Anticoagulants: Emerging Candidates for Neurovascular Protection in Alzheimer´s Disease
by Klaus Grossmann*
Center for Plant Molecular Biology (ZMBP), University of Tübingen, 72076 Tübingen, Germany.
*Corresponding author: Klaus Grossmann, Center for Plant Molecular Biology (ZMBP), University of Tübingen, 72076 Tübingen, Germany.
Received Date: 16 April, 2026
Accepted Date: 22 April, 2026
Published Date: 24 April, 2026
Citation: Grossmann K (2026) Anticoagulants: Emerging Candidates for Neurovascular Protection in Alzheimer´s Disease. Int J Cerebrovasc Dis Stroke 9: 206. DOI: https://doi.org/10.29011/2688-8734.100206
Abstract
Alzheimer’s disease (AD) is the consequence of neurodegenerative, but also vasculopathic and hemostatic changes in the brain. By targeting thrombin, whose production is increased by proteins of amyloid-ß (Aß), anticoagulants could prevent effects of elevated thrombin and fibrin clot formation leading to inflammatory, thrombotic, and hemorrhagic vascular disorders in AD. Such therapeutic efficacy could sustain vascular function, improve cerebral perfusion and nutrient supply, reduce Aß accumulation, and preserve the central nervous system and its cognitive performance for longer. Vascular- and neuroprotective potential of anticoagulants is evident from recent research in animal models and from clinical observational studies on antithrombotic treatment (i) with oral anticoagulants (OACs) in atrial fibrillation patients and (ii) in users of parenteral heparin. However, randomized controlled trials evaluating therapeutic value of anticoagulants against neurovascular dysfunction, possibly extending their approval to AD, are still pending. The primary reasons for reluctance are the increased risk of intracerebral bleeding in AD and the necessity for specific patient selection, stratification, and monitoring strategies. OACs with reduced risk of increasing intracranial hemorrhage, such as the thrombin inhibitor dabigatran and inhibitors of blood clotting factor Xa in synthesis like apixaban, could be an option for hypothetical clinical trials. In future, factor XI/XIa inhibitors, which are currently being investigated for antithrombotic use and promise an even lower risk of bleeding, might also be of interest.
Keywords: Alzheimer’s disease, Neurovascular dysfunction, Amyloid-ß proteins, Thrombin, Fibrin(ogen), Anticoagulants
Introduction
Alzheimer’s disease (AD) is considered the most common form of dementia, affecting more than 50 million people worldwide [1-3]. In this disease, misfolded proteins of amyloid-ß (Aß) are deposited in the brain parenchyma in addition to neurotoxic tau proteins [2]. Accumulation of Aß damages neuron cells and their synaptic connections in the central nervous system, causing cognitive abilities, such as perception, memory, and thinking, to be lost. Usually, the disease process begins 10-20 years before cognitive impairment becomes apparent [1-3]. In parallel, Aß, originating from neurons, but also synthesized by cerebrovascular endothelial cells and activated platelets, accumulate in the blood and stimulate the blood clotting process generating a procoagulant state [4-6]. The serine protease thrombin is increasingly produced, leading to elevated formation of fibrin clots and thus a prothrombotic state [4-6] (Figure 1).

Simplified model according to [1, 4-10].
Among sequelae of elevated levels of Aß and proinflammatory and prothrombotic thrombin and fibrin(ogen) in the blood, the cerebral blood vessels in particular suffer - a disease phenomenon that has only recently been studied in more detail [1, 4-6, 10-13]. In fact, deposition of vascular Aß is considered a driver of AD pathogenesis and is the hallmark of a vasculopathy known as Aßtype cerebral amyloid angiopathy (CAA) [1, 9, 13]. CAA is found in over 80-95% of AD patients and is central to the vasculopathic biology of AD, directly relevant to thrombosis, fibrin (ogen), and microhemorrhagic risk [1, 4, 5, 9, 12, 13]. Blood vessels become inflamed, increasingly damaged and bleeding, blocked, and dysfunctional [1, 4-6, 10-13] (Figure 1). As a result, cerebral blood flow (CBF) chronically decreases in AD patients by about 25% and in some brain regions even by more than 50% of the normal level [12, 14]. The brain is no longer sufficiently perfused and supplied with oxygen and nutrients [1, 4-6, 10-13]. In addition, the endothelium of the vessel walls for selective exchange of substances with the central nervous system, the blood-brain barrier (BBB), is increasingly damaged during AD progression [11, 12]. Small molecules and proteins such as thrombin and fibrinogen can leak from the blood into the brain parenchyma where fibrinogen is converted to fibrin, forming neurotoxic deposits [9, 10-12]. Conversely, neurotoxic proteins such as Aß can no longer be transported into the blood and be removed from the brain through the process of perivascular clearance [11,12]. An inflammatory milieu evolves in the brain parenchyma that damages neurons and synaptic transmission in the nervous system, thereby impairing brain function [1, 10-12] (Figure 1). Preferentially, the neocortex and hippocampus of the brain are affected - in other words, areas that are important centres of information processing for cognitive, behavioural and motor skills [1, 2, 12].
