Absolok® Haemostatic Evaluation in Colorectal Surgery: A Single-Centre Randomized Open-Label Pilot Clinical Trial
by Vincenzo Tondolo1,2, Gianluca Rizzo1,2*, Giada Livadoti1, Federica Marzi1, Luca Emanuele Amodio1, Giuseppe Quero2,3, Ilaria Simonelli4, and Sergio Alfieri2,3
1UOC Chirurgia Digestiva e del Colon-Retto, Ospedale Isola Tiberina Gemelli Isola, Rome, Italy
2Università Cattolica del Sacro , Rome, Italy
3UOC Chirurgia Digestiva, Fondazione Policlinico Universitario Agostino Gemelli IRCCS, Rome, Italy
4Centro di Ricerca Clinica, Ospedale Isola Tiberina Gemelli Isola, Rome, Italy
*Corresponding Author: Gianluca Rizzo, UOC Chirurgia Digestiva e del Colon-Retto – Ospedale Isola Tiberina Gemelli Isola Via di Ponte Quattro Capi, 39 - 00186 Rome, Italy
Received Date: 06 April 2026
Accepted Date: 10 April 2026
Published Date: 13 April 2026
Citation: Tondolo V, Rizzo G, Livadoti G, Marzi F, Amodio LE, et al. (2026) Absolok® Haemostatic Evaluation in Colorectal Surgery: A Single-Centre Randomized Open-Label Pilot Clinical Trial. J Surg 11: 11605 DOI: https://doi.org/10.29011/2575-9760.011605
Abstract
Key objectives: Major intra and postoperative bleeding remains a critical challenge in colorectal surgery. Primary aim of this pilot randomized study was to compare the safety and haemostatic power of Absolok® and Hem-o-Lok® clips during colorectal surgical procedures.
Methods: A single-centre, open-label, randomized (1:1) pilot trial study was conducted on patients undergone to mini-invasive or open colorectal resection from September 2023 to April 2025. All patients were randomly allocated 1:1 to Absolok® (experimental) or Hem-o-lok® (control) group. A comparative analysis of the haemostatic efficacy for each clipped vessel (graded with Siegel’s scale) intraoperative and short-term postoperative outcomes was performed.
Results: Two hundred patients were enrolled in the study (100 in Absolok® group; 100 in Hem-o-Lok® group). Baseline characteristics were comparable between the groups. Grade 1 haemostasis (no bleeding) was achieved in 100% of the vessels in both cohorts (p:1.0). No significant differences were found in terms of the median operative time (p:0.418) and intraoperative complications, which occurred, overall, in 7 patients (3.5%) (p:0.999). The overall short-term postoperative morbidity rate was 18%, and major postoperative complications were recorded in 4% of the patients. No significant differences in overall and major (grade³3) postoperative morbidity were detected between the 2 groups (p:0.198). No significant differences in time to first postoperative flatus (p:0.708) and in overall postoperative hospital stay (p:0.118) were detected between the 2 groups. No clip-related adverse events occurred.
Conclusions: In colorectal surgery, Absolok® clips seem to provide the same haemostatic effect of Hem-o-Lok® clips.
Keywords: Absolok® Clip; Hem-O-Lok® Clip; Intraoperative Bleeding; Postoperative Morbidity; Randomized Controlled Trial; Surgical Clips
Introduction
Intraoperative bleeding is not a negligible complication of colorectal surgery. Haemorrhage can be significant and difficult to manage with conventional methods of haemostasis. Severe and not rapidly controlled cases of haemorrhage may result in patient fatality [1-5]. Therefore, appropriate preoperative planning, a meticulous surgical technique and the utilization of haemostatic instruments and devices are considered mandatory to minimize the risk of severe blood loss. Haemostatic control is strongly recommended in colorectal surgery, in which postoperative bleeding ranging between 1% and 14% [6-8]. Although postoperative bleeding is rarely fatal, it could represent the beginning of related complications which significantly influence the rate of postoperative morbidity and mortality, the length of postoperative hospital stay and, consequently, the health care costs [9-11]. In this context, the intraoperative achievement of haemostasis represents a fundamental step. Metallic surgical clips are widely used for this purpose but have disadvantages, including a propensity to slip or act as a nidus for infection [12-15]. This led to the more recent introduction of non-metallic clips with a locking mechanism with tactile and auditory feedback, such as Hem-oLok® and Absolok® clips, which represent the two most widely used non-metallic clips. Currently, the two most widely available no-metallic products, are the Hem-o-Lok® and the Absolok® clips. Hem-o-Lok® clips are composed of a nonabsorbable, inert, nonconductive and radiolucent polymer that has no interference during X-ray, computed tomography or magnetic resonance imaging. Hem-o-Lok clips have a lock engagement feature, as well as teeth in the jaws that provide good security. Although the use of Hem-o-Lok® clips close to the upper urinary tract (renal collecting system/ureter) is traditionally considered safe, in urological surgery rare cases of complications due to clip migration into other hollow organs have been reported [16-19]. Furthermore, the appearance of a fibrous reaction around the nonabsorbable clip has been described in rare case reports as a radiological finding that may mimic recurrence or granuloma; Even if the true incidence of this phenomenon is currently