A Hypothesis of the Unique Pathogenesis of a Horizontal Atitudinal Visual Field Defect
by Christian Albrecht May1*, Paul Rutkowski2
1Department of Anatomy, Medical Faculty Carl Gustav Carus, TU Dresden, Germany.
2Retired ophthalmologist from New York, USA.
*Corresponding Author: Christian Albrecht May, Department of Anatomy, Medical Faculty Carl Gustav Carus, TU Dresden, Germany.
Received Date: 11 March 2026
Accepted Date: 06 April 2026
Published Date: 08 April 2026
Citation: May CA, Rutkowski P (2026) A Hypothesis of the Unique Pathogenesis of a Horizontal Atitudinal Visual Field Defect. Ophthalmol Res Rep 10: 175. DOI: https://doi.org/10.29011/2689-7407.100175
Abstract
In a minority of adults, a laminar arterial circle perfused by a single laminar paraoptic artery or a ciloretinal artery may occur. These two different choroidal vascular variations arise during the 4th month of fetal gestation and result from either a marked elevated SpO2 (single laminar paraoptic artery) or depressed SpO2 levels (cilioretinal artery). A single laminar paraoptic artery divides laminar perfusion into a superior and an inferior hemisphere. A cilioretinal artery arising from a short posterior ciliary artery or the arterial circle of Zinn-Haller perfuses a section of the optic nerve head and adjacent retina. Our hypothesis is that in older adults increasing laminar hypoxia causes ischemia to nerve fiber axons in one laminar hemispheres that leads clinically to a horizontal altitudinal visual field (HAVF) defect. If the nerve fiber axons in both laminar hemispheres become ischemic a double HAVF defect occurs. This vascular variation can be detected in aging adults with normal visual fields using the Pixlr Al Powered Photo Editor’s color program in conjunction with a fundus photograph.
What is already known on this topic
In adults an arterial circle of Zinn-Haller perfused by a single laminar paraoptic artery was demonstrated by four authors in the 1990’s [1-4].
What this study adds:
A Pixlr Al Powered Photocolor Program noninvasive procedure and a fundus photograph gives one the ability to detect indirectly in older adults with normal visual field an intraocular optic nerve
