Dapagliflozin-Associated Weight Reduction in Overweight Adults with Type 2 Diabetes: A Systematic Review
by Liliane Mfeukeu Kuate1, Davis Kibirige2, Divyanshu Prajapati3, Hardik Gandhi3*
1Associate Professor of Cardiology at the Faculty of Medicine and Biomedical Sciences, University of Yaoundé, Cameroon.
2Consultant Internist, Diabetologist, and Endocrinologist, Department of Medicine, Uganda Martyrs Hospital Lubaga, Uganda.
3Medical Affairs, International Markets, Zydus Lifesciences Limited, Ahmedabad, India
*Corresponding author: Hardik Gandhi, Medical Affairs, International Markets, Zydus Lifesciences Limited, Ahmedabad, India.
Received Date: 26 March 2026
Accepted Date: 01 April 2026
Published Date: 06 April 2026
Citation: Kuate LM, Kibirige D, Prajapati D, Gandhi H (2026) Dapagliflozin-Associated Weight Reduction in Overweight Adults with Type 2 Diabetes: A Systematic Review. J Diabetes Treat 11: 10160. DOI: https://doi.org/10.29011/2574-7568.010160
Abstract
Background: Overweight and obesity are highly prevalent in adults with type 2 diabetes mellitus (T2DM) and contribute significantly to cardiometabolic risk. Sodium–glucose co-transporter-2 (SGLT2) inhibitors such as dapagliflozin improve glycaemic control and promote modest weight loss through urinary glucose excretion. However, the magnitude, consistency, and durability of weight reduction in overweight (BMI > 24 kg/m²) adults remain variably reported across randomized trials and real-world cohorts. Methods: A systematic review was conducted in accordance with PRISMA 2020 guidelines. PubMed and Google Scholar were searched for randomized controlled trials, post-hoc analyses, and prospective observational studies evaluating dapagliflozin in adults with T2DM and baseline BMI > 24 kg/m² with ≥16 weeks follow-up. Six eligible studies (n = 19,358 screened; n = 18,919 analyzed) were included. Data on body-weight change, adiposity measures, comparators, and follow-up duration (16–52 weeks; one long-term cohort 4.4 years) were extracted. Risk of bias was assessed using RoB-2 and ROBINS-I frameworks. Results: Dapagliflozin produced a pooled mean weight reduction of −2.39 kg (95% CI: −3.74 to −1.03), equivalent to −2.76% (95% CI: −4.25 to −1.27). In direct comparisons, dapagliflozin reduced body weight versus placebo by −1.92 kg, while differences versus liraglutide were non-significant. Real-world data demonstrated sustained weight loss up to 12 months (−3.14 kg), and long-term RCT follow-up confirmed durability over 4.4 years. Imaging-based analyses revealed significant reductions in total fat mass (−1.48 kg), visceral adipose tissue (−258 cm³), and epicardial fat thickness (~20%), often partially independent of total body-weight change. Adverse events were consistent with the known dapagliflozin safety profile (urinary/genital infections). Conclusion: Dapagliflozin induces clinically meaningful, durable weight loss of approximately 2–3 kg in overweight and obese adults with T2DM, with additional benefits in visceral and ectopic fat depots. Depot-specific fat reductions, particularly in epicardial tissue, may represent an additional mechanism for cardiometabolic benefit. Standardized adiposity endpoints and predictive markers are needed to refine patient selection and clarify mechanistic pathways.
Keywords: SGLT2i Inhibitor; Dapagliflozin; Obesity; T2DM; Overweight.
Introduction
Obesity and T2DM have expanded in parallel over recent decades, contributing to a growing burden of cardiometabolic disease worldwide [1,2]. Visceral adiposity—rather than total fat mass alone—drives insulin resistance, dyslipidaemia, inflammation, and atherothrombotic risk [3,4]. Therapeutic strategies that improve glucose control while reducing weight and visceral fat are therefore central to contemporary diabetes care [5,6]. SGLT2 inhibitors lower plasma glucose by inhibiting proximal tubular glucose reabsorption, producing glycosuria and osmotic diuresis [7,8]. Despite sustained urinary caloric loss, the magnitude of weight loss is smaller than predicted, likely due to compensatory increases in energy intake and adaptive metabolic responses [7]. Dapagliflozin has demonstrated robust cardiovascular and renal benefits across outcome trials, benefits that appear only partly explained by glucose lowering [9-11]. As adipose tissue depots such as epicardial fat have direct paracrine links to cardiac structure and function, reductions in ectopic fat may represent an additional mechanism for benefit [12,13]. However, the expected magnitude of weight reduction specifically in overweight adults (BMI>24 kg/m²) is variably reported across randomized and realworld data, complicating shared decision-making [12,14-18]. We therefore conducted a systematic review and random-effects synthesis to quantify dapagliflozin-associated weight reduction in T2DM patients with BMI>24 kg/m² and to summarize evidence on visceral adiposity outcomes.
