A Multicentre, Prospective, Observational Study onEfficacy and Safety of Telmisartan + Bisoprolol for Management of Hypertension in Coronary Artery Disease (CAD) Patients in India – SUPERIOR
by Uday M Jadhav1, Arindam Pande2, Hanish Gupta3, Sandipan Halder4, Ketan L Pakhale5, Soumya Kanti Dutta6, Nitin Zalte7*
1Dr Jadhav’s Clinic, Mumbai, India
2Cardiac Care, Kolkata, India
3Life Aid Clinic, Delhi, India
4The Clinic P-2, Kolkata, India
5Metabol – Lifestyle clinic for metabolic syndrome, Mumbai, India
6Kolkata Heart Foundation, Kolkata, India
7Department of Medical Affairs, Mankind Pharma, Delhi, India
*Corresponding author: Dr Nitin Zalte, Department of Medical Affairs, Mankind Pharma, Delhi, India.
Received Date: 03 January, 2026
Accepted Date: 10 February, 2026
Published Date: 12 February, 2026
Citation: Jadhav UM, Pande A, Gupta H, Halder S, Pakhale KL, et al. (2026) A Multicentre, Prospective, Observational Study on Efficacy and Safety of Telmisartan + Bisoprolol for Management of Hypertension in Coronary Artery Disease (CAD) Patients in India – SUPERIOR. Cardiol Res Cardio vasc Med 11:294. DOI: https://doi.org/10.29011/2575-7083.100294
Abstract
Background: Coronary artery disease (CAD) is a common heart condition with hypertension (HTN) as a major independent risk factor. The study shows that the telmisartan (40 mg)–bisoprolol (2.5/5 mg) fixed-dose combination is effective and well tolerated in Indian patients with coronary artery disease and uncontrolled hypertension. The complementary RAAS blockade and β1-selective inhibition may provide synergistic cardiovascular benefits while improving treatment adherence. However, a combination of antihypertensive regimens is required for sustaining effective long-term HTN control. Objective: The study aimed to evaluate the effectiveness and safety of the combination of Telmisartan 40 mg + Bisoprolol 2.5/5mg for the management of HTN in CAD patients in India. Method: This was an investigator-initiated, multicenter, prospective, observational, population-based study that included adult patients with CAD and long-standing uncontrolled HTN (blood pressure (BP) > 140/90 mmHg) on existing therapy of ACE-I/ARB + BB who were switched to Telmisartan 40mg + bisoprolol 2.5/5mg as part of routine clinical practice. The primary objective was to assess the percentage of patients achieving the target blood pressure (Systolic BP (SBP):120-130 mmHg/ Diastolic BP (DBP):70-80 mmHg) at the end of 12 weeks of treatment. Results: A total of 157 subjects were recruited in the study, of which 82 (52.2%) were male. The mean age of the study population was 56.58(11.34) years. A total of 34 (21.5%) had dyslipidemia, and 25 (15.8%) patients had CAD. Mean SBP as well as DBP demonstrated statistically significant reductions at each follow-up visit from baseline, in sitting, supine, and standing positions. The baseline mean sitting SBP/DBP was 158.11(12.43)/93.41(3.74) mmHg; at the end of 12 weeks with Telmisartan 40mg + bisoprolol 2.5/5mg treatment, BP reduced to 134.43(6.85)/77.44(7.13) mmHg. The study demonstrated a significant reduction from baseline by 23.68/12.01 mmHg. At the final visit, the available data did not record any clinically significant changes in ECG or echocardiography. Laboratory parameters (available) reported statistically significant reductions in mean serum potassium (3.19 mEq/L), cholesterol (5.99), HbA1c (0.19%), and random blood glucose (0.68) at the final visit. An improvement in eGFR by 0.6 ml/min/1.73m2 and a reduction of serum creatinine by 0.02 mg/ dL were noted, although statistically non-significant. There were no adverse events reported during the study period. Conclusion: The study shows that the telmisartan (40mg) + bisoprolol (2.5/5mg) combination therapy could be effective and safe in Indian patients with CAD and uncontrolled HTN. Long-term studies are required to establish a strong safety profile.
Keywords: Hypertension, CAD, combination antihypertensive treatment, ARB, β-blockers, bisoprolol, telmisartan, Indian hypertensive patients with CAD.
Abbreviations: ARBs: Angiotensin receptor blockers; BP: Blood pressure; CAD: Coronary artery disease; DBP: Diastolic blood pressure; HF: Heart failure; HTN: Hypertension; SBP: Systolic blood pressure
Introduction
Coronary artery disease (CAD) is a common heart condition that involves atherosclerotic plaque formation in the vessel lumen leading to impairment in blood flow and thus oxygen delivery to the myocardium [1]. It can progress to congestive heart failure (HF) [1]. In 2022, the age-standardized prevalence of CAD was 3605 (2892 to 4454) per 100000 [2]. Studies have shown that 50–60% of CAD patients had comorbid hypertension (HTN), and 13% HTN patients had comorbid CAD [3]. Epidemiological studies have established a strong association between HTN and CAD. HTN is a major independent risk factor for the development of CAD, stroke, and renal failure [4]. Pathophysiological mechanisms of blood pressure as a risk factor for CAD are complex and include the influence of blood pressure as a physical force on the development of atherosclerotic plaque, and the relationship between pulsatile hemodynamic/arterial stiffness and coronary perfusion [5]. A meta-analysis by Lewington S et al reported that at ages 40-69 years, each difference of 20 mmHg usual SBP (or 10 mmHg usual DBP) is associated with more than a twofold difference in the stroke death rate, and with twofold differences in the death rates from ischemic heart diseases (IHD) and from other vascular causes [6]. 2017 AHA/ACC guidelines for HTN management lowered the BP target to BP <130/80 mmHg in the population with CAD [7].
β-Blocker administration remains the standard of care in hypertensive patients with angina pectoris or prior MI, or LV dysfunction with or without symptoms of HF unless contraindicated. The β-blockers carvedilol, metoprolol, and bisoprolol have been shown to improve outcomes in patients with HF [8]. Bisoprolol is reported to be well-suited as the first-line treatment of angina in primary care [9]. Bisoprolol can effectively reduce resting heart rate in Asian CAD patients with comorbid HTN and hence, improve composite cardiac clinical outcome [10].
Several angiotensin receptor blockers (ARBs) have been shown to reduce the incidence or severity of IHD events, the progression of renal disease in type 2 diabetes mellitus, and cerebrovascular events [8]. Telmisartan Randomised Assessment Study in ACE Intolerant Subjects with Cardiovascular Disease (TRANSCEND) study reported that Telmisartan was well tolerated in patients unable to tolerate ACE inhibitors and modestly reduced the risk of the composite outcome of cardiovascular death, myocardial infarction, or stroke [11]. ARB is one of the five classes of antihypertensive drugs recommended as first-line treatment for stage 1–2 HTN by Indian guidelines on HTN-IV [12].