(IOON) perfused by a single laminar and prelaminar paraoptic artery.
1. Fetal Hypoxic levels and the fetal hypoxic crisis
The fetal hypoxic level is measured indirectly by subtracting the measurable fetal SpO2 level from 100%. In general, the Sp02 levels remains low in the 1st and 3rd trimester of pregnancy (Figure #1) [5,6]. However, beginning in the 2nd trimester there is a spike in the Sp02 level from a low of 20% to 60% followed by lower SpO2 levels in the third trimester until shortly before birth.
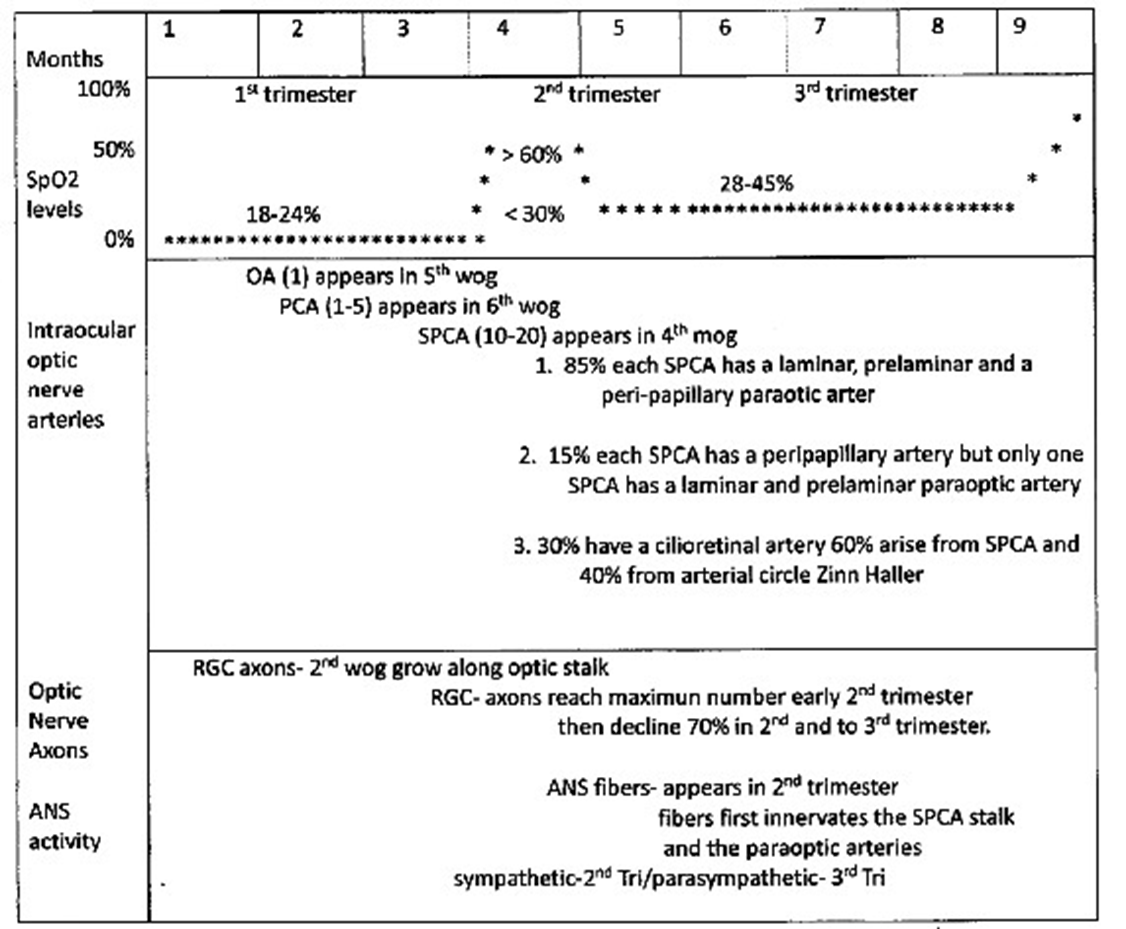
Figure 1: Time Line Fetal Intraocular Optic Nerve Development; SPCA-Short Posterior Ciliary Artery; RGC- Retinal Ganglion Cell; OA- Ophthalmic Artery; ANS- Autonomic Nervous System; PCA- Posterior Ciliary Artery; SpO2- Oxygen Saturation Level.
2. Fetal and adult choroidal blood perfusion
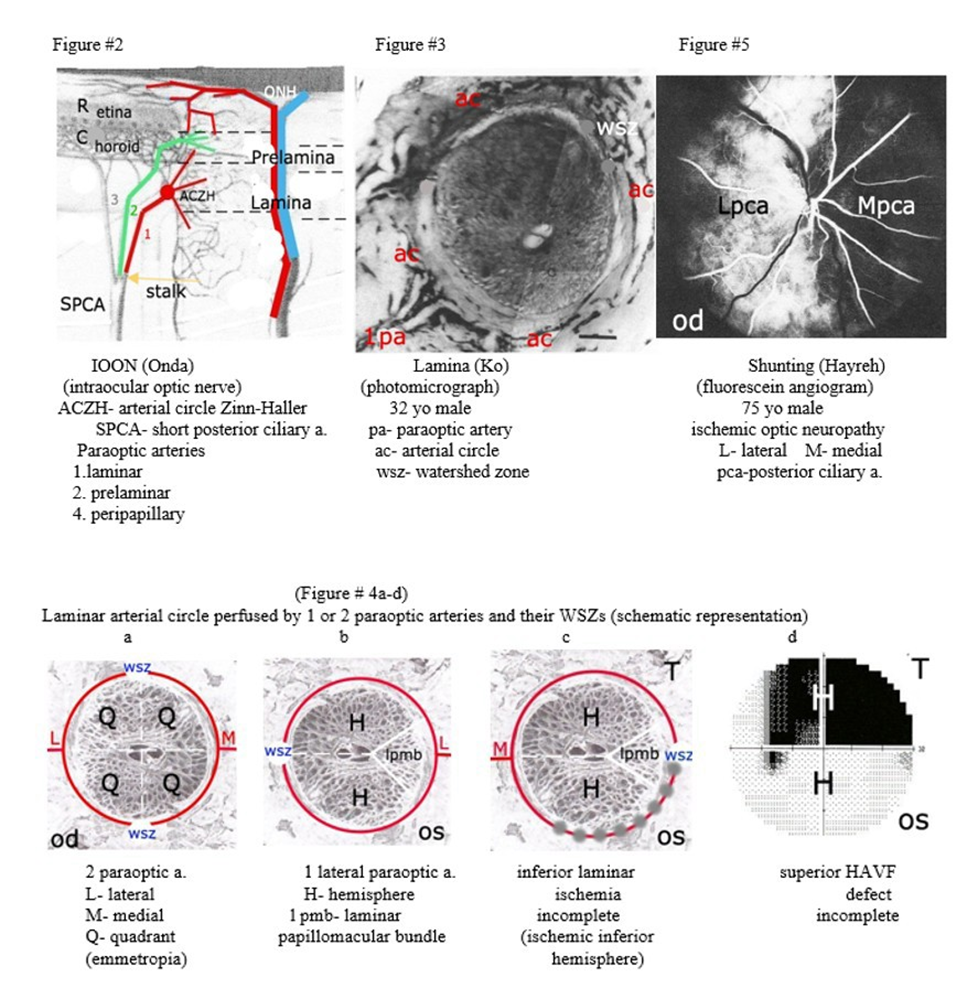
The ophthalmic artery (OA) appears in the 5th week of gestation (wog) [7] and branches into 1 to 5 posterior ciliary arteries (PCAs) in the 6th wog [8]. The multiple PCAs branch into 10 to 20 short posterior ciliary arteries (SPCAs) in the 4th month of gestation (mog) [9] that connect to the developing choriocapillaris. Two of the SPCAs, the medial and lateral, form three paraoptic arteries. A laminar paraoptic artery perfuses the arterial circle of Zinn-Haller and posterior prelamina, a prelaminar paraoptic artery perfuses the remainder of the prelamina and a peripapillary paraoptic artery perfuses the peripapillary choriocapillaris (Figure #2) [10]. In adults a single paraoptic artery perfusing the laminar arterial circle was demonstrated by 4 authors in the 1990’s. Two authors [1,2] used corrosion casting technique in human cadaver eyes, one [3] used indocyanine green in patients with pathologic myopia and a fourth [4] performed histopathological examination of the arterial circle of Zinn-Haller (Figure #3).
3. Fetal Neuronal Development
