Concurrent Chemoradiotherapy Combined with Immunotherapy for Rare Tracheal Tumors: Two Case Reports
by Hongxiang An1,2#, Jin Li3#, Lei Deng1*
1Department of Radiation Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing 100021, China
2The second radiotherapy ward of the Fourth Hospital of Fushun City, Liaoning Province, Fushun 113000, China
3Department of Pathology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing 100021, China
#Hongxiang An and Jin Li contributed equally to this work.
*Corresponding author: Lei Deng, Department of Radiation Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing 100021, China
Received Date: 06 November 2025
Accepted Date: 11 November 2025
Published Date: 13 November 2025
Citation: An H, Li J, Deng L. (2025). Concurrent Chemoradiotherapy Combined with Immunotherapy for Rare Tracheal Tumors: Two Case Reports. Ann Case Report. 10: 2445. https://doi.org/10.29011/2574-7754.102445
Background
Primary tracheal tumors are extremely rare, accounting for less than 0.1% of cases [1]. Most of these tumors are malignant, with squamous cell carcinoma (SCC) being the most common pathological type [2]. The current recommended treatment strategy for advanced tracheal tumors includes surgery and radiation therapy. Meanwhile, chemotherapy, laser treatment, cryotherapy, intraluminal therapy, and tracheal stent implantation are also potential options.
Due to the lack of guidance from relevant randomized clinical trials, the choice of treatment methods and the optimal treatment sequence remain undetermined. Research into new therapies for primary tracheal tumors is also limited due to the low incidence of this condition. However, treatment patterns from induction chemotherapy for locally advanced head and neck squamous cell carcinoma and the PACIFIC regimen for locally advanced non-small cell lung cancer could offer new strategies [3, 4]. Therefore, to further enhance our understanding of the treatment of advanced primary tracheal tumors, we report the application of concurrent chemoradiotherapy combined with immunotherapy in two rare cases of tracheal tumors.
Case 1: A 59-year-old male patient presented to an external hospital for "cough, hoarseness, and shortness of breath for 1 month." He has a 30-year history of smoking and no history of cardiothoracic surgery or tuberculosis. Cervicothoracic CT revealed a mass in the soft tissues adjacent to the trachea at the thoracic inlet, tracheal stenosis, and a mediastinal mass. Bronchoscopy showed new growth in the membranous part of the airway, spanning zones I-II (intraluminal, mural, and extramural components), with luminal stenosis of approximately 80%. Carbon dioxide extraction identified some newly detected organisms, and a pathological examination was conducted. After cauterizing the tumor with an argon beam, a rigid endoscope was used to place a stent in the airway spanning zones I-III. After treatment, the lumen was wider than before, with stenosis reduced to approximately 40%. Our hospital's pathological consultation confirmed high-grade squamous cell carcinoma, but the sample was insufficient for PD-L1 testing. Pulmonary infection occurred 3 days post-operation and improved with symptomatic treatment. A whole-body 18F-FDG PET-CT scan showed that the tracheal stent had changed, occupying space on the right side of the trachea and was considered malignant. Multiple lymph node metastases were noted in the 1L, 2R, 4R, and 4L areas of the mediastinum. The diagnosis was primary tracheal squamous cell carcinoma with mediastinal lymph node metastasis. The clinical stage was cT4N1M0, stage IV, as defined by the Bhattacharyya criteria [5].ECOG: 1 point. The patient received chest volume rotational intensity-modulated radiation therapy from October 2023 to December 2023, with the following prescription doses: 95% PGTV: 60.2 Gy/2.15 Gy/28 fractions, and 95% PTV: 50.4 Gy/1.8 Gy/28 fractions. Radiotherapy was paused for 3 weeks due to a lung infection during treatment, and the bronchial stent was removed during the pause. Synchronous chemotherapy was administered for two cycles during radiotherapy, consisting of albumin-bound paclitaxel plus carboplatin. During treatment, Grade I anemia and Grade II esophagitis occurred, which improved with symptomatic treatment. Concurrent chemoradiotherapy was followed by durvalumab immunotherapy maintenance. Follow-up until June 2025 showed no tumor progression for 18 months (Figure 1).
