A Rare Case of Intramyocardial Abscess without Infective Endocarditis: Imaging Findings
by Yildiz Sengul*, William Fenner Griffin, Todd Richard Hazelton
Department of Radiology, University of North Carolina, 101 Manning Dr, Chapel Hill, NC 27514, USA
*Corresponding author: Yildiz Sengul, Clinical instructor, Department of Radiology, University of North Carolina, 101 Manning Dr, Chapel Hill, NC 27514, USA
Received Date: 02 March 2026
Accepted Date: 06 March 2026
Published Date: 09 March 2026
Citation: Sengul Y, Griffin WF, Hazelton TR (2026) A Rare Case of Intramyocardial Abscess without Infective Endocarditis: Imaging Findings. Ann Case Report. 11: 2554. DOI: https://doi.org/10.29011/2574-7754.102554
Abstract
Myocardial abscess is a rare but potentially life-threatening infection of the myocardium, typically resulting from infective endocarditis or septic embolism. This condition can lead to tissue necrosis myocardial rupture, and disturbances in cardiac conduction, highlighting the importance of timely and accurate diagnosis for patient outcomes. While myocardial abscesses related to infective endocarditis have been well-documented in the literature, it is increasingly uncommon for these abscesses to develop in the context of a disseminated infection.
In this case report, we present a myocardial abscess located in the interventricular septum, extending into the right ventricle, due to a disseminated fungal infection caused by Cunninghamella, which is a rare form of mucormycosis. Diagnosing a myocardial abscess can be challenging and often necessitates comprehensive imaging studies. We highlight the complementary roles of chest CT, cardiac MRI, and PET/CT in establishing diagnosis, assessing the extent of the disease, and guiding clinical management.
Keywords: Myocardial abscess; Systemic fungal infection; Chest CT; Cardiac MRI; Immunocompromised host; PET/CT
Introduction
Myocardial abscess is an uncommon but serious suppurative infection of the heart muscle, often resulting from infective endocarditis, sepsis, or invasive cardiovascular procedures, most commonly bacterial [1, 2]. Fungal myocardial abscesses are exceptionally rare and usually occur in immunocompromised patients [3]. Early diagnosis is challenging due to non-specific symptoms including fever, bacteremia, or new conduction disturbances that may be leading to delays in diagnosis and limited echocardiographic sensitivity for intramyocardial lesions. Advanced cross-sectional modalities, such as cardiac magnetic resonance imaging (CMRI) and metabolic imaging with FDG PET/CT, have emerged as valuable tools for characterization, determination of extent, and assessment of associated complications [4]. Prompt recognition of myocardial abscesses is essential due to the risks of rupture, fistula formation, valvular destruction, and potentially life-threatening arrhythmia and myocardial infarction [5-7]. In this case report, we present a myocardial abscess diagnosed using a multimodal imaging approach. This case highlights the complementary roles of both structural and metabolic imaging in establishing an accurate diagnosis and guiding appropriate management.
Case Report
A 50-year-old male with diagnosed T-cell acute lymphoblastic leukemia (T-ALL) on Hyper-CVAD (hyper-fractionated cyclophosphamide, vincristine, doxorubicin (Adriamycin), and dexamethasone) chemotherapy cycle 2, presented with fever, chills, and malaise. On admission, he was neutropenic (ANC <100/µL) and hemodynamically stable. Empiric broad-spectrum antibiotics were initiated for febrile neutropenia. Chest CT revealed a 1.2 cm cavitary nodule in the right upper lobe (Figure 1A) and ill-defined, lobulated hypoattenuating lesion measuring approximately 7×3 cm within the interventricular septum (Figure 1B), raising suspicion for fungal infection in the setting of profound neutropenia. During hospitalization, the patient developed a new-onset tremor. Brain MRI demonstrated a 3.4 cm mass in the left frontal lobe with surrounding vasogenic edema, showing ring-like enhancement on post-contrast sequences, consistent with a possible fungal abscess or metastatic septic embolus. Stereotactic brain biopsy pathology confirmed fungal infection (broad, aseptate hyphae morphologically compatible with Mucorales). Peripheral blood cfDNA testing identified Cunninghamella species (a member of the Mucorales), supporting the diagnosis of invasive cerebral mucormycosis (Cunninghamella). A PET/CT scan, conducted to assess disseminated fungal infection, identified a hypermetabolic fluid collection along the inferior wall of the heart, abnormal FDG uptake in all four cardiac chambers, and a small pericardial effusion (Figure 1C). These findings raised concerns for cardiac involvement, suggesting a possible myocardial abscess and associated inflammatory activity. Transthoracic echocardiogram reported anthracycline-induced cardiotoxicity with reduced ejection fraction EF: 35%, no vegetations visualized on the valves. A cardiac MRI (Siemens 1.5T) was performed using stacked shortaxis and four-chamber cine steady-state free precession (SSFP) sequences to assess morphology, function, and wall motion. Tissue characterization included pre- and post-contrast T1-weighted imaging, first-pass perfusion, and late gadolinium enhancement (10 minutes post-contrast) with phase-sensitive inversion recovery (PSIR), which provide improved nulling of normal myocardium and better delineation of abnormal tissue such as inflammation, necrosis, or abscess. Short-axis cine SSFP imaging demonstrated a 7×3 cm hyperintense intramyocardial lesion in the mid-toapical interventricular septum extending into the right ventricle (Figure 1D), with associated myocardial edema, regional hypokinesis, and reduced systolic function (LVEF: 30%). There was no evidence of infective endocarditis involving the native heart valves, with no valvular vegetations, abnormal thickening, or perivalvular extension identified (Figure 1E). First-pass perfusion revealed a non-enhancing, multilobulated lesion without viable microvasculature in the corresponding region (Figure 1F). Late gadolinium enhancement with PSIR demonstrated a septal intramyocardial mass with a central non-enhancing core consistent with abscess formation, surrounded by a rim of peripheral enhancement and mid-wall adjacent myocardial uptake, indicating associated inflammatory involvement (Figure 1G). The case was discussed at a multidisciplinary conference attended by specialists in radiology, cardiothoracic surgery, and interventional cardiology. Due to the lack of a safe surgical or percutaneous method to evacuate the intramyocardial abscess, the team decided to pursue nonoperative management. This approach involves targeted antifungal therapy and regular imaging surveillance to closely monitor the condition. A cardiac MRI conducted at a fourweek interval showed a reduction in the size of the interventricular myocardial abscess, which now measures 3.5x1.5 cm compared to its previous dimensions. Cine SSFP sequences indicated a decrease in perilesional edema, while post-contrast delayed enhancement imaging revealed a reduction in both the thickness and intensity of the surrounding rim (Figure 1H). Additionally, there was a slight improvement in left ventricular systolic function, with a left ventricular ejection fraction (LVEF) of 35% and no new regional wall motion abnormalities detected. It was recommended to continue antifungal therapy and perform imaging surveillance at short intervals. Despite interval radiologic improvement of the interventricular myocardial abscess, the patient subsequently developed superimposed Pseudomonas aeruginosa bacteremia complicated by septic shock and ultimately succumbed to multiorgan failure.

