Acute Myeloid Leukemia with MECOM Rearrangement Presenting with Dim B-Lineage Antigen Expression: A Diagnostic Pitfall in Flow Cytometric Lineage Assignment
by Chen Glait Santar*, Ben-Zion Katz
The Cellular Immunophenotyping Unit, Hematology Institute, Tel Aviv Sourasky Medical Center, Tel Aviv, Israel
*Corresponding author: Glait-Santar Chen, The Cellular Immunophenotyping Unit, Hematology Institute, Tel Aviv Sourasky Medical Center, Tel Aviv, Israel
Received Date: 08 March 2026
Accepted Date: 12 March 2026
Published Date: 16 March 2026
Citation: Santar CG, Katz BZ (2026) Acute Myeloid Leukemia with MECOM Rearrangement Presenting with Dim B-Lineage Antigen Expression: A Diagnostic Pitfall in Flow Cytometric Lineage Assignment. Ann Case Report. 11: 2561. DOI: https://doi.org/10.29011/2574-7754.102561
Abstract
Multiparameter flow cytometry is central to lineage assignment in acute leukemia; however, aberrant antigen expression may create diagnostic ambiguity. We report a 38-year-old man presenting with leukocytosis, severe anemia, and thrombocytopenia. Standardized EuroFlow immunophenotyping demonstrated 90% circulating blasts expressing dim CD19 and dim cytoplasmic CD79a, partial surface and cytoplasmic CD22, nuclear TdT, CD34, bright CD117, CD13, CD123, CD7, CD4, and HLA-DR. CD10, cytoplasmic IgM, CD33, and myeloperoxidase were negative. The immunophenotype suggested pro-B acute lymphoblastic leukemia (ALL) or acute leukemia of ambiguous lineage. Conventional cytogenetics revealed monosomy 7 and inv (3)(q21q26)/t(3;3)(q21;q26). Molecular studies identified FLT3-ITD and additional myeloid-associated mutations (NRAS, BCOR, WT1). These findings established acute myeloid leukemia (AML) with MECOM rearrangement according to current WHO and ICC classifications. This case highlights a diagnostic pitfall in which dim B-lineage antigens expression within a standardized framework may obscure an AML-defining genomic abnormality, underscoring the necessity of integrated morphologic, immunophenotypic, and genetic evaluation.
Keywords: Leukemia classification; Flow cytometry; Diagnostic pitfall; EuroFlow; Ambiguous lineage; Chromosomal rearrangement.
Introduction
Accurate lineage assignment in acute leukemia relies on integration of cytomorphology, multiparameter flow cytometry (MFC), cytogenetics, and molecular genetics. Standardized EuroFlow panels have improved reproducibility and sensitivity in acute leukemia diagnostics [1, 2]. Nevertheless, lineage assignment remains challenging when blasts demonstrate cross-lineage or aberrant antigen expression.
Recent classification systems prioritize genetically defined AML entities, including AML with MECOM rearrangement associated with inv(3)(q21q26.2) or t(3;3)(q21;q26.2) [3, 4]. These rearrangements involve the MDS1 and EVI1 complex locus at chromosome 3q26.2, resulting in dysregulated EVI1 oncogene expression. In such cases, genomic findings supersede immunophenotypic ambiguity for disease classification. We describe a case in which standardized immunophenotyping suggested B-lineage leukemia, whereas cytogenetic and molecular findings established AML with MECOM rearrangement, illustrating a clinically significant diagnostic trap.
Case Presentation and Cytopathologic Findings
A 38-year-old previously healthy male presented with recurrent oropharyngeal infection and constitutional symptoms. Complete blood count revealed anemia with 5.8 g/dL hemoglobin, leukocytosis (41.5×10³/µL), and thrombocytopenia (80×10³/µL). Peripheral blood smear (Figure 1A) demonstrated numerous circulating blasts with high nuclear-to-cytoplasmic ratio, finely dispersed chromatin, inconspicuous nucleoli, and scant basophilic cytoplasm without Auer rods. Some of the blasts had a "hand mirror" morphology (arrows), typical to acute lymphoblastic leukemia. Bone marrow aspirate (Figure 1B) revealed approximately 80% blasts with similar morphology, some mitotic forms, without definitive cytoplasmic granulation, rendering lineage determination by morphology alone inconclusive.
