medications 2 to 5 times, and 27 (1.96%) patients requiring more than 5 switches before initiating with the FDC. Lifestyle modifications were advised to 950 (68.84%) patients, with dietary changes (449, 47.26%) and physical exercise (408, 42.95%) being the most frequently recommended interventions. Details of prior treatment history, medication switching, and lifestyle advice provided to patients are summarized in (Table 3).
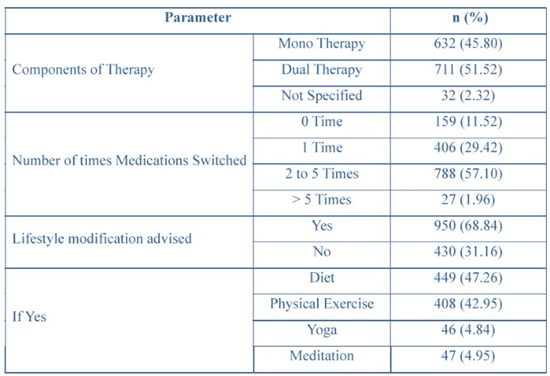
Table 3: Treatment history of medication switching and lifestyle advice
Comorbidities in study participants
The study population of 1380 patients showed a wide range of comorbid conditions. The most prevalent condition was hypertension, affecting 350 (25.36%) patients, with a mean duration of 6.45 ± 4.46 years. Obesity was present in 235 (17.03%) patients, with a mean duration of 7.16 ± 5.54 years. Other common conditions included cardiovascular disease in 137 (9.93%) patients, with a mean duration of 7.05 ± 5.40 years, and dyslipidaemias in 78 (5.65%) patients, with a mean duration of 4.82 ± 3.25 years. Regarding chronic kidney disease (CKD), 15 (1.09%) patients were diagnosed with varying stages of CKD, with the percentage distribution across stages calculated based on the total CKD population. Among these, 8 (53.33%) were in CKD Stage G1, 1 (6.67%) in CKD Stage G2, 3 (20.00%) in CKD
Stage G3a, 1 (6.67%) in CKD Stage G3b, and 2 (13.33%) in CKD Stage G4. No patients were classified in CKD Stage G5. The mean duration of CKD in this cohort was 6.47 ± 4.78 years. Other notable conditions included asthma in 69 (5.00%) patients, with a mean duration of 6.63 ± 5.16 years, and anxiety/depression in 82 (5.94%) patients, with a mean duration of 3.68 ± 3.48 years. The prevalence of comorbidities among study participants is presented in (Table 4).
|
Therapeutic Area |
n (%) |
Mean Duration (Years) (Mean ± SD) |
|
Hypertension |
350 (25.36) |
6.45 ± 4.46 |
|
Dyslipidaemia |
78 (5.65) |
4.82 ± 3.25 |
|
Obesity |
235 (17.03) |
7.16 ± 5.54 |
|
Cardiovascular disease |
137 (9.93) |
7.05 ± 5.40 |
|
Asthma |
69 (5.00) |
6.63 ± 5.16 |
|
Obstructive sleep apnea |
25 (1.81) |
5.54 ± 3.32 |
|
Metabolic associated |
26 (1.88) |
5.38 ± 3.44 |
|
fatty liver disease (MAFLD) |
||
|
Chronic kidney disease |
15 (1.09) |
6.47 ± 4.78 |
|
CKD Stage G1 |
8 (53.33) |
— |
|
CKD Stage G2 |
1 (6.67) |
— |
|
CKD Stage G3a |
3 (20.00) |
— |
|
CKD Stage G3b |
1 (6.67) |
— |
|
CKD Stage G4 |
2 (13.33) |
— |
|
CKD Stage G5 |
0 (0.00) |
— |
|
Anxiety/Depression |
82 (5.94) |
3.68 ± 3.48 |
|
Others |
15 (1.09) |
5.13 ± 4.58 |
|
Weakness |
1 (6.67) |
— |
|
Hyperthyroidism |
1 (6.67) |
— |
|
Not specified |
13 (86.67) |
— |
Table 4: Comorbidities in study participants
Clinical Outcomes Glycemic Control
Among the 921 patients, the mean Fasting Blood Glucose (FBG) level decreased from baseline (164.01 ± 38.84 mg/dL) to 139.28 ± 30.77 mg/dL at 3 months (P < 0.0001), indicating a significant improvement in fasting blood sugar levels. The Postprandial Blood Glucose (PPG, n=923) level also decreased from 244.88 ± 62.69 mg/dL to 202.98 ± 43.79 mg/dL (P < 0.0001), demonstrating a marked reduction in postprandial glucose levels. HbA1C levels significantly improved from 8.35 ± 0.96% to 7.57 ± 0.95% (n=905, P < 0.0001), indicating better long-term glycemic control. A significant reduction in HbA1c, FPG, and PPG levels was observed at 3 months compared to baseline (Figures 1 and 2).