unknown, it could represent a serious diagnostic challenge in a case previously operated on for colon cancer with a Hem-o-Lok® clip [20-21]. Absolok® clips are constructed from bioabsorbable polymers that degrade by hydrolysis over a period of approximately [6-7] months and are characterized by a latch-closure mechanism [22] comparable to that adopted for Hem-o-Lok clips. In the literature, only one nonrandomized study comparing these two different types of non-metallic clips has been published, but it was performed in the context of urological surgery [23], where the clips were used as a preloading device to anchor monofilament or multifilament sutures; thus, the clip was not used directly as a tool to close the vessels. No comparative study between Absolok® and Hem-oLok® clips exists in terms of their proper use and in the context of colorectal surgery. The aim of this prospective monocentric randomized study was to perform a comparative analysis of the safety and haemostatic power of Absolok® and Hem-o-Lok® clips during colorectal surgical procedures. The secondary aim was to compare Absolok® and Hem-o-Lok® clips in terms of operative time and intraoperative (other than intraoperative bleeding) and postoperative outcomes.
Materials and Methods
The study was a monocentric, prospective, randomized, openlabel and pilot clinical trial. All patients who underwent colorectal resection for benign or malignant disease at Isola TiberinaGemelli Isola Hospital during the study period were included in this study. The integral version of the protocol is reported. All patients who underwent colorectal resection for benign or malignant disease surgery from September 26, 2023, to April 11, 2025, at Isola Tiberina-Gemelli Isola Hospital were included in the study. The exclusion criteria were as follows: patients aged < 18 years; inability to give informed consent; emergent surgery; previous colorectal surgery; uncontrollable diabetes mellitus that needs continuous intravenously administered insulin; a history of myocardial infarction or unstable angina pectoris within 6 months; cardiac failure; New York Heart Association (NYHA) III degree; anticoagulant therapy; liver cirrhosis; Child‒Pugh class C; active hepatitis; and chronic renal failure requiring haemodialysis. Exclusion criteria were chosen to minimize major confounding factors for bleeding and postoperative morbidity in a pilot trial. Conditions associated with markedly increased haemorrhagic risk or impaired healing (e.g. anticoagulant therapy, advanced liver diseases, haemodialysis, severe cardiac failure) were excluded to improve internal validity and patient safety. Patients enrolled in the study, without exclusion criteria, were randomized into two groups (1:1 ratio): the experimental group, in which the Absolok® clip was used, and the control group, in which the Hem-o-Lok® clip was used. The allocation of the subjects to either the experimental or the control arm used a random sorting randomization algorithm. The allocation sequence was generated by the software PASS 2019. The sub Investigator (VT) and two members of the team (GR and GL) controlled the random allocation sequence. This study was designed as a pilot randomized trial because no comparative data between the two types of clips were available in colorectal surgery, and the expected rate of clinically relevant clip-related bleeding is low. The main objectives of the study were to evaluate the feasibility, safety and estimation of event rates to perform a future adequately powered non-inferiority trial. Therefore, a formal calculation of the sample size was not performed a priori: we planned to enrol a total of 200 consecutive colorectal resections as a pragmatic sample to obtain precise estimates of bleeding and complication rates and their confidence interval for future power calculations. All patients who underwent colorectal resections (categorized as right hemicolectomies, left hemicolectomies and rectal resections) were included in the study, independent of the type of approach to be performed (open, laparoscopic, or robot assisted). All surgical procedures were performed by the same group of colorectal surgeons (Unit of Digestive and Colorectal Unit at Isola Tiberina Gemelli Isola Hospital) experienced in minimally invasive surgery. All procedures were standardized, adopting, in all cases, the same steps, independent of group assignment. The details of surgical procedures performed are reported. During a right hemicolectomy 4 major vessels were clipped: ileo-colic vein and artery, right branch of middle colic vein and artery. During a left hemicolectomy and during rectal resection 2 major vessels were clipped: inferior mesenteric artery and vein.
The primary endpoint of the study was haemostatic performance at the transection site for each clipped vessel, assessed immediately after vessels division using Siegel scale [24]. The comparative analysis between Absolok® and Hem-o-Lok® in terms of haemostatic efficiency was performed evaluating the percentage of adverse bleeding events for each vessel clipped and dissected, according to Siegel’s score, which classified the entity of bleeding into four categories [24] as follows:
- Grade 1: no bleeding at the transection site.
