Clinical and Electrophysiogical Profile of Gullian Barre Syndrome in Children
by Ashutosh Kolewar1, Ghanshyam Das2*, Ajay Gaur3
1Pg student, Department of paediatrics Gajra raja medical college, Gwalior, India
2professor paediatrics, Department of paediatrics Gajra raja medical college, Gwalior, India
3professor and Head paediatrics, Department of paediatrics Gajra raja medical college, Gwalior, India
*Corresponding author: Ghanshyam Das, Department of paediatrics, Gajra raja medical college, Gwalior, India
Received Date: 18 March 2026
Accepted Date: 04 April 2026
Published Date: 07 April 2026
Citation: Kolewar A, Das G, Gaur A (2026) Clinical and Electrophysiogical Profile of Gullian Barre Syndrome in Children. Arch Pediatr 11: 341. DOI: https://doi.org/10.29011/2575-825X.100341
Abstract
Background: Guillain-Barre Syndrome (GBS) is an immune-mediated neuropathy that leads to motor weakness and may result in paralysis. This study aims to evaluate the clinical presentations, electrophysiological subtypes in GBS patients. Methods: This prospective observational study was done in the Paediatric Intensive Care Unit. Children were diagnosed with GBS based on standard diagnostic criteria, and data were collected and statistical analysis was done. Results: Out of 52 children of GBS, with a female predominance n=27 (51.9%), Ascending paralysis was common. Electrophysiological studies classified as acute motor axonal neuropathy (AMAN) n=20 (38.5%), acute inflammatory demyelinating polyneuropathy (AIDP) n=15 (28.9%). Ventilatory support was required in n=4 (7.7%) patients. Full recovery was achieved n=30(57.6%) at discharge, at eight-week follow-up full recovery was n=41(78.9%) , with mild to moderate residual disabilities in 11(21.1%) children. Conclusions: AMAN as the most common subtype in Paediatric GBS. Residual disabilities was in about 21.1% participants.
Keywords: Guillain-Barre Syndrome; Neuropathy; AMAN; Electrophysiology
Introduction
Guillain-Barre Syndrome (GBS) is a post infectious, monophasic, immune‐mediated polyradiculoneuropathy. About a third of patients with GBS develop generalised neuropathy, and require mechanical ventilation for respiratory failure [1]. GBS affects adults and children, although it is less common in paediatric population, where incidence estimates range from 0.4 to 1.4 cases per 100,000 children [2]. This condition presents after an antecedent viral or bacterial infection, with half of paediatric cases reporting a recent respiratory or gastrointestinal illness, underscoring the autoimmune pathogenesis of GBS [3]. While some patients experience a monophasic course, others may have relapses or secondary complications due to autonomic dysfunction or respiratory muscle weakness, necessitating intensive care [3].
Since the introduction of therapies such as plasmapheresis and intravenous immunoglobulin (IVIG) in the 1980s, clinical outcomes for GBS have improved, reducing the need for ventilatory support and the rate of mortality [4]. These treatments are particularly beneficial in paediatric cases, as children tend to show a faster recovery rate and better overall prognosis than adults [5]. However, despite therapeutic advancements, the evidence base on optimal management and long-term outcomes in GBS is limited, particularly in developing countries [6].
Clinical presentations of GBS vary such as acute inflammatory demyelinating polyneuropathy (AIDP) and acute motor axonal neuropathy (AMAN) displaying distinct neurophysiology [4,7]. AIDP, the commonest subtype, involves demyelination, while AMAN primarily affects axons and is prevalent in Asia [2,8]. Electrophysiological studies, are critical in differentiating subtypes and can guide prognosis and management [4,7]. There is a scarcity of studies focusing on paediatric GBS in India. This study aims to gain insight into the clinical characteristics, diagnostic challenges, and treatment approaches for paediatric GBS.
Material and Methods
This study was a prospective observational study. Participants were selected for the study using a non-probability convenience sampling method. Participants admitted in Paediatric intensive care unit (PICU) Kamla Raja Hospital, Gwalior with sign and symptom of GBS who met National institute of neurological and stroke Criteria (NINDS) [4]. were included in this study after taking informed consent from their parents.
Detailed demographic profile, history and clinical examination were done in all study participants. Electrophysiological studies were done by using Neuro MEP 4 Neurosoft system after stabilization of the participants in the Neurology department of Gajra Medical College and Hospital and was reported by an expert. Management strategies were documented. Disability grading was done by using Hughes disability score at the time of admission, discharge and at 8 weeks follow-up. Outcome was measured at the time of discharge and after 8 weeks.