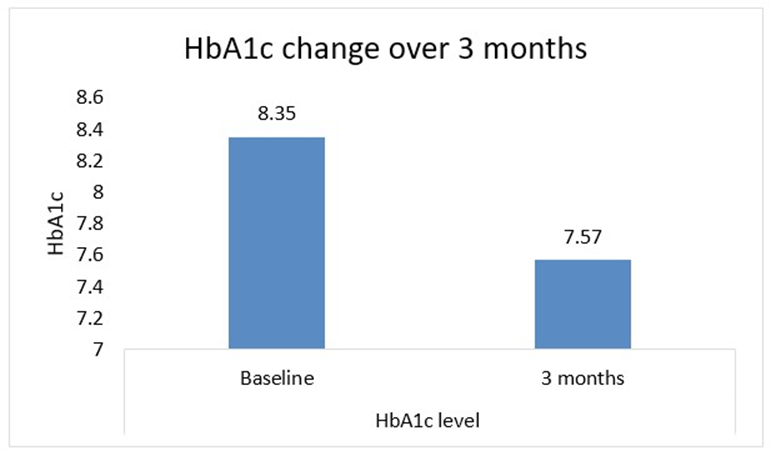
Figure 1: HbA1c change from baseline to 3 months
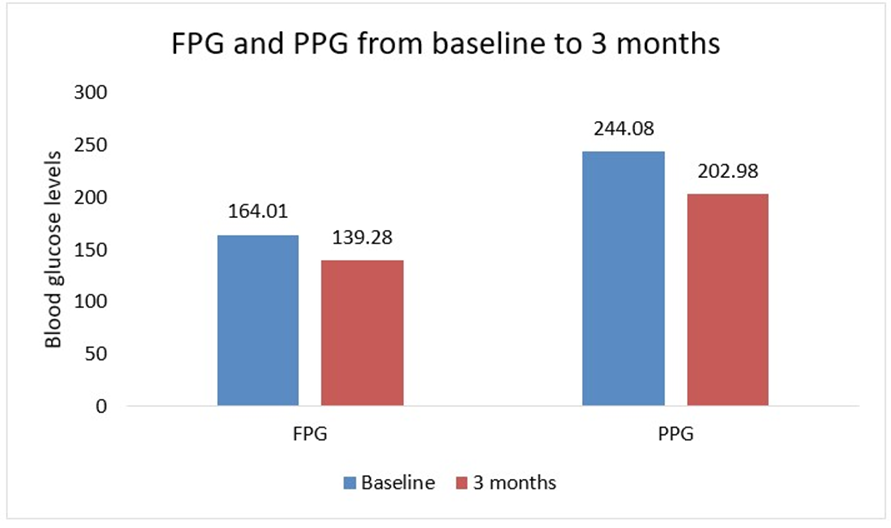
Figure 2: FPG and PPG change from baseline to 3 months
Renal Function
In the analysed population (n = 165), eGFR declined slightly from 91.59 ± 16.15 to 87.53 ± 13.21 (P = 0.0102), although the difference was statistically significant. Serum Creatinine improved from 1.05 ± 0.25 mg/dl at baseline to 0.92 ± 0.24 mg/dl at 3 months (n=154, P < 0.0001), indicating better renal health. Other renal parameters, including Creatinine clearance (n=19) and Blood Urea Nitrogen (BUN, n=96), remained stable or improved, with BUN significantly decreasing from 18.54 ± 5.99 mg/dl to 14.46 ± 2.05 mg/dl (P < 0.0001). Renal function parameters, including serum creatinine and eGFR, remained stable over the study period (Figures 3 and 4).