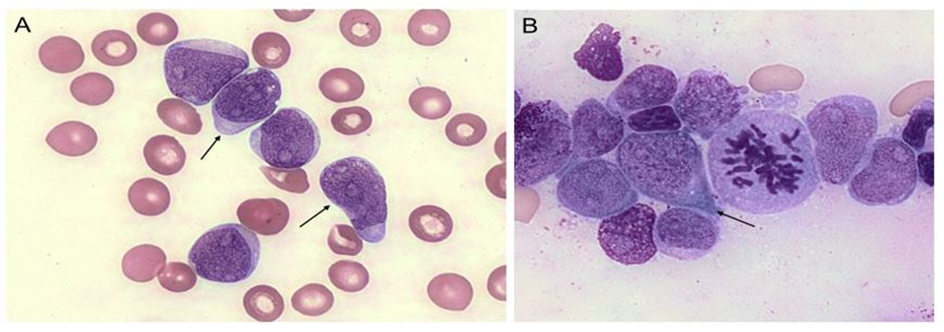
Figure 1: Peripheral blood and bone marrow cytomorphology demonstrating blast predominance without definitive lineage-specific features. (A) Peripheral blood smear showing numerous circulating blasts characterized by high nuclear-to-cytoplasmic ratio, finely dispersed chromatin, inconspicuous nucleoli, and scant to moderate basophilic cytoplasm. Arrows: "hand mirror" morphology. (B) Bone marrow aspirate smears reveal sheets of morphologically similar blast cells with frequent mitotic figures. The blasts lack cytoplasmic granulation and Auer rods. Arrow: "hand mirror" morphology. Overall, the cytomorphologic findings confirm acute leukemia but do not demonstrate clear myeloid differentiation, underscoring the need for integrated immunophenotypic and genetic evaluation for accurate lineage assignment (Wright-Giemsa stain).
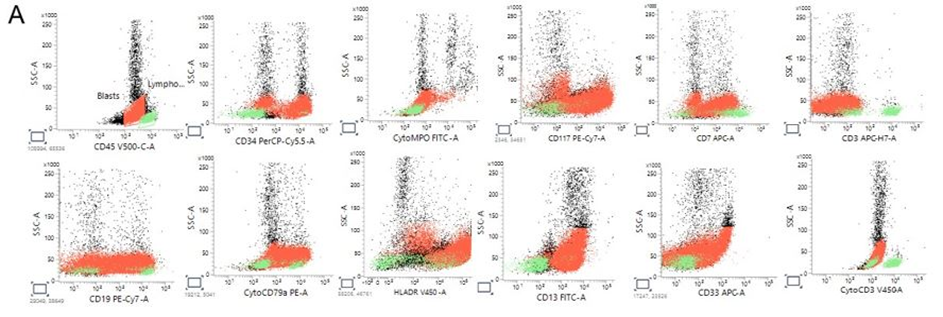
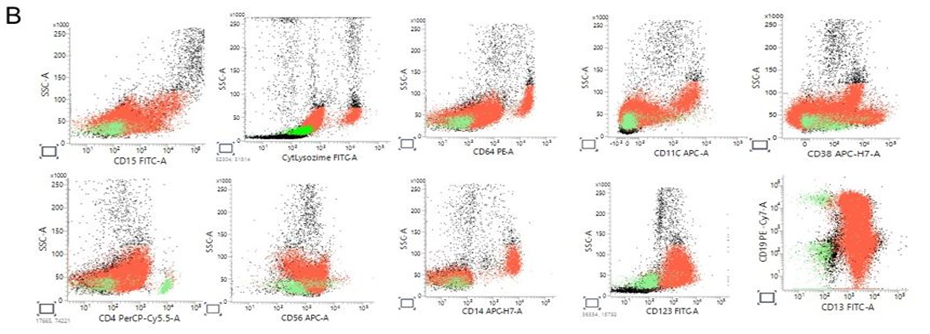
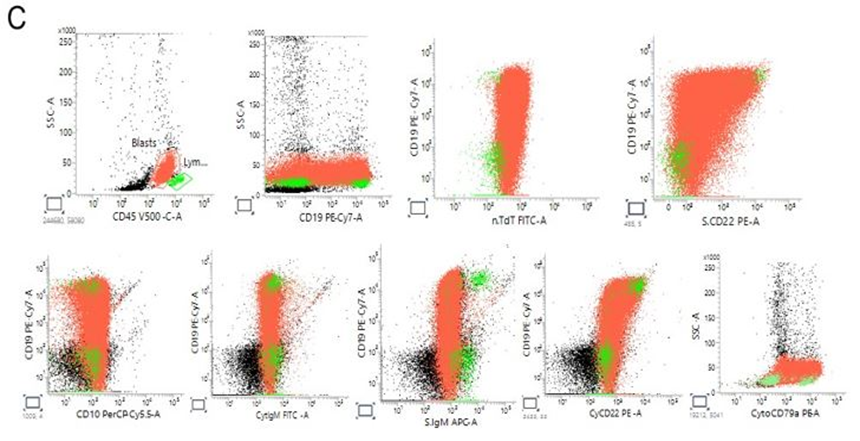
Figure 2: Flow cytometry analysis. (A). Peripheral blood multiparameter flow cytometry demonstrating aberrant blast population with mixed immunophenotypic features. Blasts (red) are identified on CD45 versus side scatter (SSC) by dim CD45 expression and low SSC, distinct from residual lymphocytes (green). The blast population shows bright CD117 expression and positivity for CD13 and CD33, with partial CD15 expression. Cytoplasmic MPO is negative. HLA-DR is expressed. Notably, the blasts demonstrate dim CD19 and dim cytoplasmic CD79a expression with partial surface and cytoplasmic CD22 positivity, while CD10 and cytoplasmic IgM are negative. Surface CD3 and cytoplasmic CD3 are negative. The overall immunophenotype demonstrates predominant myeloid antigen expression with aberrant, incomplete B-lineage marker expression, raising diagnostic complexity and prompting cytogenetic correlation. (B). Extended immunophenotypic characterization highlighting lineage ambiguity. Additional analysis confirms absence of definitive T-cell differentiation (CD3 negative; CD4, CD8 negative) and absence of monocytic differentiation (CD64, CD14 negative; lysozyme negative/low). CD34 expression is variable, without a clearly expanded CD34-bright compartment. (C). B-lineage markers show dim