♦ Inclusion Criteria
i. Children admitted in PICU with 1 to 15yrs of age, satisfying NINDS criteria for GBS[4].
NINDS diagnostic criteria
- Progressive symmetrical weakness of more than one limb.
- Progression of weakness for 2-4 weeks.
- Areflexia or hyporeflexia.
- Autonomic dysfunction.
- Mild sensory symptoms and sign.
- Absence of fever.
- Cranial nerve involvement.
♦ Exclusion Criteria
- Children with other causes of neuropathy, such as porphyria, botulism, post- diphtheritic neuropathy, drug or toxin induced neuropathy, or poliomyelitis, trauma.
- Children diagnosed with neuromuscular junction disorders or myasthenia gravis.
Study Flow Chart
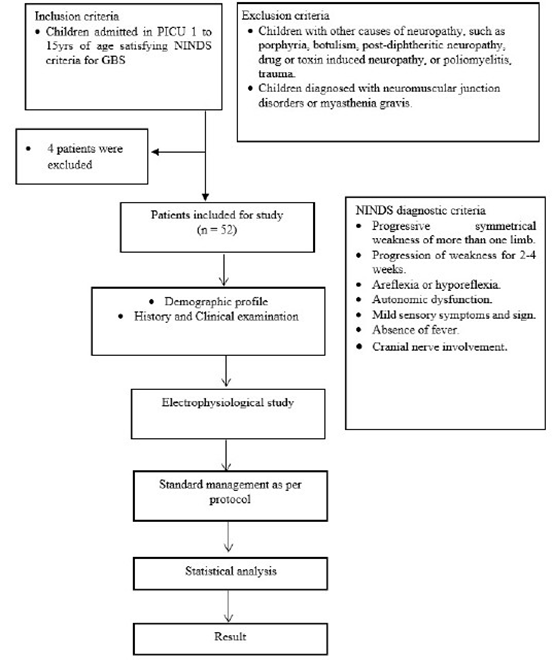
Statistical and graphical analyses for this study were undertaken by stata software version 17.0. Descriptive statistics were used to summarize demographic variables and clinical features, while inferential tests, such as chi-square tests and t-tests, were applied to assess relationships between independent and dependent variables. Multivariate analyses were performed to control for potential confounders, providing adjusted estimates for prognostic indicators in gbs patients.
Results
A total of 52 children from 1year up to 15 years, diagnosed with Guillain-Barré Syndrome based on clinical and electrophysiological criteria, were enrolled after fulfilling the inclusion and exclusion criteria.Relevant demographic data, clinical presentations, disability scores, electrophysiological findings, need for ventilatory support, and short-term recovery outcomes were recorded.
(Table 1) illustrates that the majority of participants were in the 6–10 years age group (42.31%), followed by 1–5 years (36.54%) and 11–15 years (21.15%). There was a slight female predominance, with 51.92% girls and 48.08% boys. Most children (61.5%) belonged to rural areas, while 38.5% were from urban settings. Seasonal distribution showed the highest incidence during the rainy season (32.69%), followed by autumn (26.92%), summer (23.07%), and winter (17.30%).
|
Variables |
Number (n) |
Percentage (%) |
|
Age |
||
|
1-5 Years |
19 |
36.54 |
|
6-10 Years |
22 |
42.31 |
|
11-15 Years |
11 |
21.15 |
|
Gender |
||
|
Boy |
25 |
48.08 |
|
Girl |
27 |
51.92 |
|
Residence |
||
|
Rural |
32 |
61.5 |
|
Urban |
20 |
38.5 |
|
Seasons |
||
|
Rainy |
17 |
32.69 |
|
Autumn |
14 |
26.92 |
|
Summer |
12 |
23.07 |
|
Winter |
9 |
17.30 |
|
Antecedent illness |
||
|
History of Diarrhea Before Onset |
15 |
28.85 |
|
History of Upper Respiratory Infection Before Onset |
18 |
34.62 |
|
Recent Immunization |
09 |
17.30 |
|
Types of paralysis |
||
|
Ascending Paralysis |
23 |
44.23 |
|
Descending Paralysis |
10 |
19.23 |
|
Salient clinical features |
||
|
Paresthesia, sensory involvement and pain |
||
|
Limited to fingers or toes |
(12, 17, 2) |
(23.08, 32.69, 3.85) |
|
Extended to ankle or wrist |
(14,14,18) |
(26.92, 26.92, 34.62) |
|
Extended to knee or elbow |
(14, 16, 18) |
(26.92, 30.77, 34.62) |
|
Above knees or elbows |
(12, 5, 11) |
(23.08, 9.62, 21.15) |
|
Facial Involvement |
(0, 0,3) |
(0, 0, 5.77) |
|
Motor symptoms |
||
|
Difficulty in Hand grip |
11 |
21.15 |
|
Difficulty in combing, reaching up to shelf |
19 |
36.54 |
|
Difficulty in turning in bed |
11 |
21.15 |
|
Bulbar symptoms |
7 |
13.46 |
|
Difficulty in gripping foot wear, footwear slipping off |
4 |
7.69 |
|
Cranial nerve involvement |
||
|
Facial |
4 |
7.69 |
|
Bulbar |
6 |
11.53 |
|
Requirement for Mechanical ventilation |
4 |
7.69 |
Table 1: Baseline demographic characterstics of study participants.