- Grade 2: minor bleeding at the transection site; no intervention was needed.
- Grade 3: minor bleeding at transection site, mild intervention needed
- Grade 4: significant bleeding requiring major intervention such as extensive coagulation, ligation or additional haemostatic measures.
The secondary endpoint of the study was the evaluation of whether the clip type could influence the occurrence of intraoperative and postoperative complications. For this purpose, the following outcome variables were evaluated:
- Operative time (min)
- Intraoperative complications
- Time (postoperative day) to flatus
- Postoperative complications within 30 days of surgery were graded according to Clavien’s classification [25].
- Major postoperative complications (if the grade was ≥3 according to Clavien’s classification [25]).
- Length of hospital stay (days)
- Postoperative bleeding within 30 days (any clinical bleeding requiring transfusion, endoscopic/radiologic/surgical intervention or causing haemodynamic instability
- Reoperation within 30 days
- Mortality within 30 days from surgery
Moreover, all adverse events related to the use of each type of clip observed during the study were recorded. Adverse device events were defined as any device malfunction or deficiency, including failure to lock, clip breakage, clip dislodgement/slippage, need for additional clips due to inadequate vessel control, or any intraoperative/postoperative complication judged as directly related to the clip. This trial was approved by the Ethical Committee of Lazio 3 (ID 6090) and was funded by Ethicon (UK). Ethicon (UK) had no role in study design, data collection, analysis, manuscript writing and decision to submit for publication. All the authors declared no conflicts of interest. Informed consent was obtained from all individual participants included in the study. This study is reported in accordance with the CONSORT 2010 guidelines. The trial was registered on the ISRCTN Registry (The UK’s Clinical Study Registry) as ISRCTN79285407 on 05 February 2026 (retrospectively registered). All the variables are presented as descriptive statistics. In depth, the data are reported as absolute and percentage frequencies for qualitative variables which include: sex (male vs female), ASA class, CCI category, indication for surgery (benign vs malignant), surgical approach (open vs miniinvasive), conversion rate, intraoperative complications (Y vs N), postoperative complications (Y vs N, both overall than major), anastomotic leakage (Y vs N), reoperation (Y vs N) and adverse events (Y vs N). Quantitative data distribution was assessed using the Shapiro‒Wilk test. Hence, the data are expressed as the mean, standard deviation (SD) or median and interquartile range (IQR: 25th–75th percentile), as appropriate. For the primary endpoint, the difference between Absolok® and Hem-o-Lok® in terms of the achievement rate of Grade ≤3 haemostasis for each vessel clipped and dissected (dichotomous variable) was assessed by Fisher’s exact test. The single grading scale was further reported. In the case of qualitative variables, between-group differences were assessed by Fisher’s exact test and the Chi-square test, with Yates correction, as appropriate. Quantitative data were assessed by either Student’s t test or the nonparametric Mann‒Whitney U test, as appropriate. With respect to the secondary endpoints, the potential associations between the type of clip employed and the intraoperative and postoperative courses were computed by using the same test, as previously mentioned. To evaluate differences between the two groups in terms of operation time, logarithmic transformation was applied to reduce the variability of the data distribution, and a t test was applied. A p value <0.05 was considered to indicate statistical significance. All analyses were performed by using R version 4.4.3. The anonymized dataset that supports the findings of this study is available from the corresponding author and principal investigator and sub investigator on reasonable request.
Results
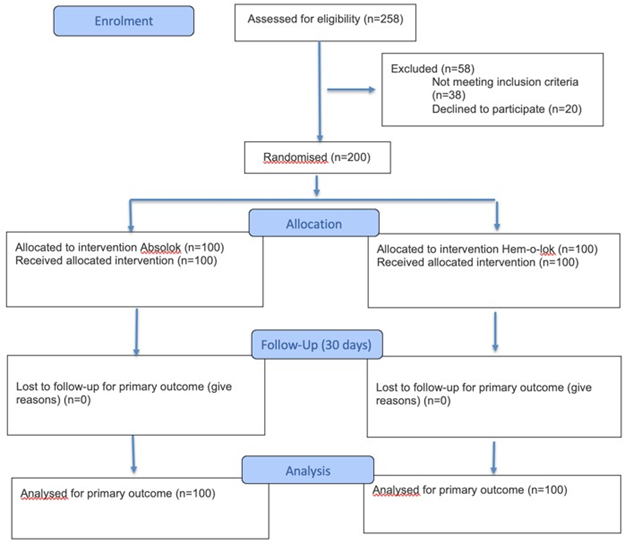
Figure 1: Flow diagram of the progress through the phases of a randomised trial of two groups (that is,enrolment, intervention allocation, follow-up, and data analysis).