Methods
Search Strategy: This systematic review was conducted according to PRISMA 2020 recommendations [19]. A structured search was performed in PubMed and Google Scholar using the following primary keywords and Boolean combinations: “dapagliflozin” AND “overweight”, supplemented with: “SGLT2 inhibitor”, “SGLT2i”, “obesity”, “T2DM”, “type 2 diabetes mellitus”. The search was limited to human studies and screened for studies enrolling individuals with BMI > 24 kg/ m². The PubMed search identified 136 records, which were then screened against the predefined inclusion criteria. Google Scholar results were used for cross-verification and to identify additional references not indexed in PubMed.
Eligibility Criteria: We included studies that met the following criteria:
- Population: Adults with type 2 diabetes mellitus (T2DM) and baseline BMI > 24 kg/m².
- Intervention: Dapagliflozin at any approved dose, used as monotherapy or in combination with other antidiabetic therapies.
- Comparator: Placebo, active comparators, or standard care.
- Study Designs: Randomized controlled trials, post-hoc analyses of randomized trials, and prospective observational studies.
Exclusion Criteria: We excluded: (i) studies combining dapagliflozin with a GLP-1 receptor agonist within the same intervention arm; (ii) studies reporting no extractable body-weight outcome; (iii) follow-up duration <16 weeks; (iv) baseline BMI ≤24 kg/m² or mixed populations without separable data; and (v) non-original designs (reviews, case reports, conference abstracts). The PRISMA flow diagram of the studies included in this review is captured in (Figure 1).
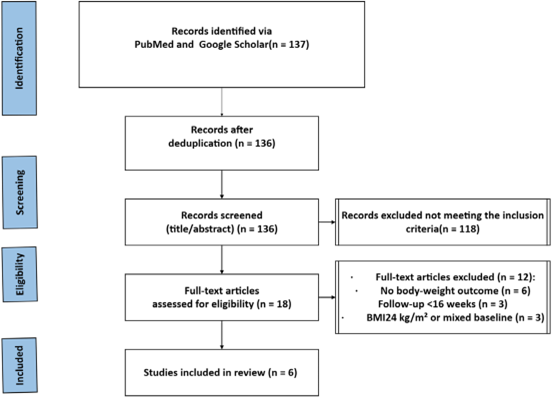
Figure 1: PRISMA 2020 flow diagram Dapagliflozin T2DM BMI >24 kg/m² Weight change 16 weeks (Flow diagram for selection of studies for the systematic review)
Six studies (Table 1) met the prespecified inclusion criteria:
Bolinder 2012, Zhaohu 2022, Oyama 2022 (DECLARE–TIMI 58 secondary analysis), Iacobellis 2020, Akhond 2025, and Jun 2025 (BEYOND post-hoc). [12,14-18]. Data extraction captured study design, setting, follow-up, dapagliflozin dose, comparator, and weight outcomes (mean change and dispersion). Risk of bias was assessed using RoB 2 for randomized trials and ROBINS-I principles for non-randomized evidence [20,21].