This syndrome is known as neurovascular dysfunction and is increasingly recognized as an important driver of Aß pathology in AD pathogenesis [1, 4, 10, 12] (Figure 1). Consequently, research focus appears to gradually change from a solely neuronaloriented to a neurovascular, holistic view. A paradigm shift, which is encouraged by recent animal studies demonstrating that accumulation of Aß in cerebral blood vessels is causally connected with perivascular inflammation, vascular damage, and cognitive decline in AD [15]. But what should not be forgotten: Alois Alzheimer already reported on vascular dysfunction in the AD brain in 1906, and later, cerebrovascular amyloid was the starting material for the first isolation of Aß [1].
Therefore, this perspective article is focusing on neurovascular dysfunction caused by elevated thrombin as part of the Aß pathology in AD. The aim is to provide a short overview of the current state of research and to inspire discussion, why it might be worth bringing anticoagulants as a novel disease-modifying therapy in the centre of attention. For getting a more detailed survey on the topic, reference is made to recent, comprehensive reviews [1, 4, 5, 10, 16, 17].
Elevated Thrombin and Neurovascular Dysfunction - “Partners in Crime”
The main biological function of thrombin in the blood is - at the end of the coagulation cascade - to convert the soluble protein fibrinogen into insoluble fibrin, which forms a fibrous network with integrated red blood cells and platelets [10, 18] (Figure 1). A blood clot (thrombus) develops, which has the task of stopping bleeding from injured vessels and, in the course of its degradation, healing wounds [18]. However, in cardiovascular diseases such as atrial fibrillation (AF), the formation of fibrin clots can occlude blood vessels and trigger thrombosis or - if the clots are displaced in the vascular system - embolism in organs that can lead to heart attack or stroke [18]. Anticoagulants, commonly known as “blood thinners”, have been used for decades to treat and avoid thromboembolic events and associated emergencies [18]. These pharmaceuticals include heparins with parenteral action for shortterm treatment and the orally effective oral anticoagulants (OACs) that are used for temporarily as well as permanent treatment [18]. The drugs stop thrombin effects and thus inhibit blood clot formation and vascular inflammation in people at risk [18]. However, as adverse side effect, use of anticoagulants increases the risk of bleeding [1, 16-18]. In Germany alone, OACs are currently administered to over 3 million patients, who are mostly over the age of 70, also including patients with dementia like AD [1]. According to a Finnish cohort study, prevalence of OAC use is 23% among people with AD, especially administered because of AF [19].
In AD, vascular Aß activate factor XII (FXII) in the coagulation cascade, leading to elevated production of thrombin and subsequently fibrin [4-9, 20] (Figure 1). This creates an inflammatory, vessel damaging, and thrombosis-promoting milieu within the cerebral vascular system (Figure 1). In the last years, hemostatic dysregulation has been recognized as a vasculopathic component of Aß pathology, which is involved in AD onset and progression [4-7, 10, 12, 17]. Triggered sequelae include inflammation, hemorrhage, occlusion, and BBB dysfunction of cerebral vessels, which restricts CBF, perfusion, and nutrient supply of the brain, as well as impairs perivascular clearance of Aß [1, 4-6, 10-12, 14, 17] (Figure 1). As a result, accumulation of Aß, thrombin, fibrin(ogen), and fibrin complexes with Aß occurs both in the vasculature and brain parenchyma [4-7, 10, 12]. Neuroinflammatory and neurotoxic alterations and tau pathologies gradually increase, leading eventually to damage of the nervous system and decline of the brain function [4-7, 10, 12] (Figure 1). While these consequences of hemostatic dysregulation are well documented in Aß pathology, the underlying molecular mechanisms are not completely understood and require further research in animal models and AD patients [1, 5, 6, 17, 21, 22]. In addition, the temporal dynamics (i) in the developing procoagulant state, (ii) in the accumulation of thrombin, fibrin(ogen), and Aß in the vascular and parenchymal brain tissue, and (iii) in the resulting processes implicated in neurovascular dysfunction and cognitive impairment have to be studied in more detail [1, 5, 17, 21].