Combination antihypertensive drug therapy is typically needed to achieve and sustain effective long-term BP control [4]. Two neurohormonal systems, the sympathetic nervous system (SNS) and renin–angiotensin–aldosterone system (RAAS), are intricately involved in the progression of disease throughout the cardiovascular disease continuum [13]. Therefore, there is a considerable rationale for combining pharmacological therapies that target both neurohormonal pathways [13].
There is insufficient evidence on the efficacy and safety of the bisoprolol with telmisartan combination and its potential in treating CAD patients with uncontrolled HTN. To date, limited real-world evidence is available evaluating the effectiveness and safety of the telmisartan and bisoprolol combination in Indian patients with CAD and uncontrolled hypertension. Thus, the purpose of this study was to determine the effectiveness and safety of Telmisartan 40mg+ Bisoprolol 2.5/5mg for the management of HTN in Indian CAD patients.
Method
This was an investigator-initiated, multicenter, prospective, observational, population-based study conducted across 50 sites in India from July 2024 to Dec 2024 to understand the effectiveness and safety of Telmisartan 40mg+ Bisoprolol 2.5/5mg for the management of HTN in CAD patients with uncontrolled HTN.
The study was initiated after receiving approval from the Suraksha Ethics Committee. The study was conducted following ‘The Declaration of Helsinki’ principles (World Medical Association) and Good Clinical Practice (GCP) Guidelines issued by the ICMR & DCG(I), Govt. of India. The study was explained to the patients in detail, and they were recruited only after receiving voluntary written informed consent.
The inclusion criteria were adult patients of either sex aged ≥18 and ≤85 years; diagnosed with CAD with or without heart failure with normal cardiac function or with HFrEF (≤40% LVEF), and long-standing uncontrolled HTN (blood pressure (BP) > 140/90 mmHg) on existing therapy of ACE-I/ARB + BB; and patients who were switched from existing therapy to Telmisartan 40mg + Bisoprolol 2.5/5mg as per the prescription of treating physicians and at investigator discretion as part of routine clinical practice.
Exclusion criteria were pregnant or breastfeeding female patients; patients suspected of or known to have an intolerance to any of the drugs prescribed; presence of any other clinically significant disease or laboratory findings that in the Investigator’s opinion may affect the study outcomes or continued participation of the patient in the study; and the patients participating in another study concurrently or within 4 weeks before the Screening Visit.
Study medications were Telmisartan 40mg+ Bisoprolol 2.5/5mg as per the prescription of treating physicians, and the study duration was 12 weeks.
Primary outcome: Percentage of patients achieving the target blood pressure (Systolic BP (SBP):120-130 mmHg/ Diastolic BP (DBP):70-80 mmHg) at the end of the treatment after 12 weeks.
Secondary outcomes:
|
Assessment |
Visit 1 – Screening & Enrolment (Day 0) |
Visit 2 – Follow-up |
Visit 3 – Follow-up |
Visit 4 – Follow-up |
|
(Day 28 ± 3) |
(Day 56 ± 3) |
(Day 84 ± 3) |
||
|
Patient information & Informed consent |
√ |
|||
|
Demographics & Medical history |
√ |
|||
|
Vital parameters |
√ |
√ |
√ |
√ |
|
Physical examination |
√ |
√ |
√ |
√ |
|
Inclusion/Exclusion |
√ |
|||
|
Enrolment Heart Rate Monitoring |
√ |
|||
|
√ |
√ |
√ |
||
|
√ |
√ |
√ |
||
|
Laboratory & Diagnostic Assessment |
√ |
Optional |
Optional |
√ |
- Percentage of patients achieving the target blood pressure (SBP:120-130 mmHg/DBP: 70-80 mmHg) after 4 weeks and 8 weeks of treatment
- Mean reduction in seated SBP and DBP after 4, 8, and 12 weeks of treatment (Average of 3 BP readings measured using an Automated Office BP Machine)
- Mean reduction in SBP and DBP from supine to standing position after 4, 8, and 12 weeks of treatment (average of three BP readings measured using an automated office BP machine)
- Evaluation of echocardiography, ECG, HbA1c, random glucose, serum creatinine, lipid profile, and eGFR at baseline and 12 weeks of treatment; serum electrolytes/potassium (optional at 4 & 8 weeks), echocardiography, serum potassium & ECG
- Physician’s global assessment of treatment post-completion of the treatment regimen
- Safety and tolerability assessment of treatment postcompletion of the treatment regimen (Serious ADRs and any other ADRs )
- Mean Change in score of SF-36 Quality of Life Questionnaire after 4, 8, and 12 weeks of treatment
Eligible patients on enrollment in the study underwent diagnostic and laboratory tests at baseline and 12 weeks (ECG, ECHO, HbA1c, random glucose, serum creatinine, eGFR, serum electrolytes, lipid profile). Patients were requested to visit the site once every 4 weeks for follow-up. During the follow-up visit, the patient’s disease status (heart rate (HR) and BP) was evaluated. The physician’s global assessment of the treatment post-completion of the treatment regimen was done at 12 weeks. The detailed schedule of procedures is provided in Table 1.
Table 1: Schedule of procedures. |
Statistical Analysis
Demographic Characteristics: Continuous data such as age and weight were summarized with n, mean, SD, and range. Categorical data such as sex were depicted with count (%).
Efficacy Analysis: For continuous variables such as SBP & DBP at different clinical postures, heart rate, serum electrolytes, HbA1c, random glucose, eGFR, and serum creatinine, mean change from baseline was derived and paired t-test was applied to assess statistical significance. For categorical variables such as the percentage of patients achieving target blood pressure at 4, 8, and 12 weeks, heart rate & ECG, ECHO, other laboratory parameters, and safety & tolerability, results were summarized with count (%). If applicable, chi-square test was performed to assess statistical significance. A p-value of <0.05 was considered statistically significant.
Safety Analysis: Incidences of adverse events were summarized with count (%). Other safety measures such as physical examination and vital signs were summarized using descriptive statistics and change from baseline, if applicable.
Results
A total of 157 eligible subjects were enrolled in the study, of whom data were considered for final analysis. The CONSORT flowchart detailing patient enrolment is presented in Figure 1.
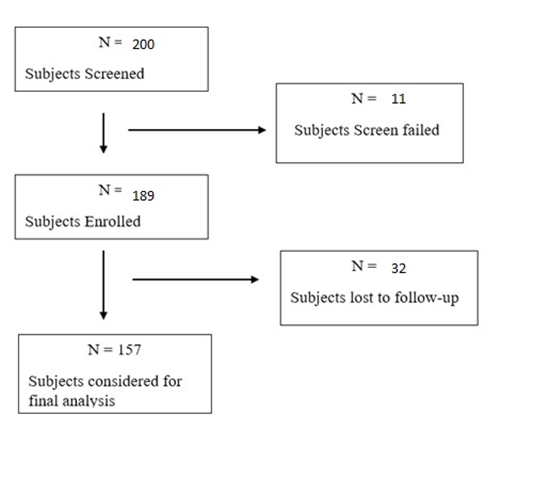
Figure 1: CONSORT Flowchart of Participant Recruitment
Patient Demographics
Out of 157, 82(52.2%) were male and 75 (47.8%) were female. The mean age of the study population was 56.58(11.34) years, and the mean weight was 72.43(8.92) kg. A total of 43 (27.2%) of the patients had diabetes mellitus, 34 (21.5%) had dyslipidaemia, and 25 (15.8%) patients had CAD.