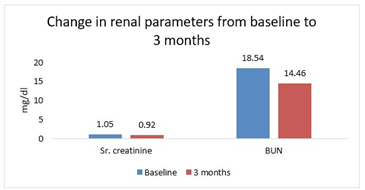
Figure 3: Creatinine and BUN change from baseline to 3 months
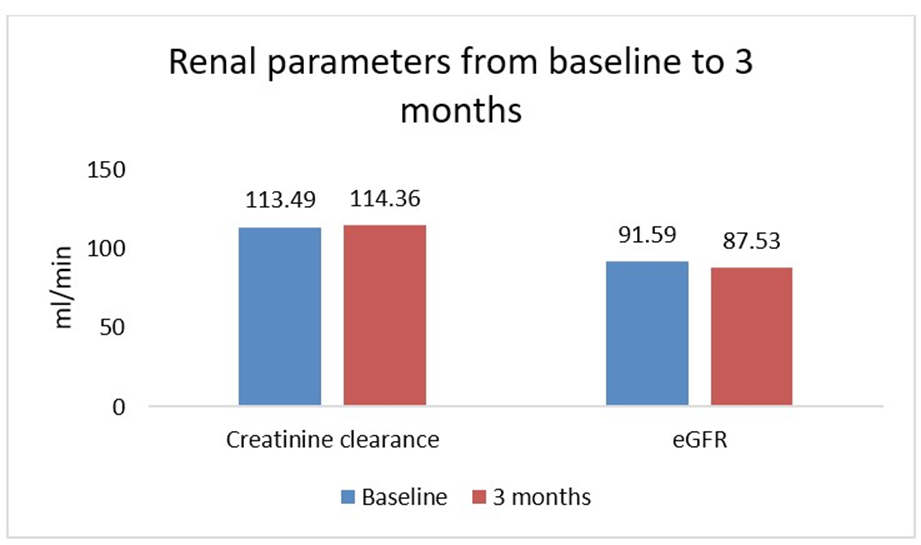
Figure 4: Creatinine clearance and eGFR change from baseline to 3 months
Lipid Profile
Among 1560 patients, there was a significant improvement in serum cholesterol levels, with total cholesterol decreasing from 184.39 ± 37.49 mg/dL to 158.65 ± 36.23 mg/dL (n=153, P < 0.0001) and LDL cholesterol decreasing from 95.58 ± 18.94 mg/dL to 83.69 ± 22.55 mg/dL (n=150, P < 0.0001). HDL cholesterol, however, showed a decrease from 55.75 ± 28.53 mg/dl to 46.52 ± 25.88 mg/dl (n=149, P = 0.0019). Triglycerides significantly decreased from 148.59 ± 32.77 mg/dl to 123.50 ± 33.26 mg/dl (n=137, P < 0.0001), reflecting an improvement in lipid metabolism. Improvements in lipid parameters, including reductions in total cholesterol, LDL-C, and triglycerides, are presented in (Figure 5).
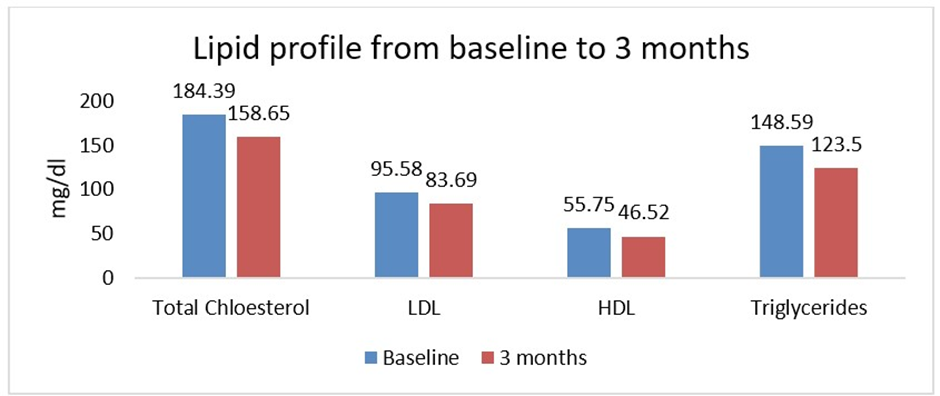
Figure 5: Lipid change from baseline to 3 months
Hepatic Parameters
Liver function markers, including ALT, AST, ALP, GGT, and Total bilirubin, showed minimal or no significant changes. Specifically, ALT (n=14) and AST (n=11) showed slight reductions, but these changes did not reach statistical significance (P > 0.05). Albumin levels slightly improved from 4.12 ± 0.89 g/dL to 4.43 ± 0.65 g/dL (n= 13, P = 0.2419), though this change was not statistically significant. Liver function tests showed no clinically significant changes (Figure 6).