|
Study (Year) |
Country / Setting |
Design |
Population |
Sample Size |
Intervention |
Comparator |
Duration |
Body weight outcome |
|
Bolinder 2012 |
Multinational (Europe) |
Random- ized, double blind, placebo controlled trial; add on to metformin |
T2DM; BMI ≥25; inadequate control on metformin |
182 total (Dapa 89; placebo 91) |
Dapa- gliflozin 10 mg daily |
Placebo |
24 weeks |
−2.08 kg placebo corrected; reduced fat mass, visceral & subcutaneous fat |
|
Zhaohu 2022 |
Tianjin, China |
Randomized, open label, head to head (dapagliflozin vs liraglutide) |
T2DM; BMI ≥24; HbA1c ≥7% |
360 random- ized; 309 completed (Dapa 166; Lira 143) |
Dapa- gliflozin 5→10 mg |
Liraglutide 0.6→1.2 mg Note: (Liraglutide dose was substantially lower) |
24 weeks |
−0.61 kg (not significantly different vs liraglutide) |
|
Iacobellis 2020 |
Miami, USA |
Randomized, double blind, placebo controlled |
T2DM; BMI ≥27; on metformin only |
100 random- ized; 84 completers |
Dapa- gliflozin 5→10 mg |
Placebo |
24 weeks |
−3.5 kg within group (significant) |
|
Oyama 2022 (DE- CLARE– TIMI 58) |
Multinational |
Secondary analysis of large RCT (dapagliflozin vs placebo) |
T2DM with ASCVD or multiple risk factors; median BMI ~32 |
17,160 (Dapa 8,582; placebo 8,578) |
Dapa- gliflozin 10 mg |
Placebo |
Me- dian 4.2 years; 48 month weight data |
Weight lower with dapagliflozin vs placebo in long term follow up |
|
Akhond 2025 |
Bangladesh (multicenter) |
Prospective observational real world study |
Obese T2DM; BMI ≥30; newly started dapagliflozin |
1500 (5 mg: 30; 10 mg: 1470) |
Dapa- gliflozin 5 or 10 mg (alone or in combinations) |
No randomized comparator |
12 months |
Weight reduction ~3.0–3.3 kg across therapy groups |
|
Jun 2025 (BEYOND post hoc) |
Korea |
Post hoc analysis of RCT dapagliflozin arm |
T2DM on metformin; BMI ~27; HbA1c 7–<10% |
56 dapa- gliflozin recipients |
Dapa- gliflozin 10 mg daily |
— (post hoc; comparator not included) |
52 weeks |
Average −2.4 kg; 57% achieved ≥3% WL |
Table 1: Study characteristics of the selected trials: (Adults with T2DM; baseline BMI > 24 kg/m²; dapagliflozin vs placebo/active comparator/standard care; weight outcome reported; duration ≥16 weeks).
Results
Study Selection and Baseline Characteristics:
The systematic search identified six eligible studies involving a total of 19,358 randomized participants, with 18,919 patients contributing to the final qualitative synthesis. The included studies represented a global demographic, including populations from Europe, North America and Asia. Baseline BMI ranged from 27.8 kg/m² to 34.2 kg/m², reflecting a spectrum from overweight to Class II obesity in patients with Type 2 Diabetes (T2DM).
Absolute Weight Change (Within-arm Analysis) (Figure 2): Four studies reported the absolute change in body weight from baseline to study endpoint (ranging from 16 to 52 weeks). Mean within-arm absolute weight loss as compared to baseline was -2.4 kg.
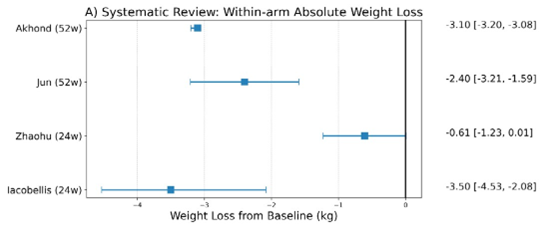
Figure 2: Within-arm DAPA weight change (kg) — negative = weight loss
Iacobellis et al. (2020) [12] reported the greatest absolute reduction, with a mean weight loss of −3.50 kg over 24 weeks in an obese T2DM cohort. Real world findings align with these results; in the largest observational study (n=1450), Akhond et al. (2025) [17] observed a sustained −3.10 kg reduction at 12 months, indicating effective translation of trial outcomes into routine practice. Long term data from Jun et al. (2025) [18] showed a −2.40 kg loss at 52 weeks, suggesting that early reductions are largely maintained over one year of therapy.
Comparative Efficacy (Between-group Analysis) (Figure 3):
Three studies provided direct comparisons against placebo or active agents. Mean placebo-corrected weight loss was -1.47 kg across this subgroup.