The study population’s demographics, along with vitals at baseline and concomitant medical history, are presented in (Table 2).
|
Parameter |
Count (N=157) |
|
Age (years) |
|
|
N |
157 |
|
Mean (SD) |
56.58(11.34) |
|
Gender |
|
|
Male |
82(52.2%) |
|
Female |
75(47.8%) |
|
Height (Cms.) |
|
|
N |
155 |
|
Mean (SD) |
161.82(8.92) |
|
Weight (Kgs.) |
|
|
N |
155 |
|
Mean (SD) |
72.43(10.53) |
|
Body Temperature (°C) |
|
|
N |
118 |
|
Mean (SD) |
36.92(.33) |
|
Heart Rate (bpm) |
|
|
N |
120 |
|
Mean (SD) |
80.65(13.41) |
|
Respiratory
Rate (bpm) |
|
|
N |
119 |
|
Mean (SD) |
19.85(1.50) |
|
Any
concomitant medical history (if available) |
|
|
Diabetes
Mellitus |
43 (27.2%) |
|
Dyslipidaemia |
34 (21.5%) |
|
Hypothyroidism |
22 (13.9%) |
|
Hyperthyroidism |
3 (1.9%) |
|
CAD |
25 (15.8%) |
|
Osteoporosis |
1 (0.6%) |
|
CKD |
2 (1.3%) |
|
Thyroid |
1 (0.6%) |
|
Anemia |
2 (1.3%) |
|
Anxiety |
1 (0.6%) |
|
Vertigo |
1 (0.6%) |
Table 2: Summary Statistics of Demographics
Baseline prescribed dosage of FDC, Telmisartan (40mg) + Bisoprolol (2.5/5mg)
A total of 85 (54.14%) of the subjects were prescribed Telmisartan (40mg) + Bisoprolol (5mg), while 75 (45.85%) were prescribed Telmisartan (40mg) + Bisoprolol (2.5mg).
Effectiveness Parameters
No. of subjects achieving target BP
The primary objective of the study was to evaluate the percentage of patients achieving the target blood pressure (120-130 mmHg SBP/70-80 mmHg DBP) at the end of 12 weeks of the Telmisartan 40mg + Bisoprolol 2.5/5mg treatment.
At the end of 12 weeks of study treatment, 37 (23.56%) of the subjects reached the target SBP <130 mmHg, and 108 (68.78%) of the subjects achieved the target DBP of 70-80 mmHg in the sitting position.
A total of 20 (12.74%) of the subjects attained the target SBP/DBP of <130 mmHg / 70-80 mmHg (sitting) at the end of 12 weeks.
A summary of the number (%) of study subjects achieving target SBP and DBP at each follow-up visit in sitting, supine, and standing positions is provided in Figure 2 (A and B).
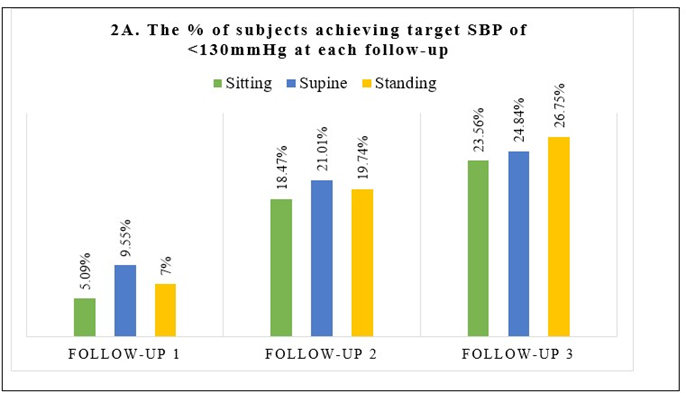
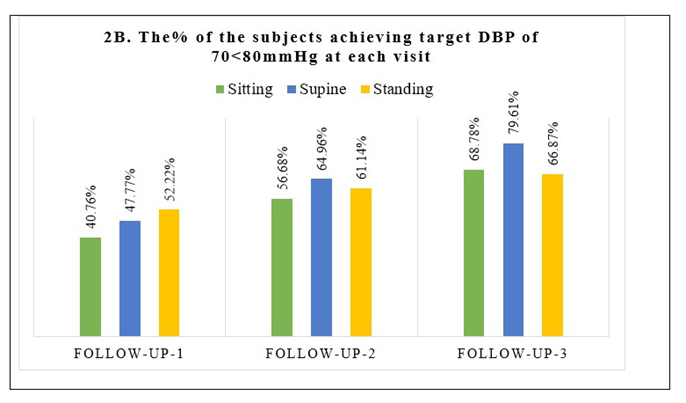
Figure 2 (A and B): A summary of the % of subjects achieving target SBP and DBP at each follow-up visit.
The secondary objective was to understand the mean change in SBP and DBP from baseline to each follow-up visit in sitting, supine, and standing positions.
Changes in SBP: One of the effectiveness parameters recorded for this clinical study was the mean change in sitting, supine, and standing SBP (mmHg) for all patients during Baseline Visit, Follow-up Visit 1, 2 and 3 (Final Visit).
With study treatment, a statistically significant reduction (p < 0.001) was observed at each visit.
Sitting SBP: The mean baseline sitting SBP decreased from 158.11(12.43) mmHg to 134.43(6.85) mmHg at follow-up visit 3, i.e., at the end of the 12-week study treatment.
Supine SBP: The mean supine SBP at baseline was 157.86(12.83) mmHg. It reduced to 134.11(8.06) mmHg at final visit.
Standing SBP: The mean standing SBP at baseline was 155.82(12.75) mmHg, which decreased to 133.03(7.044) mmHg with 12 weeks of Telmisartan 40mg+ Bisoprolol 2.5/5mg treatment.