Antecedent illness was notable in several cases, with a history of upper respiratory tract infection reported in 34.62% and diarrhea in 28.85% of participants. Recent immunization was documented in 17.30% of cases. Ascending paralysis emerged as the most common form 44.23%, while descending paralysis was observed in 19.23%. Pain, paresthesia, sensory involvment varied in extent, with pain (26.92%) paresthesia (30.77%) sensory (36.62%) among participants had extending above the knees or elbows.
Motor symptoms were frequently reported, with difficulty in combing or reaching overhead being the most prevalent (36.54%), followed by difficulty in hand grip and turning in bed (21.15% each). Bulbar symptoms were present in 13.46%, and foot weakness, indicated by slipping off of footwear which was noted in 7.69%. Cranial nerve involvement was observed in both facial (7.69%) and bulbar nerves (11.53%). Notably, 7.69% required mechanical ventilation during their hospital stay, while the majority (92.31%) did not need any ventilatory assistance. This suggests in some cases of paediatric GBS can be severe, most children can be managed without the need for respiratory support when diagnosed and treated promptly.
(Table 2) illustrates that Acute motor axonal neuropathy (AMAN) was the most common subtype, accounting for 38.46% of cases, acute inflammatory demyelinating polyneuropathy (AIDP) at 28.85%, and acute motor-sensory axonal neuropathy (AMSAN) at 23.08%, 9.62%, fell into other less common subtypes.
|
Electrophysiological Findings |
Number (n) |
Percentage (%) |
|
AMAN |
20 |
38.46 |
|
AIDP |
15 |
28.85 |
|
AMSAN |
12 |
23.08 |
|
OTHERS |
5 |
9.62 |
|
Total |
52 |
100.00 |
Table 2: Electrophysiological Findings.
(Table 3) Highlights the disability scores at the time of admission, at discharge and after 8 weeks followup. Twenty two children (42.30%) had grading score of 1, four children (7.69%) had grading score of 5 at the time of admission. Thirty children (57.69%) had grading score of 0, four children (7.69%) children had grading score of 4 at the time of discharge. Forty-one children (78.84%) had grading score of 0, one child had grading score of 3 at the time of follow-up after 8 weeks.
|
Score grading |
Disability score |
On admission |
At Discharge |
After 8 weeks follow-up |
|||
|
n |
% |
n |
% |
n |
% |
||
|
0 |
A healthy state |
0 |
0 |
30 |
57.69 |
41 |
78.84 |
|
1 |
Minor symptoms & capable of running |
22 |
42.30 |
8 |
15.38 |
7 |
13.46 |
|
2 |
Able to walk 10 meter or more without assistance but unable to run |
12 |
23.07 |
6 |
11.53 |
3 |
5.76 |
|
3 |
Able to walk 10 meter across open space with help |
8 |
15.38 |
4 |
7.69 |
1 |
1.92 |
|
4 |
Bedridden or chairbound |
6 |
11.53 |
4 |
7.69 |
0 |
0 |
|
5 |
Requiring assisted ventilation for at least part of the day |
4 |
7.69 |
0 |
0 |
0 |
0 |
|
6 |
Died |
0 |
0 |
0 |
0 |
0 |
0 |
|
Total |
52 |
100 |
52 |
100 |
52 |
100 |
|
Table 3: Distribution of participants according to Hughes Disability Score.