In the 2nd wog retinal ganglion cell (RGC) axons grow in parallel along the optic stalk towards the brain representing a relative retinal topography [11]. Macular nerve fiber axons occur temporally in the optic nerve head rim, the prelamina and the lamina [12]. The number of RGC axons reach their apogee early in the 2nd trimester and then decrease (70%) in number [13]. Also, in this second trimester sympathetic nerve fibers innervate the PCA’s, the medial and lateral SPCA (Figure #4) along with the paraoptic arteries [14]. Sympathetic nerve activity becomes prominent in the 2nd trimester and parasympathetic activity in the 3rd trimester of pregnancy [15].
4. Watershed Zones and the Arterial Circle of ZinnHaller
A watershed zone (WSZ) is the common capillary bed perfused by two different end arteries and is the weak link in tissue perfusion [16]. A simple laminar WSZ of the arterial circle has no anastomoses and is at greater risk for ischemia than a prelaminar complex WSZ with dense anastomoses [17]. Two laminar paraoptic arteries perfusing the arterial circle divide laminar perfusion into 4 quadrants with two simple vertical WSZs (Figure #4a). In a minority of humans, a single laminar paraoptic artery perfuses the arterial circle and divides laminar perfusion into a superior and an inferior hemisphere. Its one simple WSZ is located 180 degrees opposite from the perfusing paraoptic artery. This places the WSZ outside the intraocular optic nerve in the region of the peripapillary ring and peripapillary choroidal vasculature [18,19] (Figure #4b, c).
5. Clinical Picture of patients demonstrating a HAVF defect
Clinically, adult laminar ischemia presents with a superior or an inferior HAVF defect. In general, these eyes maintain good visual acuity and demonstrate minimal optic disc changes unless associated with pathologic myopia or retinal PMB visual field defects. If laminar ischemia worsens a double HAVF defect results that progresses to ischemic optic neuropathy and optic atrophy [20].
In the NAAION Decompression Trial Study [21] the incidence of a HAVF defect was 35% in the randomized group and 10% in the non-randomized group of the total visual fields performed in each group [22]. Hayreh found inferior HAVF defect in 34.9% of patients with ischemic optic neuropathy [23]. Since NAAION is a unilateral event, the fellow eye can be used as a control [24].