Besides their stimulating effect on thrombin synthesis, Aß have also the capacity of attaching themselves to fibrin clots and thus blocking their degradation by fibrinolysis [4, 5, 7-9, 12, 17, 23] (Figure 1). Such more stable, Aß-containing fibrin clots are deposited in the cerebral blood vessels together with small Aß aggregates, leading to vascular disease, especially CAA [5, 6, 9, 12, 13, 17] (Figure 1). Pathophysiological consequences of CAA include that affected areas of the brain are less and less supplied with blood (hypoperfusion) and tissues suffer dramatically from a lack of oxygen (hypoxia) and nutrients like glucose (hypoglycemia) [1, 4, 6, 9, 12, 13]. This milieu further stimulates the production and accumulation of pathogenic Aß in the brain parenchyma [1, 2, 4, 12]. Likewise, parenchymal deposition of thrombin, fibrin(ogen), and fibrin complexes with Aß are increasing as a result of progressing BBB dysfunction of the cerebral vessels [1, 4, 10-13]. Glial cells (e.g., microglia, astrocytes) are activated and release proinflammatory proteins (e.g., cytokines, chemokines) and reactive oxygen and nitric oxide species in the brain. Furthermore, myelinating oligodendrocytes, necessary for proper neuronal signaling, are disrupted [1, 10-13]. Like in a vicious circle, inflammatory and degenerative processes in the nervous tissue are intensified that drive each other, leading to the death of neurons and their synaptic connections and to further dementia progression.
Can Anticoagulants Help to Counteract Alzheimer´s?
For treating hemostatic dysregulation and associated neurovascular dysfunction in AD, OACs would act as diseasemodifying/preventive therapies due to their mode of action and pharmacological properties [1, 5, 10, 16-18, 21]. OACs include the classic vitamin K antagonists (VKAs), such as phenprocoumon (Marcumar®) and warfarin (Coumadin®), which are originally derivatives of the plant substance dicumarol. In addition, a new class of OACs, the direct oral anticoagulants (DOACs), has been introduced for antithrombotic treatment in the last 15 years. While the VKAs inhibit vitamin K-dependent synthesis of a variety of coagulation factors, the DOACs specifically block thrombin, either directly (dabigatran, Pradaxa®) or indirectly its synthesis by inhibition of coagulation factor Xa (apixaban, Eliquis®; betrixaban, Bevyxxa®; edoxaban, Lixiana®; rivaroxaban, Xarelto®) [18, 22] (Figure 1).
Evidence for possible therapeutic value of OACs against AD highlights mainly from preclinical studies in genetically modified mice [1, 10, 17, 21, 24-27]. These animal models of AD allow a detailed systems biology approach attempting to replicate Aß-, tau-, and CAA-associated pathologies in the human brain. Of special importance are recent studies in AD mice performed by Cortes-Canteli and co-workers [25]. The results demonstrated that long-term anticoagulation with dabigatran restrains deposition of toxic fibrin in the brain, preserves cerebrovascular function, BBB integrity, CBF, and brain perfusion, and prevents memory decline [25]. In addition, deposition of oligomeric Aß and amyloid plaques as well as the extent of glial activity and neuroinflammation in the brain parenchyma decreased. Furthermore, no intracerebral bleeding was observable [25]. Subsequent studies by Bian and colleagues [26, 27] using an AD mice model with strong CAA predisposition confirmed protective effects of OACs. Longterm administration of rivaroxaban attenuates BBB dysfunction, fibrinogen and Aß deposition, and neuroinflammation including fibrinogen-related microglia activation, oxidative stress, and demyelination. Brain white matter integrity was improved and axonal damage and memory deficits were reduced [26, 27].
Based on these findings, a model has been proposed showing the potential mechanism of action of OACs against neurovascular dysfunction in AD [1, 5, 10, 16, 17, 22] (Figure 2). By neutralizing Aß-induced elevated thrombin, OAC treatment could prevent deposition of fibrin clots and degradation-resistant Aß-containing fibrin clots (fibrin/-Aß clots) as well as perivascular inflammation in the brain vessels. Thereby, development of vasculopathies such as CAA could be mitigated and vascular and BBB function preserved, also aiding Aß clearance from the brain (Figure 2). CBF as well as perfusion and nutrient supply to the brain tissue could be improved (Figure 2). Parenchymal accumulation of pathogenic Aß, thrombin, fibrin(ogen), and fibrin complexes with Aß could be reduced and associated neuroinflammatory and degenerative changes in the nervous system and progress of dementia mitigated (Figure 2). OACs are believed to be particularly effective when administered in the early stages of Aß pathology in AD [1, 22]. Overall, this mechanistic model could be a useful basis to guide preclinical and clinical research for analyzing vascular- and neuroprotective efficacy of anticoagulants in more detail [1, 4, 10, 16, 17].