CD19 and cytoplasmic CD79a expression with partial CD22 positivity but lack of CD10 and cytoplasmic IgM, indicating incomplete B-lineage commitment. This constellation of findings underscores the discordance between morphologic features and immunophenotype and supports the need for integrated genetic evaluation for accurate lineage assignment.
Flow Cytometric Findings
Multiparameter flow cytometry was performed on peripheral blood using standardized EuroFlow acute leukemia panels [1, 2]. On CD45 versus side scatter, approximately 90% of nucleated cells formed a blast population with dim CD45 expression and low side scatter. Blasts were positive for: Dim CD19, Dim cytoplasmic CD79a, Partial surface and cytoplasmic CD22, Nuclear TdT, CD34 positivity, Bright CD117, CD13, CD123, CD7, CD4, HLA-DR. Blasts were negative for: Cytoplasmic myeloperoxidase (MPO), CD33, CD14, CD15, CD64, CD10, cytoplasmic IgM, surface and cytoplasmic CD3. The constellation of dim B-lineage markers with absence of CD10 and cytoplasmic IgM, combined with bright CD117 and CD13 expression, created an immunophenotypically ambiguous profile. Initial interpretation favored pro-B ALL versus acute leukemia of ambiguous lineage. However, definitive myeloid lineage assignment by MPO was lacking.
Cytogenetic and Molecular Findings
Conventional karyotyping demonstrated a haploid male clone with monosomy 7 and inv(3)(q21q26)/t(3;3)(q21;q26). These abnormalities are characteristic of MECOM rearrangement. The MECOM (MDS1 and EVI1 complex locus) gene at 3q26.2 encodes transcriptional regulators critical in hematopoietic stem cell self-renewal. Rearrangements juxtapose distal enhancer elements near GATA2 at 3q21 to MECOM at 3q26.2, resulting in aberrant EVI1 overexpression [5-7]. EVI1 functions as a zinc-finger transcription factor promoting proliferation, impairing differentiation, and conferring resistance to apoptosis, thereby driving leukemogenesis [6, 7]. Molecular testing revealed FLT3-ITD and mutations in NRAS, BCOR, and WT1, alterations frequently observed in high-risk AML and often cooperating with MECOM activation [8, 9]. Under current WHO 5th edition and ICC classifications, the presence of inv(3)/t(3;3) defines AML with MECOM rearrangement regardless of blast immunophenotype [3, 4].