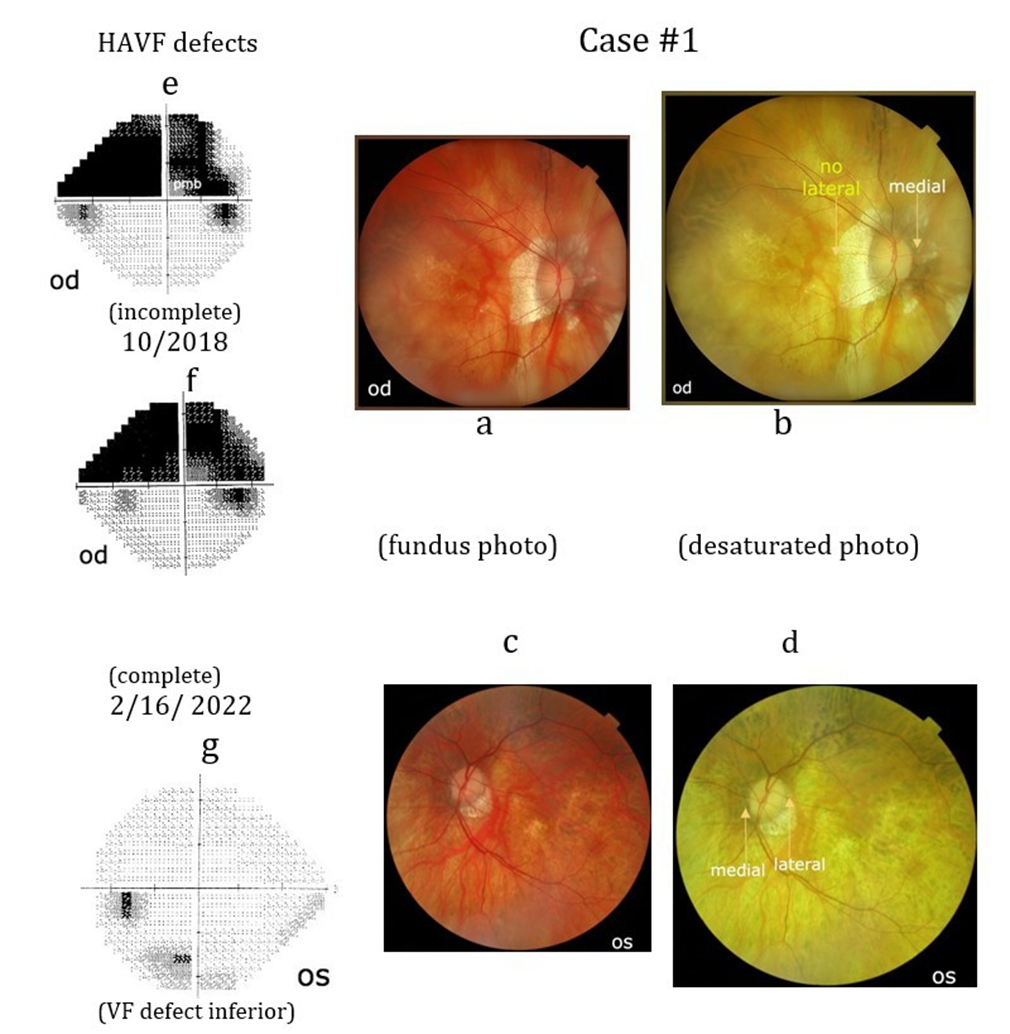
Using the Pixlr Al Program and manipulating the Hue and Vibrance settings one can obtain in older adults a desaturated fundus photograph showing the peripapillary choroidal vascular pattern around the ONH and posterior pole of the eye. The loss of this peripapillary vascular pattern adjacent to the ONH is best demonstrated in pathologic myopia (Case#1), next are adult eyes with a normal axial length (Case#2) and poorest results in acute ischemic optic neuropathy (Case #4). The adult patients are divided into 3 group: Group A. a single HAVF defect in one eye, Group B. a double HAVF defect in one eye and Group C. a visual field defect involving 3 or more quadrants in one eye. The intraocular pressure was within normal limits in all 6 cases.