Fifty-two patients (26%) underwent colorectal resection for benign disease (complicated diverticular disease or inflammatory bowel disease). All surgical procedures (100%) were performed with a minimally invasive approach, and the conversion (minimally invasive to open) rate was 2% (4 patients). In particular, were performed 59 right hemicolectomies (28 in Absolok® group vs 31 in Hem-o-Lok® group), 87 left hemicolectomies (45 in Absolok® group vs 42 in Hem-o-Lok® group) and 54 rectal resections (27 in Absolok® group vs 27 in Hem-o-Lok® group). Overall, 256 vessels were clipped by Absolok® clips, and 262 vessels were clipped by Hem-o-Lok® clips. No differences existed between the 2 groups regarding the indications for surgery (p=0.580), type of surgical approach (p=0.999) or conversion rate (p=0.620) (Table 1). With respect to the primary endpoint of the study, 100% of patients in both groups achieved Grade 1 or no bleeding at the transection site (Table 2). The median operative time was 218 minutes (IQR: 178–264.75 minutes) and did not differ between the two groups (219 min, IQR: 127–430 min; in the Hem-o-Lok® group vs. 216 min, IQR: 120–353 min; in the Absolok® group; p=0.418). In 7 patients (3.5%), intraoperative complications occurred: in 3 patients, laceration of the colonic serosa occurred with secondary laparoscopic closure of the defect; in 2 patients massive bleeding occurred: 1 of these (Absolok® group) patients was managed with laparoscopic haemostasis and the other (Hemolok® group), due to spleen damage, was managed after conversion in open surgery. In both cases, the bleeding was not attributable to clip malfunction, slippage or transection of a major vessel inadequately secured by the study device. In 1 patient, a bladder wall defect was accidentally perforated and was sutured laparoscopically; and in 1 case, after left hemicolectomy, right colon mobilization and distal transverse colon resection were necessary because of ischaemia of the residual colon. No significant differences in the rate of intraoperative complications were detected between the two groups (Table 2).
|
Hem-o-lok® |
Absolok® |
p value |
|
|
N=100 |
N=100 |
||
|
Age, median[25th-75th percentile] |
71.0 [37.9-86.6] |
69.0 [39.4-86.5] |
0.583a |
|
Sex: |
0.888b |
||
|
M, n (%) |
50 (50.0%) |
52 (52.0%) |
|
|
F, n (%) |
50 (50.0%) |
48 (48.0%) |
|
|
BMI, median[25th-75th percentile] |
25.0 [21.0-31.0] |
25.0 [19.0-34.6] |
0.836a |
|
Comorbidity: |
0.165b |
||
|
No, n (%) |
25 (25.0%) |
35 (35.0%) |
|
|
Yes, n (%) |
75 (75.0%) |
65 (65.0%) |
|
|
CCI, median[25th-75th percentile] |
4.00 [1.00-9.52] |
4.00 [1.00-11.0] |
0.827a |
|
ASA: |
0.085b |
||
|
≤2, n (%) |
84 (84.0%) |
73 (73.0%) |
|
|
>2, n (%) |
16 (16.0%) |
27 (27.0%) |
|
|
Indications for colorectal surgery: |
0.63 |
||
|
Malignant disease |
72 (72.0%) |
76 (76.0%) |
|
|
Bening disease |
28 (28.0%) |
24 (24.0%) |
|
|
Site of colorectal cancer |
0.58 |
||
|
Right Colon |
26 (26.0%) |
24 (24.0%) |
|
|
Left Colon |
19 (19.0%) |
26 (26.0%) |
|
|
Rectum |
27 (27.0%) |
26 (26.0%) |
|
|
NAD CRT (only for rectal cancer) |
15 (15.0%) |
16 (16.0%) |
0.87 |
|
Minimally Invasive Surgery |
100 (100%) |
100 (100%) |
0.999 |
|
Types of Surgical Procedures |
100 |
100 |
|
|
RH |
|||
|
LH |
31 |
28 |
|
|
RAR |
42 |
45 |
0.88 |
|
27 |
27 |
||
|
N. of clipped vessels |
262 |
256 |
- |
|
Conversion Rate |
1 (1.0%) |
3 (3.0%) |
0.62 |
a: non-parametric Mann Whitney U test; b: non-parametric Chi-square test.