Figure 1A: 50-year-old male with history of T-ALL and febrile neutropenia. A, Axial chest CT (lung window) shows a 1.2 cm cavitary nodule in the right upper lobe (arrow).

Figure 1B: 50-year-old male with history of T-ALL and febrile neutropenia. B, Axial contrast-enhanced chest CT (mediastinal window) demonstrates a hypoattenuating lesion (arrow) with indistinct margins located in the interventricular septum, measuring approximately 7.0 × 3.0 cm.

Figure 1C: 50-year-old male with history of T-ALL and febrile neutropenia. C, Axial PET/CT image demonstrates a peripherally hypermetabolic intracardiac fluid collection consistent with abscess, localized to the inferior wall and extending into the interventricular septum (arrow). The lesion shows intense peripheral FDG uptake with a centrally photopenic necrotic core (asterisk).

Figure 1D: 50-year-old male with history of T-ALL and febrile neutropenia. D, Short-axis steady-state free precession (SSFP) cardiac MR imaging demonstrates 7x3 cm hyperintense intramyocardial lesion in the mid-to-apical interventricular septum with extension into the right ventricle (arrows), associated with adjacent myocardial edema (asterisk). LV: Left ventricle, RV: Right ventricle.
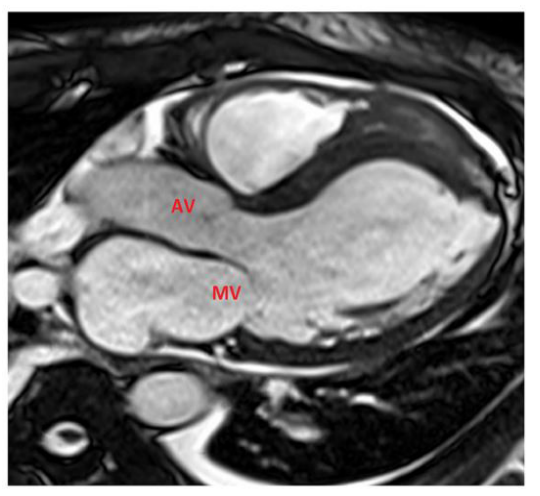
Figure 1E: 50-year-old male with history of T-ALL and febrile neutropenia. E, SSFP three-chamber cardiac MR view shows no evidence of valvular vegetations on the aortic or mitral valves. AV: Aortic valve, MV: Mitral valve.