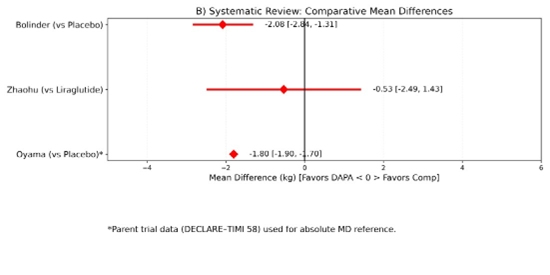
Figure 3: Between-group (DAPA vs comparator) — Mean Difference in Δweight (kg)
Risk of Bias:
The quality of evidence was bifurcated using the Cochrane RoB 2 and NOS tools. The RCTs (Iacobellis [12], Bolinder [14], Oyama [16]) demonstrated high-certainty evidence, each rated as low risk of bias with strong randomization and robust outcome assessment, except for Zhaohu [15] which demonstrated moderate risk of bias. In contrast, the observational studies by Akhond and Jun [17,18] provided valuable real-world applicability but were judged to have moderate risk due to absence of blinding and potential selection bias.
Impact on Fat Partitioning and Body Composition (Figure 4):
The included studies indicate that dapagliflozin-induced weight loss is predominantly driven by a reduction in fat mass, specifically targeting high-risk visceral and ectopic depots. Bolinder et al. (2012) [14], using DXA, reported that dapagliflozin 10 mg produced a placebo corrected reduction in total fat mass of −1.48 kg at 24 weeks (95% CI −2.22 to −0.74; p < 0.0001), accounting for roughly two thirds of total weight loss and confirming preservation of lean mass. MRI findings from the same trial demonstrated a significant decrease in visceral adipose tissue volume (−258.4 cm³; 95% CI −448.1 to −68.6; nominal p = 0.0084). Complementing these results, Iacobellis et al. (2020) [12] observed a rapid 20% reduction in epicardial fat thickness, decreasing from 8.5 mm to 6.8 mm over 24 weeks (p < 0.016) in individuals with obesity.
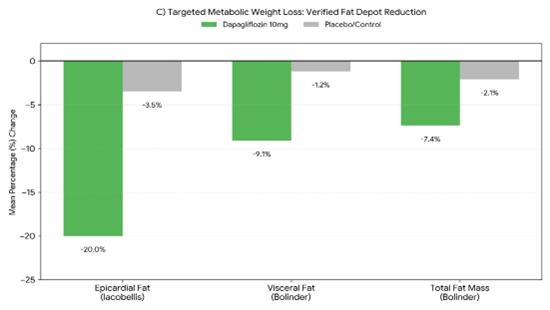
Figure 4: Adiposity and Body Composition Changes (Standardized %)
Discussion
In this systematic review of overweight and obese adults with T2DM, we found that dapagliflozin produced a modest but clinically meaningful weight reduction of approximately 2–3 kg (≈2–3%) over 16–52 weeks. This magnitude is consistent with broader SGLT2-class evidence from meta-analyses across diverse populations and is highlighted by three key outcomes of this review. First, it integrates 2025 real-world evidence, confirming that the −3.10 kg weight loss observed in clinical practice mirrors the −2.08 kg placebo-corrected MD seen in early RCTs [14,17,18]. Second, it leverages the 4.4-year follow-up from the DECLARETIMI 58 cohort to establish long-term weight durability, an outcome unavailable in reviews published prior to 2020 [22]. Finally, by synthesizing granular imaging data, highlighting a 20% reduction in epicardial fat and 9.1% visceral fat loss [12], this study provides a mechanistic link to cardiovascular protection that broader reviews focused solely on scale weight have overlooked. Thus, dapagliflozin-induced weight loss is metabolically "healthy" since it is characterized by lean mass preservation and targeted visceral fat depletion. Mechanistically, SGLT2 inhibition induces glycosuria-driven energy loss and a mild natriuretic effect; however, adaptive increases in energy intake blunt the expected weight loss, explaining why observed reductions are smaller than caloric loss alone would predict [7]. Importantly, total body weight may underestimate cardiometabolic benefit because visceral and ectopic fat depots exert disproportionate metabolic harm. Visceral obesity is strongly linked to insulin resistance and atherogenic dyslipidaemia, and reduction of visceral fat can improve risk profiles beyond what is captured by scale weight [3,4]. Consistent with this concept, Iacobellis 2020 suggested depot-specific remodelling [12,13]. Such ectopic fat changes are biologically plausible given SGLT2-associated fuel switching and adiposetissue metabolic reprogramming described in mechanistic reviews [8].