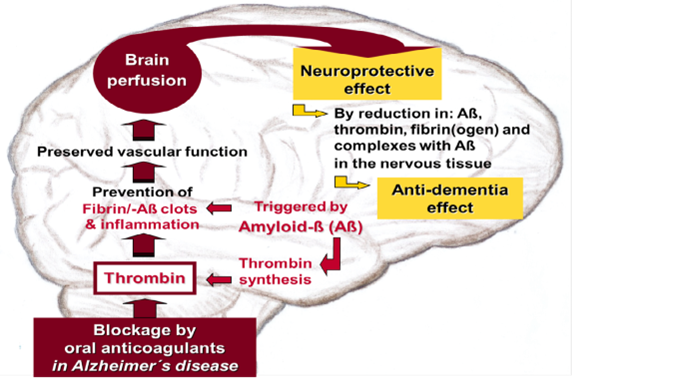
Figure 2: Proposed model on the therapeutic benefit of oral anticoagulants (OACs) in Alzheimer´disease. OACs block thrombin or its synthesis in the coagulation process, which is stimulated by toxic proteins of amyloid-ß (Aß). Normalizing Aß-induced hemostatic dysregulation by OACs could prevent effects of elevated thrombin and fibrin clot formation leading to inflammatory, thrombotic, and vascular disorders. Thus, development of vascular diseases in the brain, such as Aß-type cerebral amyloid angiopathy (CAA), could be mitigated. CAA is particularly triggered by deposition of Aß aggregates and fibrin and stable Aß-containing fibrin clots (fibrin/-Aß clots) as well as perivascular inflammation in the blood vessels. OAC treatment could preserve vascular and blood-brain barrier function, allowing cerebral blood flow and brain perfusion and nutrient supply to the nervous tissue be maintained. Thereby, parenchymal accumulation of inflammatory Aß, thrombin, fibrin(ogen), and fibrin complexes with Aß could be reduced. The nervous system could be protected from neuroinflammatory and degenerative consequences of vascular dysfunction and cognitive performance be kept for longer. Simplified model according to [1, 22].
Support for this therapeutic approach comes from numerous clinical retrospective and prospective observational studies, which provide evidence that OACs, administered due to AF, may protect patients against dementia like AD [1, 17, 22, 28-31]. Besides risk of suffering thromboembolism, AF increases the risk of AD by 3050% and doubles the risk of vascular dementia [28]. In 2019, about 60 million people were affected by AF worldwide [18]. Noteworthy, in a Swedish retrospective registry study including 444,106 AF patients (mean age above 70 years), OAC treatment was shown decreasing risk of dementia by up to 48% compared to non-OAC users (hazard ratio [HR] 0.52, 95% Cl 0.50-0.55) [29]. Early initiation of OAC treatment has been suggested to be particularly effective to preserve cognitive function [29]. In particular, elderly (>65 years), but also individuals with a low risk of AF or newly diagnosed ones, benefited from OAC treatment [30]. A recent systematic review and meta-analysis of data from a total of 22 observational studies involving 617,204 AF patients confirmed anti-dementia potential of OACs compared to non-use (HR 0.68, 95% Cl 0.58-0.80, p < 0.00001), with similar outcome for DOACs and VKAs [31]. With regard to the type of OACs administered, a recent meta-analysis of data from 9 studies involving 447,644 AF patients revealed that DOAC treatment was more effective in lowering incidence of dementia than use of VKA-type warfarin (risk ratio [RR] 0.692, 95% Cl 0.603-0.793, P=0.000), especially pronounced in AD [32]. However, in a randomized control trial (BRAIN-AF) including 1,235 AF patients with low risk of stroke
(mean age 53 years), DOAC-type rivaroxaban at reduced dose (15 mg daily) did not influence incidence of stroke and cognitive decline compared to placebo [33]. This lack of effect has been explained by the study cohort, which consisted of relatively young, low-risk participants with no real benefit from anticoagulant treatment [33]. Likewise, the underdose of rivaroxaban could have also be contributing in its ineffectiveness [33].
When compared to OACs, parenteral anticoagulants, such as the indirectly thrombin-inhibiting heparins (e.g., enoxaparin), are less suitable for long term-treating neurovascular dysfunction in AD due to pharmacological reasons [22]. Cons include invasive administration, triggering of thrombocytopenia, increased risk of bleeding, unpredictable anticoagulation by unspecific plasma protein binding, and insufficient deactivation of fibrin-bound thrombin and associated thrombus formation [22]. Nevertheless, a recent study evaluating data from different US health systems revealed that anticoagulant therapy with heparin like enoxaparin could have potential to protect from developing AD [34]. In fact, preclinical studies in enoxaparin-treated AD mice were among the first showing that anticoagulation with heparin can reduce deposition of Aß as well as inflammatory, neurotoxic, and cognitive impairment in the brain [24].