A comparison of mean SBP (sitting, supine, and standing) at each visit is presented in Table 3. A comparison of mean reductions at each visit is presented in Figure 3 (A-C).
|
Parameters |
Baseline visit (N=157) |
Follow-up visit 1 (Week 4) (N=157) |
Follow-up visit 2 (Week 8) (N=156) |
Follow-up visit 3 (Week 12) (N=157) |
|
Sitting SBP |
||||
|
Mean SBP(SD) (mmHg) |
158.11(12.43) |
145.24(10.38) |
139.53(11.40) |
134.43(6.85) |
|
Supine SBP |
||||
|
Mean SBP(SD) (mmHg) |
157.86(12.83) |
145.50(10.87) |
139.42(12.08) |
134.11(8.06) |
|
Standing SBP |
||||
|
Mean SBP(SD) (mmHg) |
155.82(12.75) |
145.22(9.58) |
139.39(10.56) |
133.03(7.044) |
Table 3: Comparison of changes in mean SBP (sitting, supine, and standing) at each visit
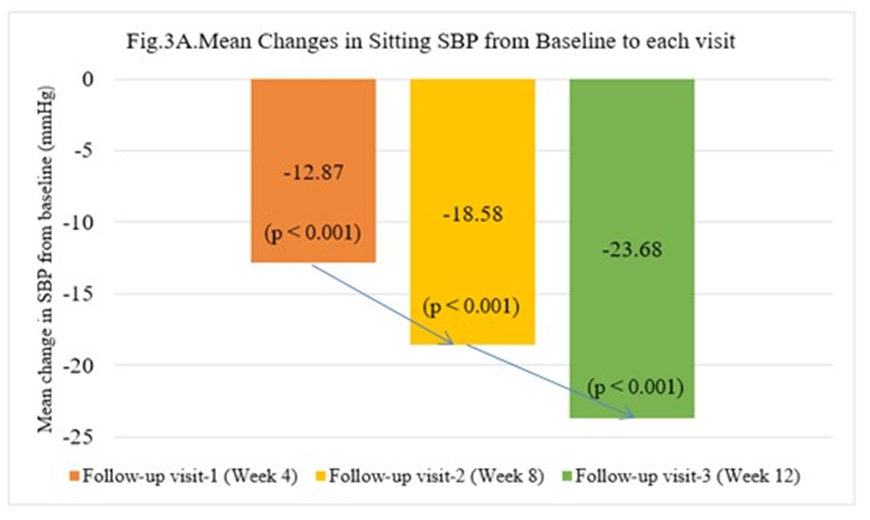
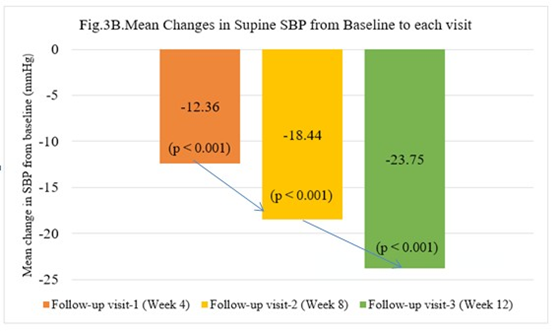
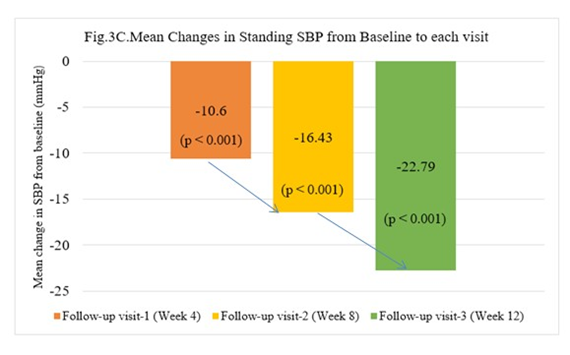
Figure 3 (A-C): A Comparison of mean reductions in SBP (sitting, supine, and standing) at each visit from baseline
Changes in the mean DBP (mmHg):
Other effectiveness parameters included the change in mean DBP. The study observed statistically significant reductions in mean sitting, supine, and standing DBP from baseline to consecutive follow-up visits at weeks 4, 8, and 12. Statistically significant (p < 0.001) reductions were observed across all follow-up visits in sitting, supine, and standing DBP.
Sitting DBP: The mean baseline sitting DBP was 93.41(3.74) mmHg. It reduced by 12.01 mmHg to 77.44(7.13) mmHg at week 12.
Supine DBP: The mean supine DBP at baseline was 92.36 (5.06) mmHg and decreased by 15.31 mmHg to 77.05 (7.51) mmHg at final visit.
Standing DBP: The mean standing DBP at baseline was 91.38 (3.82) mmHg. It decreased by 14.3 mmHg to 77.08 (6.81) mmHg at final visit.
A comparison of mean DBP (sitting, supine, and standing) at each visit is presented in Table 4. A comparison of mean reductions at each visit is presented in Figure 4 (A-C).
|
Parameters |
Baseline visit (N=157) |
Follow-up visit 1 (Week 4) (N=157) |
Follow-up visit 2 (Week 8) (N=156) |
Follow-up visit 3 (Week 12) (N=157) |
|
Sitting DBP |
||||
|
Mean DBP(SD) (mmHg) |
93.41(3.74) |
81.40(7.37) |
78.59(7.62) |
77.44(7.13) |
|
Supine DBP |
||||
|
Mean DBP(SD) (mmHg) |
92.36(5.06) |
81.09(8.04) |
78.33(7.96) |
77.05(7.51) |
|
Standing DBP |
||||
|
Mean DBP(SD) (mmHg) |
91.38(3.82) |
80.76(7.23) |
78.64(7.533) |
77.08(6.81) |
Table 4: Comparison of changes in mean DBP (sitting, supine, and standing) at each visit
Figure 4(A-C): A comparison of mean reductions at each visit in sitting, supine, and standing DBP at each visit from baseline
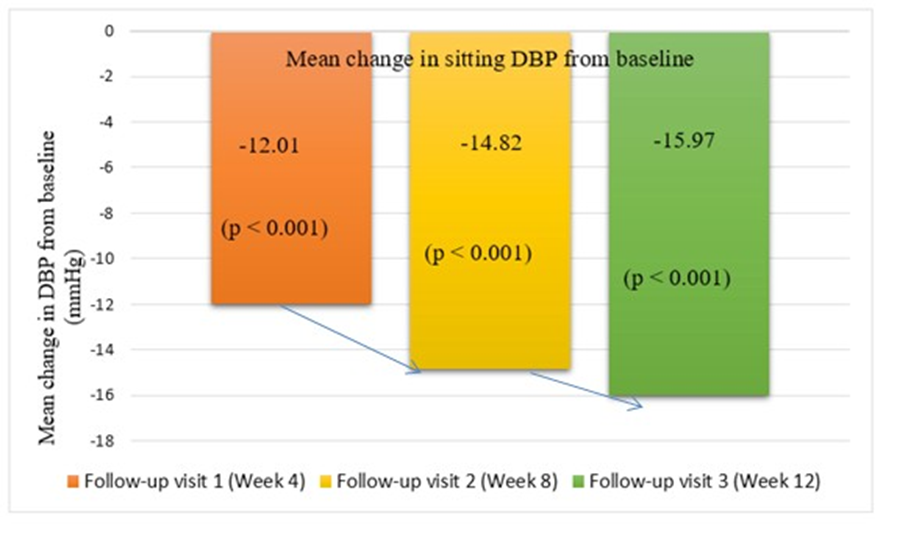
Figure 4A: A comparison of mean reductions in sitting DBP from baseline
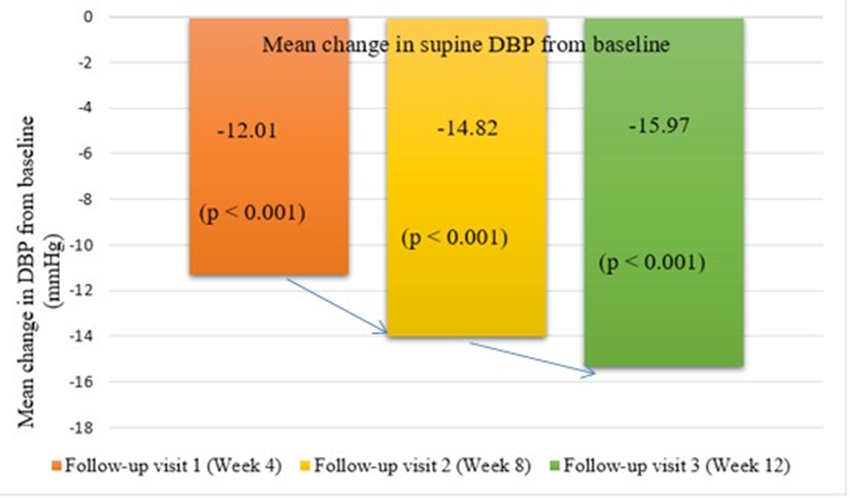
Figure 4B: A comparison of mean reductions in Supine DBP from baseline