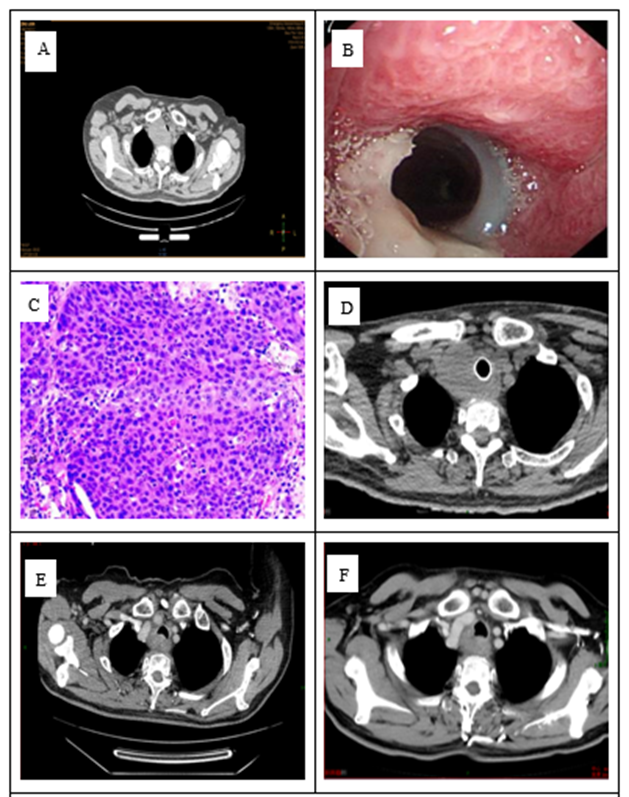
Figure 1: A: CT at diagnosis; B: Endoscopic stent placement; C: Pathological HE × 200; D: Post-stent placement CT; E: CT one month after radiotherapy completion; F: CT four months after radiotherapy completion.
Case 2: A 67-year-old male patient presented with a "tracheal mass found on physical examination one month ago." He had a history of heavy smoking and drinking, with no history of cardiothoracic surgery or tuberculosis. Chest CT revealed an irregular mass behind the tracheal wall at the thoracic inlet, with a maximum cross-section of approximately 4.2 cm by 3.5 cm. The corresponding lumen was narrowed, with the lesion mostly protruding outward. The boundary with the adjacent esophagus and thyroid was unclear, and it was closely adjacent to the thoracic 1 vertebral body. The esophagus was compressed and narrowed. Esophagoscopy showed that the esophageal inlet, located about 16-18 cm from the incisors, exhibited an extrinsic compression bulge, causing narrowing at the esophageal entrance. Bronchoscopy revealed a local protuberance in the trachea, about 2 cm past the glottis, approximately 2 cm by 3 cm in size. The mass showed infiltrative growth with no distinct boundary and was locally accompanied by ulceration and erosion on the surface. Pathological examination confirmed poorly differentiated squamous cell carcinoma. Immunohistochemistry results were as follows: PD-L1 (22C3) (TPS = 50%; CPS = 50). The diagnosis was primary tracheal squamous cell carcinoma with mediastinal lymph node metastasis. The clinical stage was cT4N1M0, stage IV, as defined by the Bhattacharyya criteria. ECOG: 1 point. After surgical consultation, the tumor was deemed unresectable. In April-May 2024, the patient underwent two cycles of induction therapy with "Pembrolizumab + Liposomal Paclitaxel + Carboplatin," resulting in a reduction of the lesion. Chest volume rotational intensity-modulated radiation therapy was administered from June to July 2024, with the following prescription doses: 95% PGTV: 60.2 Gy/2.15 Gy/28 fractions, and 95% PTV: 50.4 Gy/1.8 Gy/28 fractions. Concurrent chemotherapy, consisting of Liposomal Paclitaxel + Carboplatin, was given during radiotherapy for two cycles. During treatment, Grade I anemia and Grade II radiation esophagitis occurred, which improved with symptomatic treatment. The patient is currently undergoing maintenance treatment with pembrolizumab. Follow-up until June 2025 showed no tumor progression for one year (Figure 2).