Table 1: Baseline characteristics
|
Hem-o-lok® |
Absolok® |
p value |
|
|
N=100 |
N=100 |
||
|
N. of clipped vessels |
262 |
256 |
- |
|
Adverse Bleeding Events |
0 (0%) |
0 (0%) |
0.999 |
|
Adverse Device Events |
0 (0%) |
0 (0%) |
0.999 |
|
Operative time, minmedian [25th75th percentile] |
219 [127-430] |
216 [120-353] |
0.418a |
|
Intra-operative complications: |
0.999b |
||
|
No, n (%) |
97 (97.0%) |
96 (96.0%) |
|
|
Yes, n (%) |
3 (3.00%) |
4 (4.00%) |
|
|
First Flatus Time, daysmedian [25th- 75th percentile] |
2 [1-3] |
2 [1-4] |
0.708c |
|
Postoperative complications: |
0.198b |
||
|
No, n (%) |
78 (78.0%) |
86 (86.0%) |
|
|
Yes, n (%) |
22 (22.0%) |
14 (14.0%) |
|
|
Major postoperative complications (grade 3 or major according to Clavien class) |
5 (5%) |
3 (3%) |
0.721 |
|
Anastomotic Leak |
0.999 |
||
|
No, n (%) |
91/93 (97.8%) |
86/88 (97.7%) |
|
|
Yes, n (%) |
2/93 (2.15%) |
2/88 (2.27%) |
|
|
Postoperative bleeding within 30 days |
0 (0%) |
1 (1%) |
1 |
|
Reoperation within 30 days |
0.716 |
||
|
No, n (%) |
93 (96.9%) |
88 (95.7%) |
|
|
Yes, n (%) |
3 (3.12%) |
4 (4.35%) |
|
|
Length of hospital stay, daysmedian [25th-75th percentile] |
5.00 [4.00-12.5] |
5.00 [4.00-24.7] |
0.118c |
a: T test applied to logarithmic transformation of data; b: non-parametric Chi-square test; c: non-parametric Mann Whitney U test.
Table 2: Primary and secondary endpoints.
Overall, 36 (18%) patients experienced complications within 30 days of surgery, but only 8 (4%) patients experienced complications with a grade ≥3 according to the Clavien classification. The 30-day overall postoperative complication and major postoperative complication rates did not differ between the Absolok® group and the Hem-o-Lok® group (14% vs. 22%; p=0.198). The overall rate of anastomotic leakage was 2.1% (2.3% in the Absolok® group vs. 2.2% in the Hem-o-Lok® group; p=0.999), and the rate of reoperation within 30 days to treat postoperative complications was 4% (4.3% in the Absolok® group vs. 3.1% in the Hem-o-Lok® group; p=0.716). The overall median time to first flatus was 2 (IQR: 1–3) days and did not differ between the Absolok® (median=2 days; IQR: 1–4 days) group and the Hem-o-Lok® group (median=2 days; IQR: 1–3 days; p=0.708). The median postoperative length of stay (overall: 5 days; IQR: 5–6 days) did not differ between the two groups (Absolok® group: 5 days; IQR: 4–24.7 days vs. the Hem-o-Lok® group: 5 days; IQR: 4–12.5 days; p=0.118). None of the analysed patients in the study died within 30 days of surgery (postoperative mortality rate: 0%). Fourteen patients (7%) experienced adverse events, with an equal distribution between the two groups (7% in both the Absolok® group and the Hem-oLok® group; p=0.999) (Table 2). Surgeons noted no differences in the handling of the clip applicators, whether for open or laparoscopic techniques; no specific application issues were noted between the two clips.
Discussion
Bleeding, both intra- and postoperative, remains among the most feared complications of colorectal procedures and its incidence ranges between 1% and 14% [6-8]. While the incidence of lifethreatening haemorrhage is relatively low, even moderate bleeding can significantly compromise the postoperative outcomes [1,2]. Therefore, having disposable, safe and efficient vascular control tools, especially in high-risk patients, represents a crucial point of the surgical procedure. In recent years, Hem-o-Lok clips, composed of a nonabsorbable, inert, nonconductive and radiolucent polymer, have been largely used in minimally invasive abdominal surgery both for the closure of major vessels than for the closure of other tubular structures, such as the cystic duct or the appendicular stump [26-30]. The wide diffusion of the sliding-clip technique in minimally invasive procedures seems to be due to the benefits of knotless suturing and to the best application and distribution of the tension of the sutures on the tissues, avoiding the slippage phenomenon described with metallic clips. The sliding-clip technique for vessel closure using Hem-o-Lok® was demonstrated to reduce the operative time without affecting the postoperative outcome or length of stay [31]. However, several adverse events related to Hem-o-Lok® have been described. The nonabsorbable nature of Hem-o-Lok® means the clips remain at the level of the tumour bed, generating imaging artefacts or difficult interpretation [20,21]. Moreover, the Hem-o-Lok® clips may migrate into the abdomen. In the urological setting, ureteric or pelviureteric junction obstruction, urinary infection and stone formation have been described after Hem-o-Lok clip migration [18,19,32,33] . Moreover, in some series, several complications due to Hemo-Lok clip erosion have been described [16,17]. Absolok® clips are made from polydioxanone (PDS), a synthetic biodegradable polymer that offers excellent flexibility, biocompatibility, and gradual absorption within approximately 6–9 months in vivo.