(Table 4) Depicts the recovery status of paediatric gbs patients, which was assessed at two time points: At discharge and after 8 weeks of follow-up. At the time of discharge, 57.69% of patients had achieved full recovery, while 26.92% showed mild residual disability and 15.38% had moderate residual disability. Importantly, no patients were discharged with severe disability. At the 8-week followup, outcomes had notably improved. The proportion of patients achieving full recovery increased to 78.85%, indicating significant functional recovery post-discharge. the number of children with mild residual disability decreased to 13.46%, and those with moderate disability further declined to 7.69%. No cases of severe disability were observed at follow-up.
|
Recovery at discharge |
Recovery after 8 weeks follow-up |
|||
|
Number |
Percentage |
Number |
Percentage |
|
|
Fully Recovery |
30 |
57.69 |
41 |
78.85 |
|
Mild Residual Disability |
14 |
26.92 |
7 |
13.46 |
|
Moderate Residual Disability |
8 |
15.38 |
4 |
7.69 |
|
Severe Disability |
0 |
0.00 |
0 |
0 |
Table 4: Distribution of study subjects according to recovery at discharge.
Discussion
The findings from the current prospective observational study on paediatric Guillain-Barré Syndrome (GBS), encompassing 52 children aged 1 to 15 years, align well with existing literature on childhood GBS, while also highlighting region-specific patterns that are essential to understanding the disease spectrum in India. The study revealed a slightly higher proportion of female patients (51.9%), contrasting with most other studies, such as which reported a male predominance (male:female ratios of approximately 2.1:1 and 69% boys, respectively) [16,18]. This slight female predominance in the current study might be attributable to local epidemiological factors or sample variation.
Age distribution in this cohort demonstrated the highest prevalence in the 6–10-year age group (42.3%), with a notable representation from 1–5 years (36.5%). This finding is consistent with reports from, all of which observed that GBS most commonly affects children between 5 and 10 years of age [16,15,14], reflecting the increased susceptibility of school-aged children to immunological triggers.
Antecedent illnesses were documented in a significant number of cases, with upper respiratory tract infections in 34.6% and diarrheal episodes in 28.9% of patients. These findings align with (69.8% with antecedent illness, especially diarrhea in AMAN cases) [16], and where diarrheal illness was significantly associated with AMAN subtype [17]. This consistency across studies underscores the role of antecedent infections—particularly gastrointestinal infections likely linked to Campylobacter jejuni in the pathogenesis of axonal forms of GBS, a phenomenon welldocumented in Asian populations.
Electrophysiological classification in the present study revealed AMAN as the predominant subtype (38.5%), followed by AIDP (28.9%) and AMSAN (23.1%). This subtype distribution is highly consistent with other studies conducted in India and Asia, including, which reported AMAN in 41.5% and AIDP in 23% of cases [18], who found AMAN in 69.4% and AIDP in 25% [10], who reported AMAN in 44.2% and AIDP in 48.8% [16]. Conversely, studies from Europe and North America, including those referenced by, continue to report AIDP as the predominant subtype (often 70– 85%) [15]. These geographic differences are thought to arise from variations in environmental exposures, genetic susceptibility, and differing frequencies of infectious agents that may trigger specific immunological responses leading to GBS.
Clinically, the current study highlighted ascending paralysis as the most common presentation (44.2%), with sensory symptoms extending above the knees or elbows noted in a substantial subset (26–30%). This correlates with prospective study, where sensory symptoms, including pain, were frequently reported, though they observed an even higher rate of neuropathic pain (79%) [15]. Cranial nerve involvement was noted in 7.7% (facial) and 11.5% (bulbar) of cases, which is slightly lower than the rates reported by [15] (46% cranial nerve involvement) [16] (76.7% cranial nerve palsies). However, it remains within the spectrum reported across studies, indicating that cranial nerve involvement, while variable, is a recognized feature in paediatric GBS.
Mechanical ventilation was required in 7.7% of patients in the current study, aligning with [13] (8.3%) and slightly lower than [10] (16.6%). This highlights that, although a minority of children progress to respiratory compromise, most cases in this cohort had a relatively benign course—a trend mirrored in other regional studies, including and, both of which noted that despite AMAN’s association with more severe electrophysiological findings [18,11], the overall clinical prognosis remained favorable.
Short-term outcomes in the present study were notably positive, with 78.8% of patients achieving a complete recovery (disability score 0) at 8 weeks, and only 1.9% showing moderate disability (score 3). These results resonate with finding of 75% full recovery at approximately 9 months [15], and 97% ambulatory status at 3 months [10], reinforcing the well-recognized notion that paediatric GBS generally has a better prognosis than its adult counterpart. However, highlighted that some AMAN patients may experience delayed recovery despite eventual good long-term outcomes [11], suggesting that electrophysiological subtypes alone may not fully predict prognosis.