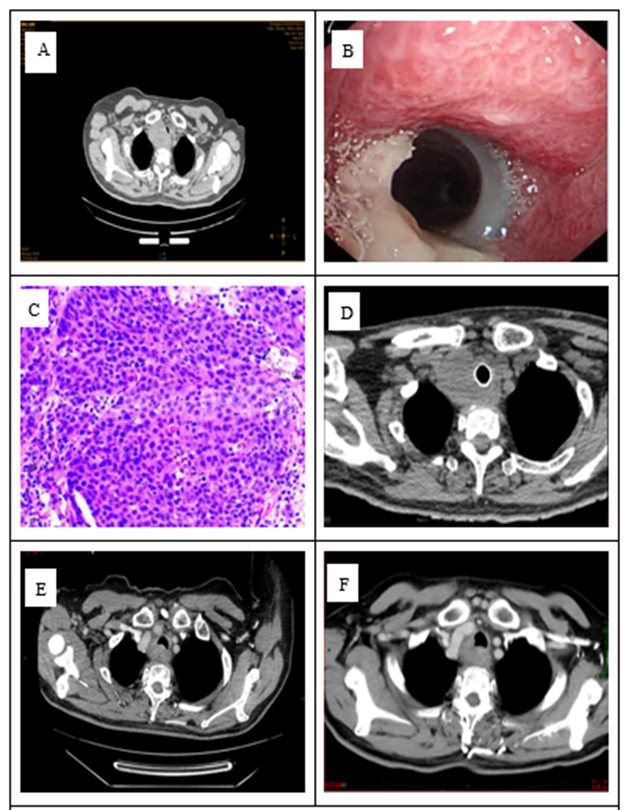
Figure 2: A: CT at diagnosis; B: Bronchoscopy findings; C: Esophagoscopy findings; D: Pathology HE × 200; E: CT after two cycles of induction; F: CT three months after radiotherapy completion.
Discussion
Due to the rarity of primary tracheal squamous cell carcinoma, the optimal treatment method remains undetermined. The treatment experience from these two cases offers a new therapeutic strategy. In both cases, combining concurrent chemoradiotherapy and immunotherapy improved disease control, increased patient treatment tolerance, and reduced the incidence of treatment-related adverse reactions. Although we provide new insights into the treatment of primary tracheal squamous cell carcinoma, further data and clinical research are needed for validation. The two cases also prompt us to consider several important questions: What is the optimal treatment approach for newly diagnosed advanced inoperable squamous cell carcinoma of the trachea prior to concurrent chemoradiotherapy? In the context of these two cases, we tend to favor the non-invasive induction therapy used in Case 2. Case 1, treated with laser and stent placement, resulted in complications that affected the continuity of treatment. In contrast, Case 2 showed good tolerance to induction chemotherapy combined with immunotherapy, followed by concurrent chemoradiotherapy. Radical radiotherapy is recommended for locally advanced inoperable squamous cell carcinoma of the trachea. Nonetheless, the optimal timing for chemotherapy, immunotherapy, and radiation therapy interventions, as well as the best combination treatment strategies and the potential benefits in terms of local control and survival, still require further research for validation.
Conclusion
In summary, this study preliminarily demonstrates the potential clinical value of concurrent chemoradiotherapy combined with immunotherapy in locally advanced primary tracheal squamous cell carcinoma. Individualized selection of systemic induction therapy or local therapy before concurrent chemoradiotherapy, based on patient characteristics, could improve local disease control, increasing treatment tolerance and enhancing survival outcomes. We hope that our study will provide valuable insights into selecting a multidisciplinary treatment strategy for locally advanced primary tracheal squamous cell carcinoma and serve as a reference for improving patient treatment outcomes.
Declaration of funding
This paper was not funded.
Declaration of financial/other relationships
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties. Peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
Author contributions
H.X.A. and L.D. contributed to the conception and design of the study. All authors collected the clinical data and interpreted data for the work. H.X.A. and J.L. contributed to the analysis and interpretation of data. H.X.A. and J.L. wrote the manuscript. All authors had access to the data, contributed to the writing of this manuscript, and gave final approval of the version to be published.
Ethics statement
For this case report, ethical committee approval was not required since only de-identified compliant data were used in the analysis. The study adhered to data protection laws and the principles of the Helsinki Declaration. Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
References
- Macchiarini P. (2006). Primary tracheal tumours. The Lancet Oncology. 7: 83-91.
- Piórek A, Płużański A, Teterycz P, Tabor S, Winiarczyk K, et al. (2024). Clinicopathological characteristics of patients with primary tracheal tumors: Analysis of eighty‐nine cases. Thoracic Cancer. 15: 878-883.
- Pfister DG, Spencer S, Adelstein D, Adkins D, Anzai Y, et al. (2020). Head and Neck Cancers, Version 2.2020, NCCN Clinical Practice Guidelines in Oncology. Journal of the National Comprehensive Cancer Network. 18: 873-898.
- Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, et al. (2017). Durvalumab after Chemoradiotherapy in Stage III Non–Small-Cell Lung Cancer. New England Journal of Medicine. 377: 1919-1929.
- Bhattacharyya N. (2016). Contemporary Staging and Prognosis for Primary Tracheal Malignancies: A Population‐Based Analysis. Otolaryngology–Head and Neck Surgery. 131: 639-642.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.