The Absolok® Polydioxanone (PDS) absorbable clips undergo biodegradation via hydrolysis, and the degradation metabolites are excreted mainly in urine (estimated retention period: 90 days) [34]. Several clinical studies have analysed the role of absorbable clips in general surgery, urology and gynaecology. In 1985, Clarke-Person et al evaluated the role of absorbable vascular ligating clips made of polydioxanone polymer in 44 patients who underwent abdominal and pelvic surgery. The authors reported that absorbable clips were superior to metallic clips in the applier when positioning to ligate vessels; moreover, polydioxanone clips were absorbed within 210 days, eliminating the risk of affecting imaging [35]. The safety of the use of absorbable clips was also demonstrated in the prospective randomized controlled study of Hawasli et al, which demonstrated that the absorbable clips were as effective as metal clips in providing haemostasis and securing the cystic duct stump [36]. The safety of Absolok® clips in general surgery was also emphasized in a large personal series from Veronese et al which included a group of major vessel ligations by clips during colorectal surgery. In this series, no biliary outflow or haemorrhages due to dislocation of absorbable clips were reported [37]. Compared with metal clips, Absolok® clips seem to support greater weight and withstand higher intravascular pressure, reducing the risk of arterial or venous slippage [38]. A comparative analysis between Absolok® and Hem-o-Lok® clips was performed in a urological setting by Rossanese et al, who evaluated the haemostatic power of Absolok® clips compared with that of Hemo-Lok® during renorraphy [23]. The study compared 57 patients in whom Absolok® clips were used with 40 patients in whom Hem-o-Lok® clips were used. No differences in intraoperative or postoperative outcomes were detected between the two types of clips; moreover, on abdominal CT performed 3 months after surgery, no Absolok® clips were shown on CT scans, highlighting one of the main characteristics of Absolok® clips [23]. This aspect is particularly relevant in surgical oncology, where postoperative imaging (especially CT) plays a key role in monitoring recurrence. Permanent clips can cause beam-hardening artefacts, confuse radiologists, or mimic recurrence. Therefore, absorbable devices could eliminate this source of ambiguity, improving diagnostic confidence in surveillance imaging. Moreover, although no clipspecific infectious complications were observed in either group, the theoretical advantage of eliminating a permanent foreign body cannot be overlooked. Permanent devices, even if inert, can support biofilm formation or become a nidus in immunocompromised or irradiated patients [22]. In contrast, polydioxanone materials, once hydrolysed, are excreted as inert byproducts via renal and hepatic metabolism [34]. In fact, no studies have performed a comparative analysis in the context of digestive surgery or, in particular, during colorectal procedures. Considering the potential advantages of absorbable clips such as Absolok® and the absence of a comparative study in the context of colorectal surgery, we performed this single-centre, open-label, randomized pilot trial with the aim of comparing the haemostatic power of Absolok® clips with that of Hem-o-Lok® clips in the context of colorectal surgery. In addition to the evidence recorded in other surgical settings, in our large series of colorectal procedures, no differences were found between Absolok® and Hem-o-Lok® clips in terms of the safety of blood vessel closure, both for malignant procedures and for benign indications for colorectal procedures. Moreover, no differences between the two groups were found in terms of intraoperative or early (within 30 days) postoperative outcomes, as no differences in the handling of the clip applicators or specific application issues were noted between the two clips. Our protocol design did not include CT abdominal scans at 3 months after the colorectal surgical procedures, which could be considered a limitation of the study, but the analysis of Absolok® absorption was performed only in a previously published study [22].
Conclusions
This pilot randomized trial demonstrates that Absolok® clips can be safely and feasibly used for vascular control in minimally invasive colorectal surgery, with haemostatic performance comparable to standard polymer clips, such as Hem-o-Lok, without safety concerns. Future work should explore their utility in higher-risk subgroups (e.g., inflammatory bowel disease, reoperations, and irradiated pelvis) and long-term endpoints such as pain, adhesionrelated obstruction, or imaging clarity. A larger multicentre phase III trial is warranted. A formal cost effectiveness evaluation was not performed in the present pilot study and costs may vary across healthcare systems and procurement contracts. Future analyses should quantify potential benefits of absorbable clips in terms of downstream resource utilization against the device acquisition cost.
Declarations
Ethics approval and consent to participate: This trial was approved by the Ethical Committee of Lazio 3 (ID 6090). The study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all individual participants included in the study.