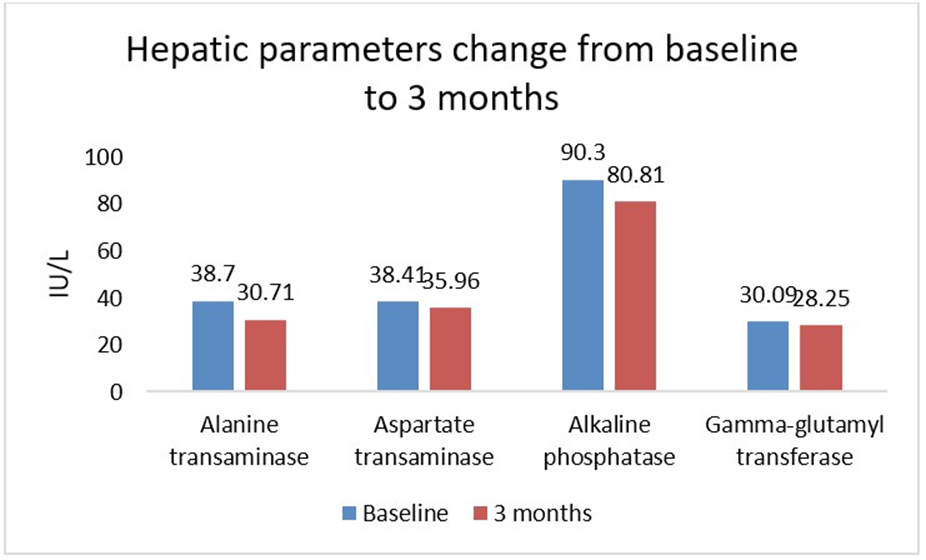
Figure 6: Hepatic enzymes change from baseline to 3 months
Adverse Events and Safety
Overall, 6.30% of patients experienced at least one AE, while the majority (93.70%) reported none. The most frequently reported AE was hypoglycaemia, occurring in 2.54% (n=35) of patients. Breathlessness was reported by 3.62% (n=50), and pedal oedema by 1.16% (n=16). Additionally, 0.87% (n = 12) of patients reported other symptoms, including weakness (0.14%), elevated blood pressure (0.14%), anxiety (0.07%), and stress (0.07%). Overall, the incidence of adverse events was low, with most patients not experiencing any significant side effects. Incidence of adverse events is shown in (Table 5).
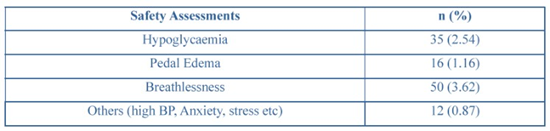
Table 5: Safety parameters in the study participants
Discussion
The present study evaluated the effectiveness and safety of the FDC of sitagliptin, metformin, and pioglitazone in patients with T2DM. The study showed notable improvements in various clinical parameters at the 3-month follow-up compared with baseline, highlighting the effectiveness of the FDC combination of sitagliptin, metformin, and pioglitazone in managing T2DM and associated health indicators.
In the present study, treatment with the FDC of sitagliptin, metformin and pioglitazone resulted in a significant improvement in glycemic control over 3 months (p<0.001). Patients demonstrated a marked reduction in FBG, PPG, and HbA1c levels, reflecting both short-term and long-term benefits. These findings are consistent with previous studies reporting a subsequent significant reduction in HbA1c with FDCs of metformin, sitagliptin, and pioglitazone (-1.64, P = 0.0208). Similar reductions were found in FPG (− 13.2 mg/dL, P = 0.0068) and PPG (− 20.83 mg/dL, P = 0.0023) [16].
Consistent with previous findings, the addition of alogliptin to metformin and pioglitazone significantly improved glycemic control in T2DM patients, achieving a mean HbA1c reduction of −1.4% (P < 0.001), suggesting triple therapy was more effective than either dual combination with metformin (P ≤ 0.001) [16]. Furthermore, Bosi et al. reported that triple therapy with a gliptin, metformin, and pioglitazone improved glycemic control, with an additional ~0.7% reduction in HbA1c over dual therapy after 52 weeks [17]. The observed reduction in HbA1c of approximately 0.8% is clinically meaningful, as even a 1% decrease in HbA1c has been associated with a significant reduction in microvascular complications of diabetes [18].