Figure 1F: 50-year-old male with history of T-ALL and febrile neutropenia. F, Resting first-pass perfusion cardiac MR image shows a non-enhancing, multilobulated intramyocardial lesion in the mid-to-apical interventricular septum (asterisk), consistent with the absence of viable microvasculature.
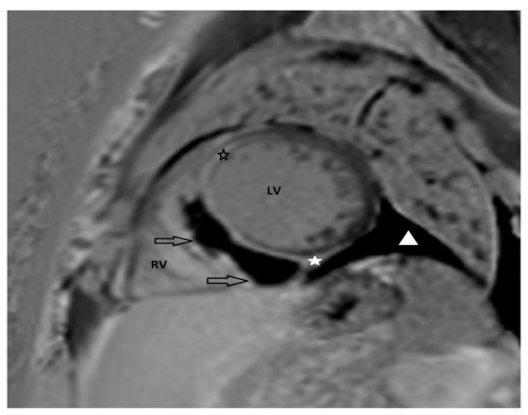
Figure 1G: 50-year-old male with history of T-ALL and febrile neutropenia. G, Late gadolinium enhancement with phasesensitive recovery (PSIR) cardiac MR image shows large septal intramyocardial lesion with central non-enhancing necrotic core and peripheral rim enhancement compatible with abscess (arrows). Mid-wall enhancement in the adjacent myocardium reflects associated inflammatory involvement (asterisks). In addition, there is a small pericardial effusion seen (triangle). LV= Left ventricle, RV= Right ventricle.
signal intensity and perilesional edema (asterisk), and improvement of rim enhancement (arrow).
Discussion
Myocardial abscesses occur in approximately 20% of patients with infective endocarditis and typically arise adjacent to the affected valve due to contiguous spread. In contrast, isolated myocardial abscesses develop independently of valvular involvement and result from septicemia-induced focal myocarditis progressing to liquefactive necrosis [8, 9]. Intramyocardial abscess is difficult to diagnose due to nonspecific and variable presentations, including fever, leukocytosis, bacteremia, chest pain, or even acute coronary syndrome [5, 10]. The integration of cardiac MRI with echocardiography, CT, and nuclear imaging methods enhances the diagnostic accuracy for myocardial abscesses. MRI complements echocardiography by offering superior tissue characterization, while CT is useful for evaluating extracardiac complications. FDG-PET provides valuable metabolic insights, especially in cases of prosthetic valve infections or disseminated disease. Utilizing multiple imaging modalities allows for a comprehensive assessment and helps guide therapeutic strategies [11-13].
In conclusion, our case emphasizes the pivotal role of multimodality imaging in the diagnosis of rare intracardiac fungal abscesses. In immunocompromised patients, particularly those with underlying cardiomyopathy in whom invasive diagnostic procedures may have substantial risk, cardiac MRI provides a safe and comprehensive noninvasive assessment. In the absence of histopathologic confirmation, CMR contributes decisively to diagnostic confidence and clinical management by distinguishing abscess from neoplasm, thrombus, hematoma, and other intracardiac masses, based on the characteristic demonstration of a central non-enhancing necrotic core, peripheral rim enhancement, and associated inflammatory signal abnormalities.
All authors declare that they have no conflicts of interest or relevant financial disclosures related to this case report.
References
- Sagar S, Liu PP, Cooper LT Jr. (2012). Myocarditis. Lancet. 379: 738747.
- Chikwe J, Barnard J, Pepper JR. (2004). Myocardial abscess. Heart. 90: 597.
- Kim TY, Jung JI, Kim YJ, Kim HW, Lee HG. (2015). CT and MRI evaluation of cardiac complications in patients with hematologic diseases: a pictorial review. The International Journal of Cardiovascular Imaging. 31: 159-167.
- Chen W, Jeudy J. (2019). Assessment of Myocarditis: Cardiac MR, PET/CT, or PET/MR? Curr Cardiol Rep. 21: 76.
- Shackcloth MJ, Dihmis WC. (2001). Contained rupture of a myocardial abscess in the free wall of the left ventricle. Ann Thorac Surg. 72: 617619.
- Khan B, Strate RW, Hellman R. (2007). Myocardial abscess and fatal cardiac arrhythmia in a hemodialysis patient with an arterio-venous fistula infection. Semin Dial. 20: 452-454.
- Lutsenko L, Sokolov M, Krychkevych V. (2023). A rare case report of fatal acute myocardial infarction as a complication of myocardial abscess. Eur Heart J Case Rep. 7: ytad167.
- Iqbal J, Ahmed I, Baig W. (2008). Metastatic myocardial abscess on the posterior wall of the left ventricle: a case report. J Med Case Rep. 2: 258.
- Crum-Cianflone NF. (2008). Bacterial, fungal, parasitic, and viral myositis. Clin Microbiol Rev. 21: 473-494.
- Kirkbride RR, Rawal B, Mirsadraee S, Galperin-Aizenberg M, Wechalekar K, et al. (2021). Imaging of Cardiac Infections: A Comprehensive Review and Investigation Flowchart for Diagnostic Workup. J Thorac Imaging. 36: W70-W88.
- Reynier C, Garcier J, Legault B, Motreff P, Ponsonnaille J, et al. (2001). Cross-sectional imaging of post endocarditis paravalvular myocardial abscesses of native mitral valves: 4 cases. J Radiol. 82: 665-669.
- Harris KM, Ang E, Lesser JR, Sonnesyn SW. (2007). Cardiac magnetic resonance imaging for detection of an abscess associated with prosthetic valve endocarditis: a case report. Heart Surg Forum. 10: E186-187.
- Jaleel J, Kaushik P, Patel C, Chandra KB, Yadav S, et al. (2023). Utility of 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography in Cardiac Infections. Indian Journal of Nuclear Medicine. 38: 140-144.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.