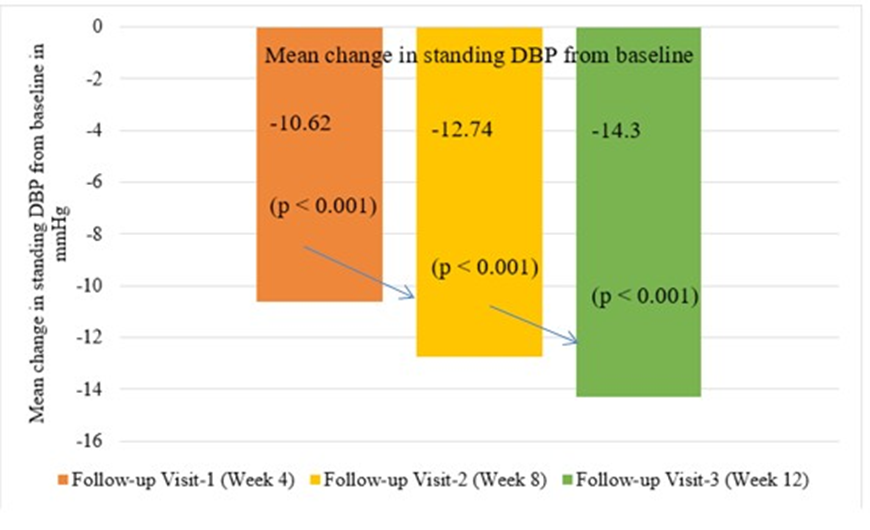
Figure 4C: A comparison of mean reductions in Standing DBP from baseline
One of the secondary objectives was to assess the changes in available ECG, echocardiograph, and laboratory parameters (HbA1c, random glucose, serum creatinine, lipid profile, and eGFR) from baseline to final visit.
Analysis of Summary Statistics of ECG and Echocardiograph
ECG: One of the safety parameters recorded for this clinical study was ECG at baseline and final visit. At baseline, out of 157 patients, ECG details of 128 patients were available. For the final visit, ECG details of 44 subjects were available. At final visit, none of the patients’ ECGs recorded atrioventricular block. The summary statistics of ECG results of the baseline and final visits are presented in Table 5.
|
ECG report |
No. of patients |
|
Baseline ECG report |
(N=128) |
|
Normal ECG |
24 (15.28 %) |
|
Sinus Rhythm (General) |
26 (16.5%) |
|
Sinus Tachycardia (General) |
6 (3.8%) |
|
Sinus Bradycardia |
8 (5%) |
|
Sinus Bradycardia, Ventricular Hypertrophy, T-wave abnormality |
1 (0.6%) |
|
Sinus Bradycardia, Left Axis Deviation |
2 (1.2%) |
|
Sinus Bradycardia, Left Anterior Fascicular Block |
2 (1.2%) |
|
Sinus Rhythm, Poor R wave progression |
3 (1.7%) |
|
Sinus Rhythm, Left Ventricular Hypertrophy |
2 (1.2%) |
|
Sinus Rhythm, Right Bundle Branch Block (RBBB) |
1 (0.6%) |
|
Sinus Rhythm, Complete Right Bundle Branch Block (CRBBB) |
1 (0.6%) |
|
Sinus Rhythm, Prolonged P-wave |
1 (0.6%) |
|
Sinus Rhythm, Non-Specific T-wave abnormality |
3 (1.7%) |
|
Sinus Rhythm, Left Axis Deviation |
3 (1.7%) |
|
Sinus Rhythm, Left Anterior Fascicular Block |
1 (0.6%) |
|
Sinus Rhythm, Septal Infarct, Probably Evolved |
1 (0.6%) |
|
Sinus Rhythm, IV Conduction Block, Myocardial Infarction |
1 (0.6%) |
|
Sinus Tachycardia, ST-T Changes in Leads 2,3,V2 |
1 (0.6%) |
|
Sinus Tachycardia, Left Anterior Fascicular Block |
1 (0.6%) |
|
Left Axis Deviation |
1 (0.6%) |
|
Right Ventricular Hypertrophy |
1 (0.6%) |
|
Left Ventricular Hypertrophy |
3 (1.7%) |
|
Hypertrophy Negative T-Wave |
1 (0.6%) |
|
NSR (Normal Sinus Rhythm) with ST-T Changes |
6 (3.8%) |
|
T-wave Inversion in Lead V1 |
3 (1.7%) |
|
ST-T Changes in Multiple Leads |
10 (6.3%) |
|
Inferior and Lateral ST-T Changes |
2 (1.2%) |
|
Ventricular Premature Contractions (VPCs) |
1 (0.6%) |
|
Right Bundle Branch Block (RBBB) |
1 (0.6%) |
|
Final Visit |
N = 44 |
|
ECG within normal limits |
16 (10.1%) |
|
Incomplete LBB |
1 (0.6%) |
|
LVH (otherwise ECG within normal limit) |
20 (12.7%) |
|
Sinus tachycardia |
2 (1.2%) |
|
ST-T changes in anterolateral leads |
4 (2.5%) |
|
ST-T depression anterior wall |
1 (0.6%) |
Table 5: Summary of ECG results
The summary statistics of the available echocardiograph reports from the baseline and final visits are presented in (Table 6).