Despite the availability of multiple antidiabetic medications, managing weight gain continues to be a major challenge in diabetes care. Obesity, particularly visceral adiposity, plays a central role in the pathogenesis of T2DM, as excess free fatty acids released from adipose tissue impair insulin signalling pathways, thereby promoting insulin resistance. Pioglitazone is known to cause weight gain. However, several studies have shown that adding sitagliptin to patients already stabilized on pioglitazone does not result in any significant additional weight change compared with placebo [17,21]. Consistent with these reports, the present study does not find a significant change in body weight, suggesting that the addition of sitagliptin, metformin, and pioglitazone FDC did not contribute to excessive weight gain, a common concern with certain antidiabetic medications.
A slight but statistically significant reduction in eGFR (91.6 to 87.5, P = 0.0102) was observed in our study, along with improved serum creatinine levels (1.05 to 0.92, P < 0.0001) and a pronounced decrease in BUN (P < 0.0001). These findings are consistent with renal effects reported in recent studies evaluating the triple Combination of metformin, sitagliptin, and empagliflozin. The study found a modest decline in eGFR (101.8 to 98, p<0.05) over 24 months, suggesting preserved renal function. Serum creatinine remained stable, and urinary protein/albumin ratios improved, supporting the renal-protective profile of the triple therapy [20].
In the present study, a significant improvement in lipid parameters was observed, with reductions in total cholesterol (184.39 to 158.65, p < 0.0001), LDL (95.58 to 83.69, p< 0.0001), and triglycerides (148.59 to 123.50, p < 0.0001), indicating a favourable impact on lipid metabolism. In contrast, HDL (55.75 to 46.52, p = 0.0019) declined, warranting further evaluation. However, a study by Lin et al. demonstrated that triple therapy with metformin, a DPP4 inhibitor, and an SGLT2 inhibitor resulted in a more pronounced improvement in the lipid profile than metformin monotherapy. Specifically, this combination achieved a significant reduction in LDL-C (p = 0.016) and total cholesterol (p = 0.049), along with a favourable increase in HDL-C (p = 0.037), findings comparable to ours [21]. Furthermore, Park et al. reported that therapy with the triple combination of metformin, sitagliptin, and empagliflozin resulted in reductions in triglycerides and LDL-cholesterol, accompanied by an increase in HDL-cholesterol after 24 months [22]. These results are consistent with the improvements in lipid profile observed in our study.
In our study, liver function markers, including ALT, AST, ALP, GGT, and Total bilirubin, showed minimal or no significant changes. Specifically, ALT (38.71 to 30.71, p = 0.287) and AST (38.41 to 35.96, p = 0.752) showed slight reductions but did not reach statistical significance. Similarly, Park et al. reported that AST and ALT levels decreased among participants receiving triple therapy with metformin, sitagliptin, and empagliflozin, although the changes in ALT were not statistically significant [22].
In this study, FDC therapy with sitagliptin, metformin, and pioglitazone appeared to stabilize or modestly improve liver fibrosis over 3 months. The proportion of patients with mild fibrosis (F0–F1) increased slightly, while those with F2 and F3 fibrosis decreased, suggesting a potential reduction in fibrosis progression. Importantly, no significant worsening was observed, with only a minimal increase in patients reaching the F4 stage. These findings suggest a stabilizing or mildly beneficial effect of triple therapy on liver fibrosis. A study conducted by Lavynenko et al. suggested that advanced fibrosis (F3/F4) was significantly lower in the triple therapy of metformin/exenatide/pioglitazone (7%) compared with conventional therapy with metformin followed by sequential addition of glipizide and then basal insulin glargine (26%), suggesting a protective effect against fibrosis progression. These findings align with our study, which demonstrates that triple antidiabetic regimens and agents, such as SGLT2 inhibitors or TZDs, may confer both metabolic and hepatic benefits [22].