Consent for publication: Consent for publication was obtained from all individual participants included in the study.
Availability of data and materials: The datasets used and/ or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests: All the authors declare that they have no competing interests.
Funding: This pilot randomized trial was funded by Ethicon (UK). Disclosures: All the authors declare no disclosures.
Author contributions: Conceptualization: Sergio Alfieri, Vincenzo Tondolo, Giuseppe Quero; Methodology: Giuseppe Quero, Gianluca Rizzo, Ilaria Simonelli; Formal analysis and investigation: Gianluca Rizzo, Ilaria Simonelli, Luca Emanuele Amodio, Federica Marzi, Giada Livadoti; Writing - original draft preparation: Gianluca Rizzo, Luca Emanuele Amodio, Federica Marzi, Giada Livadoti; Writing - review and editing: Sergio Alfieri, Vincenzo Tondolo, Giuseppe Quero; Funding acquisition: Sergio Alfieri, Vincenzo Tondolo; Resources: Gianluca Rizzo, Sergio Alfieri, Vincenzo Tondolo; Supervision: Sergio Alfieri, Vincenzo Tondolo.
Acknowledgements: not applicable
Trial Registration: ISRCTN Registry (The UK’s Clinical Study Registry), ISRCTN79285407, 05 February 2026 (Retrospectively registered).
References
- Stolfi VM, Milsom JW, Lavery IC, Oakley JR, Church JM, et al. (1992) Newly designed occluder pin for presacral hemorrhage. Dis Colon Rectum 35: 166-169.
- Germanos S, Bolanis I, Saedon M, Baratsis S (2010) Control of presacral venous bleeding during rectal surgery. Am J Surg 200: e33- e35.
- Baqué P, Karimdjee B, Iannelli A, Benizri E, Rahili A, et al. (2004) Anatomy of the presacral venous plexus: implications for rectal surgery. Surg Radiol Anat 26: 355-358.
- Hill AD, Menzies-Gow N, Darzi A (1994) Methods of controlling presacral bleeding. J Am Coll Surg 178: 183-184.
- Wang QY, Shi WJ, Zhao YR, Zhou WQ, He ZR (1985) New concepts in severe presacral hemorrhage during proctectomy. Arch Surg 120: 1013-1020.
- Golda T, Zerpa C, Kreisler E, Trenti L, Biondo S (2013) Incidence and management of anastomotic bleeding after ileocolic anastomosis. Colorectal Dis 15: 1301-1308.
- McCloskey CA, Wilson MA, Hughes SJ, Eid GM (2007) Laparoscopic colorectal surgery is safe in the high-risk patient: a NSQIP riskadjusted analysis. Surgery 142: 594-597.
- Benoist S, Panis Y, Alves A, Valleur P (2000) Impact of obesity on surgical outcomes after colorectal resection. Am J Surg 179: 275-281.
- Tartter PI (1988) Blood transfusion and infectious complications following colorectal cancer surgery. Br J Surg 75: 789-792.
- Morton J, Anastassopoulos KP, Patel ST, Lerner JH, Ryan KJ, et al. (2010) Frequency and outcomes of blood products transfusion across procedures and clinical conditions warranting inpatient care: an analysis of the 2004 healthcare cost and utilization project nationwide inpatient sample database. Am J Med Qual 25: 289-296.
- Callum JL, Waters JH, Shaz BH, Sloan SR, Murphy MF (2014) The AABB recommendations for the Choosing Wisely campaign of the American Board of Internal Medicine. Transfusion 54: 2344-2352.
- Arnaud JP, Bergamaschi R (1993) Migration and slipping of metal clips after celioscopic cholecystectomy. Surg Laparosc Endosc 3: 487-488.
- Ghavidel A (2015) Migration of clips after laparoscopic cholecystectomy; a case report and literature review. Middle East J Dig Dis 7: 45-49.
- Madhavan S, Pandey A, Masood S, Kumar S, Chauhan S, et al. (2021) Polymeric locking clips [Hem-o-lok] versus Metallic clips in elective Laparoscopic Cholecystectomy: A Retrospective study of 1496 patients. Pol Przegl Chir 93: 1-5.
- Sheffer D, Gal O, Ovadia B, Kopelman Y (2020) Cholangitis caused by surgical clip migration into the common bile duct: a rare complication of a daily practice. BMJ Case Rep 13: e231201.
- Turini GA 3rd, Brito JM 3rd, Leone AR, Golijanin D, Miller EB, et al. (2016) Intravesical Hemostatic Clip Migration After Robotic Prostatectomy: Case Series and Review of the Literature. J Laparoendosc Adv Surg Tech A 26: 710-712.