|
Final Visit (12th week) |
N = 28 |
|
LVEF (50-55%) with Gr-I diastolic dysfunction |
5 (3.1%) |
|
LVEF (50-55%) with Fair LV systolic function |
3 (1.7%) |
|
Concentric LVH, LVEF (50-55%), Gr-I diastolic dysfunction |
2 (1.2%) |
|
LVEF (50-55%) |
1 (0.6%) |
|
LVEF (50-50%), diastolic dysfunction |
1 (0.6%) |
|
LVEF (40-45%) with Mild LV systolic dysfunction |
4 (2.5%) |
|
LVEF (40-45%) with Gr-I diastolic dysfunction |
1 (0.6%) |
|
LVEF (40-45%) with Pan LV systolic dysfunction |
1 (0.6%) |
|
LVEF (40-45%) with Hypokinesia of anterior wall |
1 (0.6%) |
|
LVEF (57%) with Normal LV systolic function |
1 (0.6%) |
|
Hypokinesia of IVS (including anterior & anterolateral wall involvement) |
3 (1.7%) |
|
Hypokinesia of anterior wall with Gr-I diastolic Dysfunction |
2 (1.2%) |
|
Mild LV systolic dysfunction |
1 (0.6%) |
|
LVEF = 60%, No PH |
1 (0.6%) |
|
Normal |
1 (0.6%) |
Table 6: Summary of Echocardiography Results
Analysis of available Laboratory and Diagnostic Assessments:
The summary statistics of the mean change from baseline to the final visit (Visit-3) for HbA1c, Random Blood Glucose, eGFR, and Serum Creatinine are presented in Table 7.
The mean difference at the final visit from baseline in HbA1c was -0.19%, random blood glucose was -0.68 mmol/L, eGFR was 0.6 ml/ min/1.73m2, and serum creatinine was -0.02 mg/dL.
The difference in mean HbA1c was statistically significant.
|
Parameters |
HbA1c |
Random Blood Glucose (mmol/L) |
eGFR (ml/min/1.73m2) |
Serum Creatinine (mg/dl) |
|
Baseline visit |
||||
|
N |
157 |
157 |
157 |
157 |
|
Mean(SD) |
6.88(1.5) |
7.89(4.05) |
89.74(24.56) |
0.89(0.37) |
|
Diabetic |
65(41.4%) |
|||
|
Non-Diabetic |
92(58.6%) |
|||
|
Follow-up visit 3 |
||||
|
N |
157 |
157 |
154 |
157 |
|
Mean |
6.69(1.32) |
3.02 |
90.34(26.07) |
0.87(0.24) |
|
Diabetic |
80(50.6%) |
|||
|
Non-Diabetic |
77(48.7%) |
|||
|
Mean Change from Baseline |
-0.19 |
-0.68 |
0.6 |
-0.02 |
|
p-value |
0.003 |
0.010 |
0.613 |
0.448 |
Table 7: Summary Statistics of Laboratory and Diagnostic Assessments
Mean changes in serum potassium and serum cholesterol from baseline to week 12 (final visit) are presented in Table 8. Both parameters reported statistically significant reductions in mean values from baseline by 3.19 and 5.99, respectively.
|
Parameters |
Serum Potassium mEq/L |
Serum cholesterol |
|
Baseline Visit |
||
|
N |
157 |
157 |
|
Mean(SD) |
4.06(0.48) |
170.03(56.82) |
|
Visit 3 |
||
|
N |
157 |
154 |
|
Mean(SD) |
0.87(0.24) |
164.04(0.43) |
|
Mean Change from Baseline |
-3.19 |
-5.99 |
|
p-value |
<0.01 |
<0.01 |
Table 8: Summary of changes in mean serum potassium and cholesterol
Physician’s global assessment of treatment post-completion of the treatment regimen:
One of the efficacy parameters recorded for this clinical study was Physician’s global assessment of treatment.
A total of 77.3% of the physicians were satisfied with the improvement in the patient’s ability to perform daily activities following Bisoprolol + Telmisartan administration.
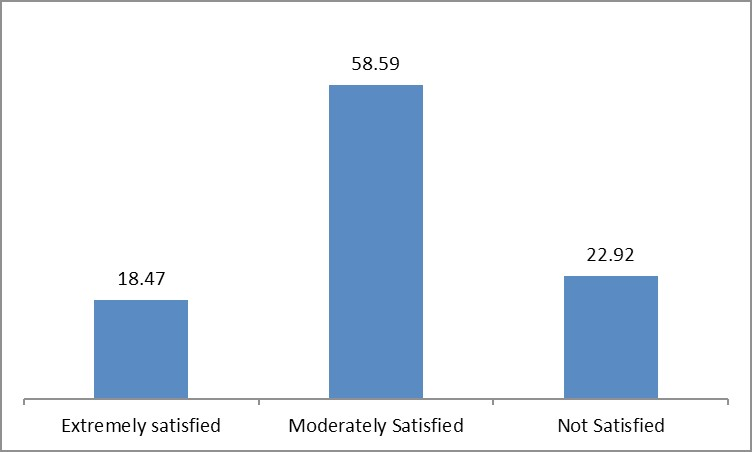
Figure 5: Graphical Representation of Physician’s global assessment of treatment: “How Satisfied are you with the improvement in the patient’s ability to perform daily activities following Bisoprolol + Telmisartan administration?”
“How likely would you recommend the usage of Bisoprolol + Telmisartan in existing coronary artery disease (CAD) patients with uncontrolled HTN?”
One of the efficacy parameters recorded for this clinical study was Physician’s global assessment of treatment: “How likely would you recommend the usage of Bisoprolol + Telmisartan in existing coronary artery disease (CAD) patients with uncontrolled HTN?” Summary statistics are presented in Table 9 and Figure 6 below.
It was evident from Table 9 that, out of 157 physicians, 63 (40.2%) were extremely satisfied, 88 (54.8%) were moderately satisfied, and 0.6% were slightly satisfied with patients’ treatment.
|
Frequency |
Percentage (%) |
|
|
N |
157 |
|
|
Extremely satisfied |
63 |
40.2% |
|
Moderately satisfied |
88 |
54.8% |
|
Slightly Satisfied |
0.6 |
0.6% |
Table 9: Summary Statistics of Physician’s Physician’s global assessment of treatment: “How likely would you recommend the usage of Bisoprolol + Telmisartan in existing coronary artery disease (CAD) patients with uncontrolled HTN?”
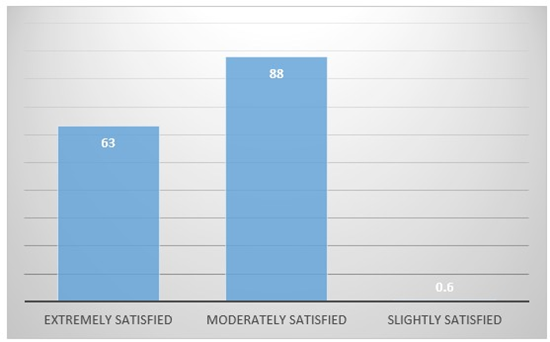
Figure 6: Graphical Representation of Physician’s global assessment of treatment: “How likely would you recommend the usage of Bisoprolol + Telmisartan in existing CAD patients with uncontrolled HTN?”