Discussion
AML with MECOM rearrangement is a rare but well-characterized, high-risk entity representing approximately 1-2% of AML cases [5]. It is frequently associated with monosomy 7 and a complex karyotype. Clinically, it confers aggressive behavior, relative chemoresistance, and poor overall survival [5, 8]. The underlying mechanism involves enhancer hijacking and deregulated expression of EVI1, leading to impaired hematopoietic differentiation and stem cell expansion [6, 7]. These leukemias often exhibit multilineage dysplasia and may arise de novo or evolve from myelodysplastic syndromes [5, 8]. Immunophenotypically, AML with MECOM rearrangement may show aberrant antigen expression, including lymphoid markers. Aberrant CD7 is common; expression of B-lineage markers is less frequent but documented [8]. In our case, dim CD19 and cytoplasmic CD79a expression within a standardized EuroFlow framework suggested B-lineage assignment. However, several features argued against classic pro-B ALL: absence of CD10 and cytoplasmic IgM, partial CD22 expression, and co-expression of bright CD117 and CD13.
Notably, although B-lineage markers were present, criteria for mixed phenotype acute leukemia were not fulfilled, as definitive myeloid lineage assignment by MPO or monocytic differentiation was absent according to WHO/ICC lineage criteria [3, 4, 10]. Instead, the AML-defining cytogenetic abnormality superseded immunophenotypic ambiguity. This case underscores an important cytopathologic principle: morphology and flow cytometry may suggest lymphoid differentiation, yet genetically defined AML entities must be excluded before establishing lineage. Reliance on dim B-lineage antigen expression without genomic correlation may result in misclassification and inappropriate therapeutic stratification.
Conclusion
AML with MECOM rearrangement involving the MDS1-EVI1 locus at 3q26.2 is a rare, high-risk leukemia driven by EVI1 overexpression and associated with aggressive clinical behavior. This case illustrates that dim B-lineage antigen expression in standardized multiparameter flow cytometry may mimic pro-B ALL, creating a diagnostic pitfall. Integrated morphologic, immunophenotypic, cytogenetic, and molecular evaluation is essential to ensure accurate classification in accordance with contemporary AML taxonomy.
Footnotes
Ethical Considerations: Informed consent was obtained from the patient involved in this study.
Conflict of interests: The authors declare no conflicts of interests.
Funding: The authors received no specific funding for this work.
References
- van Dongen JJM, Lhermitte L, Böttcher S, Almeida J, van der Velden VH, et al. (2012). EuroFlow antibody panels for standardized n-dimensional flow cytometric immunophenotyping of normal, reactive and malignant leukocytes. Leukemia. 26: 1908-1975.
- Kalina T, Flores-Montero J, van der Velden VHJ, Martin-Ayuso M, Böttcher S, et al. (2012). EuroFlow standardization of flow cytometric immunophenotyping. Leukemia. 26: 1908-1975.
- Campo E, Jaffe ES, Cook JR, Quintanilla-Martinez L, Swerdlow SH, et al. (2022). The International Consensus Classification of lymphoid neoplasms. A report. Blood. 140: 1229-1253.
- Arber DA, Orazi A, Hasserjian RP, Orazi A, Mathews V, et al. (2022). The 2022 WHO classification of tumors of hematopoietic and lymphoid tissues: myeloid neoplasms and acute leukemia. Leukemia. 36: 1703-1719.
- Gröschel S, Schlenk RF, Engelmann J, Rockova V, Teleanu V, et al. (2013). Deregulated expression of EVI1 defines a poor prognostic subset of MLL-rearranged acute myeloid leukemias: a study of the German-Austrian Acute Myeloid Leukemia Study Group and the Dutch-Belgian-Swiss HOVON/SAKK Cooperative Group. Clin Oncol. 31: 95-103.
- Lugthart S, Gröschel S, Beverloo HB, Kayser S, Valk PJ, et al. (2010). Clinical, molecular, and prognostic significance of WHO type inv(3)(q21q26.2)/t(3;3)(q21;q26.2) and various other 3q abnormalities in acute myeloid leukemia. Clin Oncol. 28: 3890-3898.
- Kataoka K, Sato T, Yoshimi A, Goyama S, Tsuruta T, et al. (2011). Evi1 is essential for hematopoietic stem cell self-renewal, and its expression marks hematopoietic cells with long-term multilineage repopulating activity. J Exp Med. 208: 2403-2416.
- Rogers HJ, Hsi ED. (2013). Myeloid Neoplasms with inv(3)(q21q26.2) or t(3;3)(q21;q26.2). Surg Pathol Clin. 6: 677-692.
- Papaemmanuil E, Gerstung M, Bullinger L, Gaidzik VI, Paschka P, et al. (2016). Genomic Classification and Prognosis in Acute Myeloid Leukemia. N Engl J Med. 374: 2209-2221.
- Fuda F, Chen W. (2025). Acute Leukemia of Ambiguous Lineage: Diagnosis and Evaluation by Flow Cytometry. Cancers (Basel). 17: 871.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.