- Cormio L, Massenio P, Lucarelli G, Di Fino G, Selvaggio O, et al. (2014) Hem-o-lok clip: a neglected cause of severe bladder neck contracture and consequent urinary incontinence after robot-assisted laparoscopic radical prostatectomy. BMC Urol 14: 21.
- Shrivastava P, Nayak B, Singh P (2017) Migrated Hem-o-Lok clips in the ureter: a rare cause of recurrent urinary tract infection. BMJ Case Rep bcr 2016219143.
- Fiard G, Peneau M, Soulimane B (2014) Migration massive de clips Hem-o-lok® après néphrectomie partielle ouverte : cas clinique et revue de la littérature [Massive migration of Hem-o-lok® clips following open partial nephrectomy: a case report and literature review]. Prog Urol 24: 616-619.
- Koba M, Kawai K, Tokuyama S, Takahashi Y, Matsui Y, et al. (2024) A Case of Scarred Tissue Caused by a Hem-o-Lok Clip Used during Descending Colon Cancer Resection. Gan To Kagaku Ryoho 51: 1785-1787.
- Fan X, Zan R, Bian L, Chen Y (2021) Hem-O-Lok clips may lead to misdiagnosis in postoperative follow-up of tumor patients: A case report. Asian J Surg 44: 1123-1124.
- Darzi A, Soin B, Coleman J, Lirici NM, Angelini L (1997) Initial experience with an absorbable laparoscopic ligation clip. Br J Surg 84: 974-976.
- Rossanese M, Crestani A, Giannarini G, Calandriello M, Alario G, et al. (2020) Absolok® versus Hem-o-Lok® clips for renorrhaphy during partial nephrectomy for parenchymal renal tumors. Minerva Urol Nefrol 72: 91-98.
- Siegel J, Cummings J, Clymer J (2014) Reproducible, Repeatable and Clinically-relevant Hemostasis Scoring. Journal of Advances in Medical and Pharmaceutical Sciences 1: 30-39
- Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240: 205-213.
- Casale P, Pomara G, Simone M, Casarosa C, Fontana L, et al. (2007) Hem-o-lok clips to control both the artery and the vein during laparoscopic nephrectomy: personal experience and review of the literature. J Endourol 21: 915-918.
- Lee JE, Kim KG, Lee DO, Seo SS, Kang S, et al. (2015) Ligation of uterine vessels in total laparoscopic hysterectomy using Hem-o-lok clips. Taiwan J Obstet Gynecol 54: 8-12.
- Kihara Y, Takeda Y, Ohmura Y, Katsura Y, Shinke G, et al. (2021) Migration of non-absorbable polymer clips in hepato-biliary-pancreatic surgery: a report of four cases. Surg Case Rep 7: 183.
- Pardolesi A, Park B, Petrella F, Borri A, Gasparri R, et al. (2012) Robotic anatomic segmentectomy of the lung: technical aspects and initial results. Ann Thorac Surg 94: 929-934.
- Nota CL, Molenaar IQ, Borel Rinkes IH, Hagendoorn J (2020) Robotic liver resection of segment 7: A step-by-step description of the technique. Surg Oncol 34: 206-207.
- Benway BM, Wang AJ, Cabello JM, Bhayani SB (2009) Robotic partial nephrectomy with sliding-clip renorrhaphy: technique and outcomes. Eur Urol 55: 592-599.
- Park KS, Sim YJ, Jung H (2013) Migration of a Hem-o-Lok Clip to the Ureter Following Laparoscopic Partial Nephrectomy Presenting With Lower Urinary Tract Symptoms. Int Neurourol J 17: 90-92.
- Lee Z, Reilly CE, Moore BW, Mydlo JH, Lee DI, et al. (2014) Stone formation from nonabsorbable clip migration into the collecting system after robot-assisted partial nephrectomy. Case Rep Urol 397427.
- Boland ED, Coleman BD, Barnes CP, Simpson DG, Wnek GE, et al. (2005) Electrospinning polydioxanone for biomedical applications. Acta Biomater 1: 115-123.
- Clarke-Pearson DL, Creasman WT (1985) A clinical evaluation of absorbable polydioxanone ligating clips in abdominal and pelvic operations. Surg Gynecol Obstet 161: 250-252.
- Hawasli A (1994) The use of absorbable clips in laparoscopic cholecystectomy. J Laparoendosc Surg 4: 333-338.
- Veronese E, Elio A, Residori C, Frigo F, Salvato S, et al. (1999) Nostra esperienza sull’utilizzo delle clips riassorbibili in polidiossanone Absolok in chirurgia laparoscopica [Our experience with the use of Absolok polydioxanone resorbable clips in laparoscopic surgery]. Minerva Chir 54: 195-197.
- Hsu TC (2006) Comparison of holding power of metal and absorbable hemostatic clips. Am J Surg 191: 68-71.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.