None of the patients reported any adverse effects with the study treatment during the study period.
Discussion
To the best of our knowledge, this is one of the first real-world evidence studies conducted in the Indian population evaluating the effectiveness and safety of the combination therapy of Telmisartan 40 mg + Bisoprolol 2.5/5mg in Indian patients with CAD and uncontrolled HTN. The study demonstrated that the combination treatment with Telmisartan 40 mg + Bisoprolol 2.5/5mg significantly (p<0.001) reduced SBP as well as DBP in sitting, supine, and standing positions at weeks 4, 8, and 12. The results showed that the dual combination therapy demonstrated effectiveness as early as 4 weeks and sustained the same till 12 weeks.
A systematic review and meta-analysis by Ettehad D et al. concluded that BP lowering significantly reduces vascular risk across various baseline blood pressure levels and comorbidities, with analysis results providing strong support for lowering blood pressure to SBP <130 mmHg and providing BP-lowering treatment to individuals with a history of cardiovascular disease, CAD, stroke, diabetes, heart failure, and chronic kidney disease [14]. Similarly, a largescale meta-analysis of randomised trials reported that a 5 mmHg reduction of SBP reduced the risk of major cardiovascular events by about 10%, irrespective of previous diagnoses of cardiovascular disease, and even at normal or high-normal blood pressure values [15]. The SUPERIOR study recorded the mean decreases from baseline in SBP/DBP (in the sitting position) at weeks 4, 8, and 12 with telmisartan/bisoprolol combination therapy as 12.87/12.01, 18.58/14.82, and 23.68/15.97 mmHg, with p-value <0.001 across all results. The study reported reductions in BP as early as week 4, which were sustained till week 12.
A phase III Indian (N=264 subjects) study examined safety, efficacy, and tolerability of the fixed-dose combination (FDC) of telmisartan 40 mg + bisoprolol 5 mg (TBP) tablets with the existing comparator FDC telmisartan 40 mg + metoprolol succinate ER 50 mg (TMS) tablets in patients with stage 1-2 HTN. It reported that the subjects receiving TBP demonstrated a statistically significant reduction of 28.00 ± 1.143 mmHg in SBP and 15.37 ± 0.710 mmHg in DBP in the seated position at the end of 12 weeks. Our study findings concur with these results, with a mean reduction of (SBP) 23.68/(DBP)15.97 mmHg [seated position] with 12 weeks of treatment with telmisartan + bisoprolol [16].
There is a paucity of literature on the effectiveness and safety of the FDC of telmisartan with bisoprolol. However, there are many studies that evaluated telmisartan and bisoprolol monotherapy.
A real-world evidence study evaluated the long-term effectiveness of bisoprolol in patients with angina and concluded that the significant long-term reduction in the risk of mortality and various cardiovascular events with bisoprolol versus other β-blockers, and drugs other than β-blockers, confirms treatment guideline recommendations that bisoprolol is particularly well suited as the first-line treatment of angina in primary care [9].
Resting heart rate is considered a strong predictor of total mortality and hospitalization due to heart failure in HTN patients. A subgroup analysis of the BISO-CAD study reported that bisoprolol can effectively reduce resting heart rate in Asian CAD patients with comorbid HTN and hence improve composite cardiac clinical outcomes without affecting their blood pressure [10].
An observational study compared bisoprolol with metoprolol in Indian patients with Stage-1 HTN. It reported that the bisoprolol group demonstrated significantly greater reductions in SBP (-19.3 ± 3.7 mmHg vs. -16.8 ± 4.1 mmHg, p=0.01) and DBP (-10.8 ± 2.6 mmHg vs. -8.9 ± 2.9 mmHg, p=0.02) compared to the metoprolol group. A higher proportion of patients in the Bisoprolol group achieved target BP (76% vs. 64%, p=0.04), and patient satisfaction was significantly higher (8.4 ± 1.1 vs. 7.9 ± 1.3, p=0.03) [17].
Bisoprolol has been reported to potentially mask the symptoms of hypoglycaemia in patients with diabetes and may rarely be associated with bradycardia and AV block [18]. However, no such events were observed in the present study, and no clinically significant changes in heart rate or ECG findings were reported during the study period.
An electronic health record-based cohort study evaluating threeyear cardiovascular outcomes of telmisartan in patients with HTN, showed that telmisartan users exhibited significantly lower visitto-visit BP variability. The adjusted 3-year MACE rate was similar between telmisartan users (4.6%) and other ARB users (4.7%, logrank P = 0.75), with comparable safety profiles [19].
A sub-analysis of a telmisartan randomised assessment study in ACE intolerant subjects with cardiovascular disease, found that the effect of telmisartan in hypertensive and non-hypertensive patients at high cardiovascular risk was not different. This posthoc analysis suggests that myocardial ischemia may be further reduced by telmisartan in hypertensive patients [20].
We observed a statistically significant reduction in serum cholesterol as well as blood glucose levels at the end of 12 weeks, which could be attributed to the positive effect of telmisartan. A study evaluating telmisartan reported that telmisartan may have favourable effects on lipid and glucose metabolism, in addition to lowering blood pressure [21].
A study to formulate a consensus statement for the utilization of bisoprolol in combination with telmisartan based on the contemporary evidence and the real-world experiences of the Indian cardiologists, observed a high preference for the combination of bisoprolol with telmisartan for use in patients with HTN with comorbidities [22].
Studies have shown that by combining two drugs with different mechanisms of action, an antihypertensive effect two to five times greater than that obtained by monotherapy is possible. Increasing the dose of monotherapy reduces coronary events by 29% and cerebrovascular events by 40%, while combining two antihypertensive agents with a different mechanism of action reduces coronary events by 40% and cerebrovascular events by 54%. Thus, the use of combination therapy provides greater protection to a target organ than increasing the dose of monotherapy [23]. A real-world evidence study in the Indian population with essential hypertension reported that in the telmisartan monotherapy group, mean SBP/DBP was significantly reduced by 13.3/7.2 mmHg [24]. An Indian study reported a significant reduction in SBP (-19.3 ± 3.7) mmHg and DBP (-10.8 ± 2.6 mmHg) with bisoprolol at the end of 12 weeks [17]. Our study with the combination therapy of telmisartan (40mg) + bisoprolol (2.5/5mg) recorded a statistically significant reduction in mean SBP/DBP of 23.68/15.97 mmHg (sitting), 23.75/15.31 mmHg (supine), and 22.79/14.3 mmHg (standing) at the end of 12 weeks. It is evident that the reductions in BP with combination therapy are greater than those of monotherapy.