DOAKs: Blood-flow-promotive, Neuroprotective, Cognitionpreserving
Besides changing lifestyle and reducing cardiovascular risk factors to counteract AD, treatment with standard-of-care drugs in patients is hitherto only able to alleviate some symptoms of the disease, improve general condition, and slow down mild cognitive impairment [3, 22]. Recently, monoclonal antibody agents against Aß (lecanemab, donanemab) were approved for treatment of AD, which give hope of combating causes of the disease [3, 35]. In clinical, 18-month phase III trials, both agents slowed down progression of cognitive and physical impairment in early ADstage-patients modestly by around 30% [3, 35]. Data indicate that anti-amyloid therapies are most effective when administered during the earliest symptomatic stages of AD [3,35]. However, these expensive therapies were not able to stop or even cure AD. Moreover, adverse side effects, referred to as amyloid-related imaging abnormalities (ARIA) involving localized brain swelling (ARIA-E for edema) and microhemorrhages (ARIA-H for cerebral hemorrhage), were observed in some patients. Most of which were mild, but some of which were severe [1, 3, 5, 35]. Fatal brain bleeding (ARIA-H) was found predominantly in AD patients with significant CAA and inflammation, but also in those receiving anticoagulation for stroke risk prevention due to AF [3, 35]. Therefore, anti-amyloid therapies are currently contraindicated in patients with severe CAA or anticoagulant use [35]. However, a recent systematic review and meta-analysis found no increased risk of ARIA among AD patients receiving anti-amyloid antibody therapies while on OAC use [36]. Extreme caution or contraindication are also required in AD patients carrying one of the strongest and most widespread genetic risk factors of AD, the e4-allele of the gene encoding apolipoprotein E (APOE) [35]. Those individuals face a three- to sixfold higher risk of developing antibody-induced ARIA [35].
Considering limitations of the usual “symptomatic” drugs and the new anti-amyloid therapies, other plausible approaches - especially cost-effective repurposing of approved drugs - remain valuable treatment options for AD [3, 22]. Particularly, OACs - due to their anti-thrombotic and anti-dementia potential - might be beneficial not only for AF or other cardiovascular diseases, but possibly also for AD. OAC treatment could neutralize - prophylactically (in cases of hereditary predisposition) or therapeutically - hemostatic dysregulation, thus preventing neurovascular dysfunction and derived cognitive impairment [1, 5, 10, 16, 17, 21, 22]. Intriguingly, first clinical studies treating dementia with VKAtype anticoagulants go back to the 1960s [37]. These still smallscale, partially placebo-controlled trials revealed a slowdown of cognitive decline in treated elderly and presenile individuals [37]. However, these positive results did not prompt subsequent clinical trials in larger cohorts [1, 10, 17, 21, 22].
When thinking about conducting clinical trials to evaluate benefits and harms of anticoagulants in AD, use of OACs from DOACtype, such as dabigatran, apixaban, and rivaroxaban, is a plausible idea. Arguments in favor of these DOACs are (i) their specific and rapid inhibition of thrombin or factor Xa in biosynthesis at short half-life and fixed dosing, (ii) a comprehensive clinical data base on antithrombotic efficacy, bleeding risk, and antidementia potential especially in elderly, (iii) no interference with vitamin K-dependent metabolism and dietary, (iv) available antidote strategies that can stop serious bleeding in minutes, and (v) their reduced risk of dangerous intracranial hemorrhage by average 50% compared to VKA treatments [1, 17, 22, 38-42]. In a comprehensive retrospective FDA study including 134,414 elderly AF patients, dabigatran decreased incidence of serious intracranial hemorrhage compared to warfarin by 66% (from 9.6 cases per 1000 person-year with warfarin to 3.3 cases with dabigatran; HR 0.34, 95% Cl 0.26-0.46) [38]. However, the risk of major gastrointestinal bleeding raised by 28% compared to warfarin (HR 1.28, 95% Cl 1.14-1.44) [38]. Therefore, in patients showing high gastrointestinal bleeding risk or severe renal dysfunction as well as in patients with mechanical heart valve implants, use of VKAs is favoured [1, 18, 22]. However, dabigatran´s safety concerning gastrointestinal bleeding might be improved by administration of lower doses [39]. Overall, in recent retrospective observational studies including elderly AF patients with dementia, particularly apixaban [41] and dabigatran [42] have proven to be the preferred OAC options with respect to stroke prevention and safety.
A new class of anticoagulants might also be suited treating hemostatic dysregulation and sequelae in AD [1,43]. These drugs, which are currently in clinical investigation, neutralize factor XI/ XIa in blood clotting [43] (Figure 1). They include the DOACs asundexian and milvexian as well as parenteral antibodies like abelacimab and antisense oligonucleotides like fesomersen, which have to be administered about once a month invasively [43]. It is assumed that such inhibitors have a reduced risk of bleeding, as coagulation factor XI plays a subordinate role in hemostasis, but contributes significantly to the growth of a thrombus [43].
Conclusions
Therapeutic Potential
Whether used alone or in combination with anti-inflammatory, cardiovascular active, standard dementia, or new disease-modifying drugs, anticoagulants might have potential to become part of an integrated treatment strategy, aimed at mitigating vascular-driven neuropathogenesis and associated cognitive decline in AD (Figure 2). However, further preclinical research and primarily clinical intervention trials are necessary (i) in getting more information how anticoagulants can protect against neurovascular dysfunction, (ii) in evaluating their therapeutic potential and bleeding risk in AD, and (iii) in working out guidelines for their possible use [1, 17, 21, 22, 34]. Currently, especially DOACs, such as dabigatran, apixaban, and rivaroxaban, could be suggested for this new medical indication, since they are approved drugs in anti-thrombotic cardiology, have a known safety profile, and are applied over more than 15 years.