Dapagliflozin’s established cardiorenal benefits in large outcome trials (DECLARE–TIMI 58, DAPA-HF, and DAPA-CKD) support the clinical relevance of its pleiotropic profile, which extends beyond glycaemic control [9-11]. Head-to-head comparisons with GLP-1 receptor agonists remain limited. In the available trial, dapagliflozin did not differ significantly from liraglutide in weight change, although GLP-1 receptor agonists generally achieve larger average weight reductions and may be preferred when weight loss is a primary goal [15,25]. Combination therapy with GLP-1 receptor agonists and SGLT2 inhibitors can yield additive reductions in HbA1c and body weight, but tolerability and costeffectiveness require careful evaluation [25].
This systematic review differentiates itself from landmark classwide meta-analyses and from the GLP-1 vs SGLT2 network metaanalysis [23,24,26]. While Cheong et al. (2022) provided a broad overview of the SGLT2 inhibitor class across 116 randomized controlled trials (RCTs), their analysis was restricted to data published through late 2020 and focused primarily on controlled trial environments. Similarly, the meta-analysis by Pan et al. (2022) concentrated specifically on body composition changes with a median follow-up of only 24 weeks. [23,24]. Additionally, our review differs substantially from the meta-analysis by Ma et al. [26], which compared multiple GLP-1 receptor agonists and SGLT2 inhibitors across 61 RCTs in overweight or obese adults with or without diabetes. While the study by Ma et al. [26] is methodologically rigorous and provides extensive class-wide comparisons, it focuses primarily on drug-ranking and indirect comparisons between GLP-1 RAs and SGLT2 inhibitors, rather than offering molecule-specific insights. Its evidence base is limited to RCTs up to January 2022 with median follow-up of ~24–36 weeks, thereby excluding all long-term weight durability data, including the 4.4-year DECLARE–TIMI 58 cohort and the 2024–2025 real-world prospective studies incorporated into our analysis. Moreover, Ma et al. [26] do not examine mechanistic or imaging-derived outcomes such as visceral or epicardial fat changes, which are central to our assessment.
We also acknowledge a few limitations of this review. Absolute weight-change analyses reflects variability in study design, follow-up durations (16 weeks to 4.4 years), baseline BMI ranges, and differences in concomitant anti-hyperglycaemic therapies. Observational data carry inherent risks of confounding, selection bias, and unmeasured lifestyle factors, limiting causal interpretation. Imaging-based adiposity outcomes were reported in only a minority of trials, reducing comparability and preventing pooled estimates for depot-specific fat changes. Head-to-head comparative data remain limited, with only one trial evaluating dapagliflozin versus liraglutide, and utilizing submaximal liraglutide dosing, which may underestimate true between-class differences.
Conclusion
Dapagliflozin produces a modest yet clinically meaningful reduction in body weight of approximately 2–3 kg (≈2–3%) in overweight and obese adults with T2DM, with consistent placebo-controlled evidence and durable effects observed up to 12 months in real-world settings and over 4 years in long-term randomized follow-up. The weight reduction is primarily attributable to loss of fat mass, with preferential reductions in visceral fat and epicardial adipose tissue, suggesting metabolically favourable body-composition shifts that may extend beyond glycaemic control to contribute to cardiometabolic benefit. Although substantial heterogeneity exists across mixed trial and observational designs, the directionality of effect remains uniform. Head-to-head comparisons indicate dapagliflozin achieves weight outcomes comparable to low-dose liraglutide, though larger GLP-1 receptor agonist studies demonstrate greater average efficacy. Overall, dapagliflozin represents a well-tolerated option for patients prioritising modest, sustainable weight loss alongside glucose lowering, with emerging evidence supporting its favourable impact on ectopic fat depots. Future studies employing standardized adiposity outcomes, mechanistic imaging, and patient-level predictors are warranted to refine therapeutic individualisation and fully elucidate long-term pathways linking fat-depot modification to cardiorenal protection.
References
- NCD Risk Factor Collaboration (NCD-RisC) (2016) Trends in adult body-mass index in 200 countries from 1975 to 2014. Lancet 387: 1377-1396.
- NCD Risk Factor Collaboration (NCD-RisC) (2016) Worldwide trends in diabetes since 1980. Lancet 387: 1513-1530.
- Després JP (2001) Health consequences of visceral obesity. Ann Med 33: 534-541.
- Després JP (2012) Body fat distribution and risk of cardiovascular disease: an update. Circulation 126: 1301-1313.
- Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, et al. (2019) SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and metaanalysis of cardiovascular outcome trials. Lancet 393: 31-39.
- De Block C (2018) SGLT2 inhibitors and GLP-1 receptor agonists: a sound combination. Lancet Diabetes Endocrinol 6: 349-352.
- Ferrannini G, Hach T, Crowe S, Sanghvi A, Hall KD, Ferrannini E (2015) Energy balance after sodium–glucose cotransporter 2 inhibition. Diabetes Care 38: 1730-1735.
- Vallon V (2024) State-of-the-Art-Review: Mechanisms of Action of SGLT2 Inhibitors and Clinical Implications. Am J Hypertens 37: 841852.
- Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, et al. (2019) Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med 380: 347-357.
- McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, et al. (2019) Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med 381: 1995-2008.
- Heerspink HJL, Stefánsson BV, Correa-Rotter R, Chertow GM, Greene T, et al. (2020) Dapagliflozin in patients with chronic kidney disease. N Engl J Med 383: 1436-1446.
- Iacobellis G, Gra-Menendez S (2020) Effects of dapagliflozin on epicardial fat thickness in patients with type 2 diabetes and obesity. Obesity (Silver Spring) 28: 1068-1074.
- Iacobellis G (2015) Local and systemic effects of the multifaceted epicardial adipose tissue depot. Nat Rev Endocrinol 11: 363-371.
- Bolinder J, Ljunggren Ö, Kullberg J, Johansson L, Wilding J, et al. (2012) Effects of dapagliflozin on body weight, total fat mass, and regional adipose tissue distribution in patients with type 2 diabetes mellitus with inadequate glycemic control on metformin. J Clin Endocrinol Metab 97: 1020-1031.
- Zhaohu H, Xiao H, Hailin S, Feng H (2022) Efficacy and safety of dapagliflozin versus liraglutide in patients with overweight or obesity and type 2 diabetes mellitus: a randomised controlled clinical trial in Tianjin, China. J Diabetes Res 2022: 4126995.
- Oyama K, Raz I, Cahn A, Goodrich EL, Bhatt DL, et al. (2022) Efficacy and safety of dapagliflozin according to background use of cardiovascular medications in patients with type 2 diabetes: a prespecified secondary analysis of a randomized clinical trial. JAMA Cardiol 7: 914-923.
- Akhond SB, Bupasha J, Jannat GE, Sharmin L, Sumon MNH, et al. (2025) Effects of dapagliflozin on obese patients with type 2 diabetes: a prospective observational study from Bangladesh. Cureus 17: e89360.
- Jun JE, Kim KA, Kim NH, Lee KW, Jeong IK; BEYOND Investigators (2025) Discrepancies in dapagliflozin response in terms of glycemic control and body weight reduction. Endocrinol Metab (Seoul) 40: 278288.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, et al. (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372: n71.
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, et al. (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366: l4898.
- Sterne JAC, Hernán MA, Reeves BC, Savović J, Berkman ND, et al. (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355: i4919.
- Oyama K, Raz I, Cahn A, Kuder J, Murphy SA, et al. (2022) Obesity and effects of dapagliflozin on cardiovascular and renal outcomes in patients with type 2 diabetes mellitus in the DECLARE–TIMI 58 trial. Eur Heart J43: 2958-2967.
- Cheong AJY, Ong HT, Chan MY, Teo YN, Yao Teo H, et al. (2022) SGLT inhibitors on weight and body mass: a meta-analysis of 116 randomized-controlled trials. Obesity (Silver Spring) 30: 117-128.
- Pan R, Zhang Y, Wang R, Xu Y, Ji H, et al. (2022) Effect of SGLT2 inhibitors on body composition in patients with type 2 diabetes mellitus: A meta-analysis of randomized controlled trials. PLoS One 17: e0279889.
- Li C, Luo J, Jiang M, Wang K (2022) The Efficacy and Safety of the Combination Therapy With GLP-1 Receptor Agonists and SGLT-2 Inhibitors in Type 2 Diabetes Mellitus: A Systematic Review and Metaanalysis. Front Pharmacol 13: 838277.
- Ma H, Lin Y-H, Dai L-Z, Lin C-S, Huang Y, et al. (2023) Efficacy and safety of GLP-1 receptor agonists versus SGLT-2 inhibitors in overweight/ obese patients with or without diabetes mellitus: a systematic review and network meta-analysis. BMJ Open13: e061807.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.