Each antidiabetic agent has unique pharmacological properties, and their concurrent use in combination therapy may enhance efficacy or even produce outcomes superior to those expected from individual agents [23,24]. A recent review of placebo-controlled, double-blind, randomized, parallel-group, multicentre trials demonstrated that pioglitazone, administered once daily for 16–24 weeks, was well tolerated both as monotherapy and in combination with other antihyperglycemic agents. Similarly, the present study found adverse events to be infrequent, with most patients reporting no significant side effects, indicating that the FDC of sitagliptin, metformin, and pioglitazone is generally well-tolerated and has a favourable safety profile [25]. These findings are consistent with the Indian IMPACT randomized trial of the metformin/sitagliptin/ pioglitazone FDC, which also reported low overall adverse event rates (12.7% with FDC vs. 8.5% with dual therapy), with most events being non-serious, only one severe adverse event in the FDC group, and no deaths. Taken together, this evidence indicates that triple FDC therapy has an acceptable short-term tolerability profile [18].
Limitations
This study has several limitations. First, being retrospective and observational, it is subject to selection bias and missing data. Second, the duration was limited to 3 months, which restricts the assessment of long-term efficacy and safety. Third, exploratory endpoints, such as HOMA-IR and Fibroscan scores, although outlined in the protocol, could not be evaluated due to insufficient data. Finally, the actual enrollment (1380 patients across 186 sites) was lower than the protocol-planned 1560 patients across 260 sites, which may affect generalizability.
Conclusion
This retrospective, multicenter study in 1,380 Indian patients with T2DM demonstrated that the FDC of Sitagliptin, Pioglitazone, and Metformin significantly improves glycemic control over a three-month period. This improvement is reflected in reductions in HbA1c, fasting plasma glucose, and postprandial glucose levels. Additionally, secondary benefits were observed in lipid profile and renal parameters, with stable liver function and minimal weight changes. The treatment demonstrated a good safety profile, with only a small proportion of patients reporting adverse events. The most commonly observed events were mild hypoglycemia and breathlessness. Importantly, no serious adverse events were documented.
These findings suggest that the FDC of Sitagliptin, Pioglitazone, and Metformin is an effective and well-tolerated therapeutic option for patients who are not achieving adequate glycemic control with monotherapy or dual therapy. This study provides valuable realworld evidence from the Indian population, supporting the role of triple FDC therapy in optimizing diabetes management and reducing cardiometabolic risk.
Ethics Approval Statement
The work presented in this study was in accordance with the study protocol, the New Drugs and Clinical Trials Rules 2019 issued by the Government of India, the ethical principles that have their origin in the Declaration of Helsinki, International Council for Harmonization (ICH) Good Clinical Practice (GCP), and all applicable local regulatory requirements. Approval from the Independent Ethics Committee was obtained prior to study initiation and data collection.
Acknowledgements
We would like to extend our thanks to all the institutes, investigators, and team members for their support. We also appreciate the CRO support from Auriga Research for clinical trial management. We also thank Medinsights Solution LLP. for medical writing support in the study.
Funding
This study was funded by Glenmark Pharmaceuticals Limited. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interest
Dr. Sumit Bhushan, Dr. Sanjay Chaudhari, Dr. Rahee Borulkar, Ms. Prajakta Bhosale, Ms. Rujuta Gadkari, and Dr. Saiprasad Patil are employees of Glenmark. All other investigators/authors have no conflicts of interest that are directly relevant to the content of this article.
References
- International Diabetes Federation (2025) IDF Diabetes Atlas. 11th ed. 2025.
- Lu X, Xie Q, Pan X, Zhang R, Zhang X, et al. (2024) Type 2 diabetes mellitus in adults: pathogenesis, prevention and therapy. Signal Transduct Target Ther, 9:262.
- Galicia-Garcia U, Benito-Vicente A, Jebari S, Larrea-Sebal A, Siddiqi H, et al. (2020) Pathophysiology of Type 2 Diabetes Mellitus. Int J Mol Sci, 21: 6275.
- Sen A, Kaur AP, Saxena I (2024) Metformin Monotherapy With and Without Lifestyle Changes Affects Anthropometric Parameters, Blood Pressure, Blood Glucose, and Lipid Profile in Indian Patients With Newly Diagnosed Type 2 Diabetes. Cureus,16: e62131.
- Goldberg RB (2022) Diabetic dyslipidemia: causes and consequences. J Clin Endocrinol Metab,107: e369–e383.
- American Diabetes Association (2025) 11. Cardiovascular Disease and Risk Management: Standards of Care in Diabetes—2025. Diabetes Care, 48: S207–S238.
- Brown JB, Conner C, Nichols GA (2010) Secondary failure of metformin monotherapy in clinical practice. Diabetes Care,33:501-506.