Conclusion
The current study’s findings not only confirm the predominance of the AMAN subtype and its association with antecedent infections but also emphasize the relatively mild clinical course and good short-term prognosis in most children. The slight variation in female predominance and less cranial nerve involvement underscore the importance of local epidemiological and demographic factors. Collectively, these findings reinforce early electrophysiological studies, prompt diagnosis, and appropriate supportive care to ensure favourable outcome in paediatric GBS.
Conflicts of interest
None
Author contributions
Dr Ashutosh Kolewar - 1) acquisition and analysis of data 2) drafting a significant portion of the manuscript or figures
Dr Ghanshyam Das conception and design of the study,
Dr Ajay Gaur - supervision
References
- Bellanti R, Rinaldi S (2024) Guillain-Barré Syndrome: A Comprehensive Review. Eur J Neurol 31: E16365.
- Khan Sa, Das Pr, Nahar Z, Dewan Smr (2024) An Updated Review on Guillain-Barré Syndrome: Challenges In Infection Prevention and Control In Low- and Middle-Income Countries. Sage Open Med 12: 20503121241239536.
- Dimachkie Mm, Barohn Rj (2013) Guillain-Barré Syndrome and Variants. Neurol Clin 31: 491-510.
- Leonhard Se, Mandarakas Mr, Gondim Faa, Bateman K, Ferreira Mlb, et al, (2019) Diagnosis and Management of GuillainBarré Syndrome In Ten Steps. Nat Rev Neurol 15: 671-683.
- Van Den Berg B, Walgaard C, Drenthen J, Fokke C, Jacobs Bc, et al, (2014) GuillainBarré Syndrome: Pathogenesis, Diagnosis, Treatment And Prognosis. Nat Rev Neurol 10: 469-482.
- Florian Ia, Lupan I, Sur L, Samasca G, Timiș Tl (2021) To Be, Or Not To Be… Guillain-Barré Syndrome. Autoimmun Rev 20: 102983.
- Sudulagunta Sr, Sodalagunta Mb, Sepehrar M, Khorram H, Bangalore Raja Sk, et al, (2015) Guillain-Barré Syndrome: Clinical Profile and Management. Ger Med Sci 13: Doc16.
- Willison Hj, Jacobs Bc, Van Doorn Pa (2016) Guillain-Barre Syndrome. Lancet 388: 717-727.
- Ammache Z, Afifi Ak, Brown Ck, Kimura J (2001) Childhood GuillainBarré Syndrome: Clinical and Electrophysiologic Features Predictive of Outcome. J Child Neurol 16: 477-483.
- Yadav S, Jain P, Sharma S, Kumar V, Aneja S (2019) GuillainBarre Syndrome in North Indian Children: Clinical and Serial Electrophysiological Features. Neurol India 67: 724-727.
- Nagasawa K, Kuwabara S, Misawa S, Fujii K, Tanabe Y, et al. (2006) Electrophysiological Subtypes and Prognosis of Childhood GuillainBarré Syndrome in Japan. Muscle and Nerve 33: 766-770.
- Ashrafi Mr, Mohammadalipoor A, Naeini Ar, Amanat M, Tavasoli Ar, et al. (2020) Clinical Characteristics and Electrodiagnostic Features of Guillain-Barré Syndrome Among The Pediatric Population. J Child Neurol 35: 448-455.
- Akbayram S, Dogan M, Akgün C, Peker E, Sayin R, et al. (2011) Clinical Features and Prognosis with Guillain-Barr Syndrome. Ann Indian Acad Neurol 14: 98-102.
- Karalok Zs, Taskin Bd, Yanginlar Zb, Gurkas E, Guven A, et al. (2018) Guillain-Barré Syndrome in Children: Subtypes and Outcome. Childs Nerv Syst 34: 22912297.
- Korinthenberg R, Schessl J, Kirschner J (2007) Clinical Presentation and Course of Childhood Cuillain-Barré Syndrome: A Prospective Multicentre Study. Neuropediatrics 38: 10-17.
- Kannan Ma, Ch Rk, Jabeen Sa, Mridula Kr, Rao P, et al. (2011) Clinical, Electrophysiological Subtypes and Antiganglioside Antibodies in Childhood Guillain-Barré Syndrome. Neurol India 59: 727-732.
- Nachamkin I, Barbosa Pa, Ung H, Lobato C, Rivera Ag, et al. (2007) Patterns of Guillain-Barré Syndrome in Children: Results from a Mexican Population. Neurology 69: 1665-1671.
- Sankhyan N, Sharma S, Konanki R, Gulati S (2014) Childhood Guillain-Barré Syndrome Subtypes in Northern India. J Clin Neurosci 21: 427-430.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.