Limitations
When thinking about neuroprotective use of anticoagulants like the DOACs in clinical practice, best therapeutic success could probably be expected in AD patients, who are - indicated by imaging and biochemical diagnostic methods - in the early stage of Aß pathology, CAA, and procoagulant state, show low bleeding risk, and perceive none or only mild cognitive impairment [1, 17, 22]. However, not all AD patients appear to exhibit procoagulant state [20]. Therefore, diagnostic and screening strategies of procoagulant state are required to identify individuals that may benefit from anticoagulation and minimize the bleeding risk in those, who would not [17]. For details on the prerequisites, framework conditions, and diagnostic tools necessary for a random clinical trial on DOAC use in AD, reference is made to recent review articles [22, 44]. In principle, anticoagulants should only then be prescribed, if - after careful evaluation - presumed benefits for vascular and cognitive condition outweigh bleeding risk. Accordingly, a recent Taiwanese observational study showed that in AD, increased risk of intracerebral hemorrhage is further elevated by anticoagulant use, preferentially in individuals with comorbidities such as diabetes and heart failure [45]. Therefore, prerequisite for any anticoagulant use - especially in elderly with fragile blood vessels and risk of CAA - is an individualized assessment of the bleeding risk and procoagulant and CAA state by, e.g., clinical HAS-BLED score criteria and biochemical and imaging procedures. In addition, careful individual adjustment of the dosing strategy and close medical monitoring are required to prevent severe complications, particularly intracranial bleeding [1, 10, 17, 18, 22, 45].
Future Directions
Overall, the recent preclinical findings on mitigating hemostatic and neurovascular dysfunction, together with the observed antidementia potential in AF patients are good reasons for clinically investigating therapeutic value of anticoagulants in AD. However, up to now randomized controlled trials with appropriate DOACs for evaluating benefits and risks of treatment and possibly extending their approval to AD are still pending [1, 10, 17, 21, 22]. As outlined before, the primary reasons for reluctance are the increased risk of intracerebral bleeding in AD and the need for specific patient selection and stratification strategies and diagnostic AD, CAA, and safety monitoring, thereby driving up clinical trial costs. DOACs with reduced risk of increasing intracranial hemorrhage, such as the thrombin inhibitor dabigatran or inhibitors of blood clotting factor Xa in synthesis like apixaban and rivaroxaban, could be an appropriate option for a hypothetical clinical trial. In future, factor XI/XIa inhibitors, which are currently being investigated for antithrombotic use and promise an even lower risk of bleeding [43], might also be of interest. Until then, use of OACs or other anticoagulants, e.g., heparin [34], is currently only indicated within the scope of the approved, anti-thromboembolic therapies for preventing or treating blood clots under medical supervision [1, 16-18, 22]. Any potential individual off-label use of anticoagulants against AD - with the aim of improvement of this hitherto incurable disease [46] - is currently considered with reservation among neurologists in Germany. The opinion is that such therapy should initially only be carried out as part of a clinical study - optimally as a randomized controlled trial (personal communication).
Acknowledgements
Cordial thanks are due to my wife, Regina, for her helpful and encouraging support in this work and in our engagement in the care home, together with people suffering from dementia.
Funding
This work received no funding. For open access publishing of this article, the author gratefully acknowledges support by the Open Access Publishing Fund of the University of Tübingen, Germany.
Conflict of Interest
The author declares no conflict of interest in connection with this manuscript.
Authors Contributions
The author contributed to drafting and revising the article and agree to be accountable for all aspects of the work.
Ethical Approval Not applicable.
Consent to Participate Not applicable.
Consent for Publication Not applicable.
Availability of Data and Material Not applicable.
Code Availability Not applicable
References
- Grossmann K (2024) Unraveling the thrombin-Alzheimer´s connection: oral anticoagulants as potential neuroprotective therapeutics. Adv Neurol 3:3799.
- Scheres SHW, Ryskeldi-Falcon B, Goedert M (2023) Molecular pathology of neurodegenerative diseases by cryo-EM of amyloids. Nature. 621:701-710.
- Xiao D, Zhang C (2024) Current therapeutics for Alzheimer´s disease and clinical trials. Explor Neurosci 3:255-271.
- Casquero-Veiga M, Ceron C, Cortes-Canteli M (2025) Alzheimer´s disease and vascular biology - a focus on the procoagulant state. Curr Op in Cell Biol 95: 102528.
- Badimon A, Torrente D, Norris EH (2023) Vascular dysfunction in Alzheimer´s disease: alterations in the plasma contact and fibrinolytic systems. Int J Mol Sci 24:7046.