An Indian study reported a significant reduction in SBP (-19.3 ± 3.7) mmHg and DBP (-10.8 ± 2.6 mmHg) with bisoprolol at the end of 12 weeks [17].
The present study did not report any adverse events during the study period.
Study limitations
This study has certain limitations, including a relatively small sample size, lack of a comparator group, and short follow-up duration, which may limit the generalizability of the findings and evaluation of long-term safety outcomes. Larger prospective studies with longer follow-up are needed to confirm the long-term safety and cardiovascular benefits of this combination therapy.
Conclusion
The study shows that the telmisartan (40 mg)–bisoprolol (2.5/5 mg) fixed-dose combination provides effective blood pressure control and good tolerability in Indian patients with CAD and uncontrolled hypertension. The complementary mechanisms of RAAS blockade and β1-selective inhibition, along with the FDC approach, may enhance cardiovascular protection, treatment adherence, and therapeutic convenience.
Funding: Mankind Pharma
Conflict of Interest: Dr Nitin Zalte is an employee of Mankind Pharma. All other authors declare no conflict of interest related to this study.
Citations
- Shahjehan RD, Sharma S, Bhutta BS (2024) Coronary Artery Disease. In: StatPearls.
- Stark B, Johnson C, Roth GA (2024) Global prevalence of coronary artery disease: an update from the global burden of disease study. Journal of the American College of Cardiology. 83: 2320-2320.
- Chen Y, Zhou ZF, Han JM, Jin X, Dong ZF, et al. (2022) Patients with comorbid coronary artery disease and hypertension: a cross-sectional study with data from the NHANES. Annals of Translational Medicine. 10:745.
- Rosendorff C, Lackland DT, Allison M, Aronow WS, Black HR, et al. (2015) Treatment of Hypertension in Patients with Coronary Artery Disease; a scientific statement from the American Heart Association, American College of Cardiology, and American Society of Hypertension. Circulation. 131:e435-e470.
- Weber T, Lang I, Zweiker R, Horn S, Wenzel RR, et al. (2016) Hypertension and coronary artery disease: epidemiology, physiology, effects of treatment, and recommendations : A joint scientific statement from the Austrian Society of Cardiology and the Austrian Society of Hypertension. Wien Klin Wochenschr. 128:467-479.
- Lewington S, Clarke R, Qizilbash N, Peto R, Collins R (2002) Agespecific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 360:1903-1913.
- Colantonio LD, Booth JN, Bress AP, Whelton PK, Shimbo D, et al. (2018) 2017 ACC/AHA Blood Pressure Treatment Guideline Recommendations and Cardiovascular Risk. J Am Coll Cardiol. 72:1187-1197.
- Rosendorff C, Lackland DT, Allison M, Aronow WS, Black HR, et al. (2015) Treatment of Hypertension in Patients with Coronary Artery Disease: A Scientific Statement from the American Heart Association, American College of Cardiology, and American Society of Hypertension. J Am Coll Cardiol. 65:1998-2038.
- Sabidó M, Thilo H, Guido G (2019) Long-term effectiveness of bisoprolol in patients with angina: A real-world evidence study. Pharmacol Res. 139:106-112.
- Chen YD, Yang XC, Pham VN, Huang SA, Fu GS, et al. (2020) Resting heart rate control and prognosis in coronary artery disease patients with hypertension previously treated with bisoprolol: a sub-group analysis of the BISO-CAD study. Chin Med J (Engl). 133:1155-1165.
- Yusuf S, Teo K, Anderson C, Pogue J, Dyal L, et al. (2008) Effects of the angiotensin-receptor blocker telmisartan on cardiovascular events in high-risk patients intolerant to angiotensin-converting enzyme inhibitors: a randomised controlled trial. Lancet. 372:1174-1183.
- Shah SN, Munjal YP, Kamath SA, Wander GS, Mehta N, et al. (2020) Indian guidelines on hypertension-IV (2019). J Hum Hypertens. 34:745-758.
- Strauss MH, Hall AS, Narkiewicz K (2021) The Combination of BetaBlockers and ACE Inhibitors Across the Spectrum of Cardiovascular Diseases. Cardiovascular Drugs and Therapy. 37: 757–770.
- Ettehad D, Emdin CA, Kiran A, Anderson SG, Callender T, et al. (2016) Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet. 387:957-967.
- Blood Pressure Lowering Treatment Trialists’ Collaboration (2021) Pharmacological blood pressure lowering for primary and secondary prevention of cardiovascular disease across different levels of blood pressure: an individual participant-level data meta-analysis. The Lancet. 397:1625-1636.
- Wander GS, Ram B, Kumar Sonkar S, Manjunath CN, Kamath P, et al. (2024) Comparison of the efficacy, safety, and tolerability of the FDC of telmisartan + bisoprolol with telmisartan + metoprolol succinate ER combination therapy for stage 1 and stage 2 hypertension: A doubleblind, multicentric, phase-III clinical study. Indian Heart J. 76:159-166.
- Kumar ANV, Vangoori Y, T Sanjeeva Kumar Goud (2025) Comparative Efficacy and Tolerability of Bisoprolol and Metoprolol in Patients with Stage-1 Hypertension: An Observational Study. European Journal of Cardiovascular Medicine. 15:110-116.
- Bazroon AA, Alrashidi NF (2023) Bisoprolol. In: StatPearls.
- Yum Y, Kim JH, Joo HJ, Kim YH, Kim EJ (2024) Three-Year Cardiovascular Outcomes of Telmisartan in Patients With Hypertension: An Electronic Health Record-Based Cohort Study. American Journal of Hypertension. 37:429-437.
- Foulquier S, Böhm M, Schmieder R, Sleight P, Teo K, et al. (2014) Impact of telmisartan on cardiovascular outcome in hypertensive patients at high risk: a Telmisartan Randomised AssessmeNt Study in ACE iNtolerant subjects with cardiovascular Disease subanalysis. J Hypertens. 32:1334-1341.
- Inoue T, Fujimoto S, Marumoto T, Kitagawa T, Ishida K, et al. (2021) Therapeutic Potential of Vortioxetine for Anxious Depression: A Post Hoc Analysis of Data from a Clinical Trial Conducted in Japan. Neuropsychiatr Dis Treat. 17:3781-3790.
- Sawhney JPS, Makkar J, Solanki D, Guha S, Kaul U, et al. (2023) 213 Consensus towards the utilisation of bisoprolol in combination with telmisartan in indian patients with hypertension. Heart, 109.
- Guerrero-García C, Rubio-Guerra AF (2018) Combination therapy in the treatment of hypertension. Drugs Context.7: 212531.
- Khan MY, Pandit S, Abdulkutty J, Navasundi G, Hazra PK, et al. (2021) Effectiveness of Telmisartan on Blood Pressure Control in Hypertensive Patients in India: A Real-World Retrospective Study from Electronic Medical Records. Cardiol Ther. 10:255-269.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.