- RSSDI Clinical Practice Recommendations for the Management of Type 2 Diabetes Mellitus 2022. International Journal of Diabetes in Developing Countries, 42:1-143.
- Kim YG, Hahn S, Oh TJ, Kwak SH, Park KS, et al. (2013) Differences in the glucose-lowering efficacy of dipeptidyl peptidase-4 inhibitors between Asians and non-Asians: a systematic review and metaanalysis. Diabetologia, 56: 696-708.
- Rosenstock J, Perkovic V, Toto RD, et al. (2024) Efficacy and safety of sitagliptin in type 2 diabetes: a pooled analysis of recent trials. Diabetes Ther, 15: 455–468.
- Zhou Y, Huang Y, Ji L (2018) Pioglitazone for type 2 diabetes mellitus: a meta-analysis of randomized clinical trials. Clin Ther, 40:1623–1639.
- Yamauchi T, Kadowaki T (2023) Insights into the mechanism of action of thiazolidinediones from clinical and basic science perspectives. J Diabetes Investig, 14:6–15.
- Ceriello A, Johns D, Widel M, Eckland DJ, Gilmore KJ, et al. (2005) Comparison of effect of pioglitazone with metformin or sulfonylurea (monotherapy and combination therapy) on postload glycemia and composite insulin sensitivity index during an oral glucose tolerance test in patients with type 2 diabetes. Diabetes Care, 28: 266–272.
- Chawla S, Kaushik N, Singh NP, Ghosh RK, Saxena A (2013) Effect of addition of either sitagliptin or pioglitazone in patients with uncontrolled type 2 diabetes mellitus on metformin: A randomized controlled trial. J Pharmacol Pharmacother, 4: 27-32.
- Aashish M, Arindam N, Siddiqi SS, Bhosle D, Mallikarjuna VJ, et al. (2024) Efficacy and safety of fixed dose combination of Sitagliptin, metformin, and pioglitazone in type 2 Diabetes (IMPACT study): a randomized controlled trial. Clin Diabetes Endocrinol, 10: 3.
- DeFronzo RA, Burant CF, Fleck P, Wilson C, Mekki Q, et al. (2012) Efficacy and tolerability of the DPP-4 inhibitor alogliptin combined with pioglitazone, in metformin-treated patients with type 2 diabetes. J Clin Endocrinol Metab, 97:1615–1622.
- Bosi E, Ellis GC, Wilson CA, Fleck PR (2011) Alogliptin as a third oral antidiabetic drug in patients with type 2 diabetes and inadequate glycaemic control on metformin and pioglitazone: a 52-week, randomized, double blind, active-controlled, parallel-group study. Diabetes Obes Metab, 13: 1088–1096.
- Stratton IM, Adler AI, Neil HA, Matthews DR, Manley SE, et al. (2000) Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ, 321: 405-412.
- Bailey CJ, Green BD, Flatt PR (2010) Fixed-dose combination therapy for type 2 diabetes: sitagliptin plus pioglitazone. Expert Opin Investig Drugs, 19:1017–1025.
- Park YH, Sohn M, Lee SY, Lim S (2024) Two-year therapeutic efficacy and safety of initial triple combination of metformin, sitagliptin, and empagliflozin in drug-naïve type 2 diabetes mellitus patients. Diabetes Metab J, 48: 253-264.
- Lin YY, Weng SF, Hsu CH, Huang CL, Lin YP, et al. (2022) Effect of metformin monotherapy and dual or triple concomitant therapy with metformin on glycemic control and lipid profile management of patients with type 2 diabetes mellitus. Front Med (Lausanne), 9: 995944.
- Lavynenko O, Abdul-Ghani M, Alatrach M, Puckett C, Adams J, et al. (2022) Combination therapy with pioglitazone/exenatide/metformin reduces the prevalence of hepatic fibrosis and steatosis: The efficacy and durability of initial combination therapy for type 2 diabetes (EDICT). Diabetes Obes Metab, 24: 899-907.
- Arya DS, Chowdhury S, Chawla R, Das AK, Ganie MA, et al. (2019) Clinical benefits of fixed-dose combinations translated to improved patient compliance. J Assoc Physicians India, 67: 58–64.
- Gupta YK, Ramachandran SS (2016) Fixed-dose drug combinations: issues and challenges in India. Indian J Pharmacol, 48: 347–349.
- Hanefeld M, Belcher G (2001) Safety profile of pioglitazone. Int J Clin Pract Suppl, 121: 27–31.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.