- Strickland S (2018) Blood will out: vascular contributions to Alzheimer´s disease. J Clin Invest 128:556-563.
- Zamolodchikov D, Chen Z.-L, Conti BA, Renne T, Strickland S (2015) Activation of the factor XII-driven contact system in Alzheimer’s disease patient and mouse model plasma. Proc Natl Acad Sci. USA 112:4068-4073.
- Zamolodchikov D, Renne T, Strickland S (2016) The Alzheimer’s disease peptide ß-amyloid promotes thrombin generation through activation of coagulation factor XII. J Thromb Haemost 14:995-1007.
- Cajamarca SA, Norris EH, Van der Weerd L, Strickland S, Ahn HJ (2020) Cerebral amyloid angiopathy-linked ß-amyloid mutations promote cerebral fibrin deposits via increased binding affinity to fibrinogen. Proc Natl Acad Sci USA. 117:14482-92.
- Iannucci J, Grammas P (2023) Thrombin, a key driver of pathological inflammation in the brain. Cells 12:1222.
- Profaci CP, Munij RN, Pulido RS, Daneman R (2020) The blood-brain barrier in health and disease: important unanswered questions. J Exp Med. 217: e20190062.
- Sweeney MD, Montagne A, Sagare AP, Nation DA, Schneider LS, et al. (2019) Vascular dysfunction-the disregarded partner of Alzheimer’s disease. Alzheimers Dement. 15:158-167.
- Greenberg, SM, Bacskai BJ, Hernandez-Guillamon M, Pruzin J, Sperling R, et al. (2020) Cerebral amyloid angiopathy and Alzheimer disease - one peptide, two pathways. Nat Rev Neurol 16:30-42.
- Asslani I, Habeck C, Scarmeas N, Borodovac A, Brown TR, et al. (2008) Multivariate and univariate analysis of continuous arterial spin labeling perfusion MRI in Alzheimer’s disease. J Cereb Blood Flow Metab 28:725-736.
- Feng Y, Wang S, Yang D, Zheng W, Xia H, et al. (2025) Inhibition of IFITM3 in cerebrovascular endothelium alleviates Alzheimer´s-related phenotypes. Alzheimers Dement. 21: e14543.
- Grossmann K (2020) Anticoagulants for treatment of Alzheimer’s disease. J Alzheimers Dis. 77:1373-1382.
- Toribo-Fernandez R, Ceron C, Tristao-Pereira C, Fernandez-Nueda I, Perez-Castillo A, et al. (2024) Oral anticoagulants: a plausible new treatment for Alzheimer´s disease? Br J Pharmacol. 181:760-776.
- Todorova N, Mateeva A, Savova A, Kondeva-Burdina M (2025) Current insights on cardiovascular and neuro- degenerative relations of direct oral anticoagulants and their biotransformation. Pharmacia. 72:1-14.
- Ali Babar B, Vu M, Koponen M, Taipale H, Tanskanen A, et al. (2022) Prevalence of oral anticoagulant use among people with and without Alzheimer´s disease. BMC Geriatrics. 22:464.
- Cortes-Canteli M, Mattei L, Richards AT, Norris EH, Strickland S (2015) Fibrin deposited in the Alzheimer’s disease brain promotes neuronal degeneration. Neurobiol Aging 36:608-617.
- Madivalar V, Teli S, Hiremath S, Chatter V, Shrinivas G, et al. (2025) Anticoagulant therapy for Alzheimer´s disease: a review of recent research. Int J Conv Res. 2:10-23.
- Grossmann K (2022) Direct oral anticoagulants (DOACs) for therapeutic targeting of thrombin, a key mediator of cerebrovascular and neuronal dysfunction in Alzheimer´s disease. Biomedicines.10:1890.
- Cortes-Canteli M, Paul J, Norris EH, Bronstein R, Ahn HJ, et al. (2010) Fibrinogen and ß-amyloid association alters thrombosis and fibrinolysis: a possible contributing factor to Alzheimer’s disease. Neuron. 66:695-709.
- Bergamaschini L, Rossi E, Storini C, Pizzimenti S, Distaso M, et al. (2004) Peripheral treatment with enoxaparin, a low molecular weight heparin, reduces plaques and ß-amyloid accumulation in a mouse model of Alzheimer’s disease. J Neurosci. 24:4181-4186.
- Cortes-Canteli M, Kruyer A, Fenandez-Nueda I, Marcos-Diaz A, Ceron C, et al. (2019) Long-term dabigatran treatment delays Alzheimer’s disease pathogenesis in the TgCRND8 mouse model. J Am Coll Cardiol. 74:1910-1923.
- Bian Z, Liu X, Feng T, Yu H, Hu X, et al. (2022) Protective effect of rivaroxaban against amyloid pathology and neuroinflammation through inhibiting PAR-1 and PAR-2 in Alzheimer´s disease mice. J Alzheimers Dis 86:111-123.
- Bian Z, Hu X, Liu X, Yu H, Bian Y, et al. (2023) Protective effects of rivaroxaban on white matter integrity and remyelination in a mouse model of Alzheimer´s disease combined with cerebral hypoperfusion. J Alzheimers Dis 96:609-622.
- Agarwal R, Tully PJ, Mahajan R (2024) Cognitive function in atrial fibrillation: a narrative review of evidence and mechanisms. Heart Mind. 8:100-110.
- Friberg L, Rosenqvist M (2018) less dementia with oral anticoagulation in atrial fibrillation. Eur Heart J. 39:453-460.
- Friberg L, Andersson T, Rosenqvist M (2019) less dementia and stroke in low-risk patients with atrial fibrillation taking oral anticoagulation. Eu Heart J. 40:2327-2335.
- Latif F, Nasir MM, Meer KK, Farhan SH, Cheema HA, et al. (2024) The effect of oral anticoagulants on the incidence of dementia in patients with atrial fibrillation: a systematic review and meta-analysis. J Cardiol Cardiovasc Risk Prev.21: 200282.
- Zhang C, Zhang J, Zhao X, Jiang D, Liu X, et al. (2024) Association of direct oral anticoagulants and warfarin with incidence of dementia in atrial fibrillation patients: a systematic review and meta-analysis. IJC Heart Vascul. 54: 101401.
- Rivard L, Khairy P, Talajic M, Tardif JC, Nattel S, et al. (2024) Blinded randomized trial of anticoagulation to prevent ischemic stroke and neurodegenerative impairment in AF (BRAIN-AF): methods and design. Can J Cardiol. 35:1069-1077.
- Readhead B, Klang E, Gisladottir U, Vandromme M, Li L, et al. (2025) Heparin treatment is associated with delayed diagnosis of Alzheimer´s dementia in electronic health records from two large United States health systems. Molecular Psychiatry. 30:1461-1465.
- Vukmir RB (2024) Amyloid-related imaging abnormalities (ARIA): diagnosis, management, and care in the setting of amyloid-modifying therapy. Ann Clin Transl Neurol. 11: 1669-1680.
- Schlemm E, Gauthier S, Magnus T, Thomalla G, Rosa-Neto P, et al. (2025) Risk of amyloid-related imaging abnormalities associated with anticoagulant therapy in patients with Alzheimer´s disease treated with anti-amyloid monoclonal antibodies: a systematic review and metaanalysis. J Neurol Neurosurg Psychiatry.97:367-368.
- Walsh AC, Walsh BH, Melaney C (1978) Senile-presenile dementia: follow-up data on an effective psychotherapy-anticoagulant regimen. J Am Geriatr Soc. 26:467-470.
- Graham DJ, Reichman ME, Wernecke M, Zhang R, Southworth MR, et al. (2015) Cardiovascular, bleeding, and mortality risks in elderly Medicare patients treated with dabigatran or warfarin for nonvalvular atrial fibrillation. Circulation. 131:157-164.
- Ruff CT, Giugliano RP, Braunwald E, Hoffman EB, Deenadayalu N, et al. (2014) Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a metaanalysis of randomised trials. Lancet. 383:955- 962.
- Bonand C, Garcia-Blas S, Llergo JT, Fernández-Olmo R, DíezVillanueva P, et al. (2021) Direct oral anticoagulants versus warfarin in octogenarians with nonvalvular atrial fibrillation: a systematic review and meta-analysis. J Clin Med. 10:5268.
- Lin KJ, Singer DE, Bykov K, Bessette LG, Mastrorilli JM, et al. (2023) Comparative effectiveness and safety of oral anticoagulants by dementia status in older patients with atrial fibrillation. JAMA Network Open. 6: e234086.
- Fang C-W, Hsieh C-Y, Yang H-Y, Tsai C-F, Sung S-F (2025) Comparative effectiveness and safety of direct oral anticoagulants in atrial fibrillation patients with dementia. Thrombosis Res. 250: 109332.
- Capodanno D, Alexander JH, Bahit MC, Eikelboom JW, Gibson CM, et al. (2025) Factor XI inhibitors for the prevention and treatment of venous and arterial thromboembolism. Nature Rev Cardiol. 22:896912.
- Zetterberg H, Bendlin BB (2026) Biofluid biomarkers in Alzheimer´s disease and other neurodegenerative dementias. Nature. 650:49-59.
- Hsiao CH, Wu CE, Wang CM, Lin TY, Wu RY, et al. (2025) Intracerebral hemorrhage risk in Alzheimer´s disease patients on anticoagulants: a nationwide cohort study. J Alzheimers Dis. 107:769-777.
- Grossmann K (2021) Alzheimer-Krankheit - können Antikoagulantien helfen? J Neurol Neurochir Psychiat. 22:7-10.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.