Group A:
Case #1: A 57-year-old female demonstrated a superior HAVF in her right eye (Case#1e). She also demonstrated a minor inferior visual field defect in her left eye. Her vision was 20/20 in the right eye and 20/50 in the left eye secondary to recurrent heretic keratitis and laser treated myopic retinal macular edema. She had pathological myopia greater in her right eye (axial length 33.7mm) and bilateral pseudophakia with a flattened oblong ONH in both eyes. Her right lateral peripapillary choroidal pattern is absent (Case#1b).
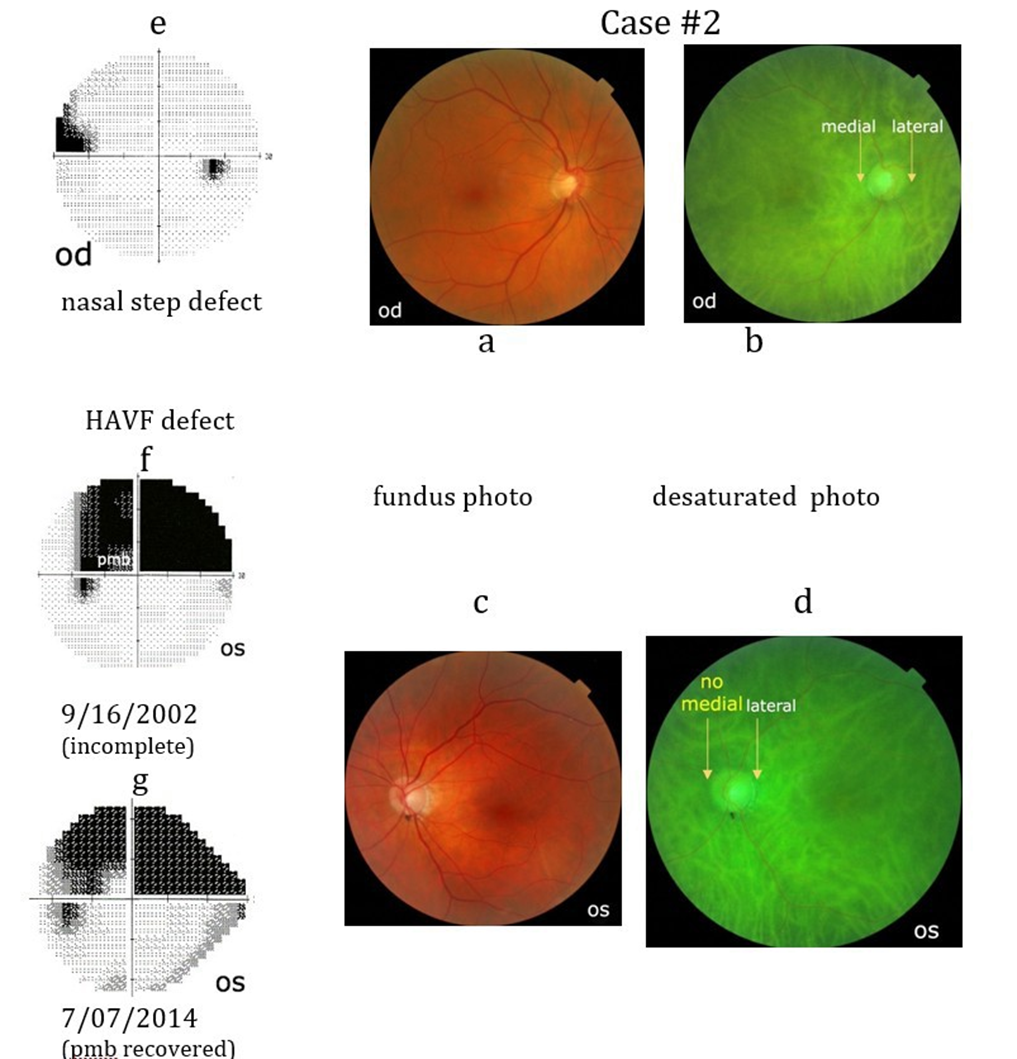
Case #2: A 61-year-old female demonstrated a superior HAVF defect in her left eye (Case#2f). Initially, the HAVF defect involved her left papillomacular bundle (PMB). She had a superior nasal step visual field defect in her right eye (Case#2e). Her vision was 20/20 with moderate myopia in each eye and thinning of the inferior temporal left OH rim. Her past history included 2 myocardial infarction and 9 coronary artery stents insertion. Her left peripapillary choroidal vascular pattern artery is absent (Case# 2d).
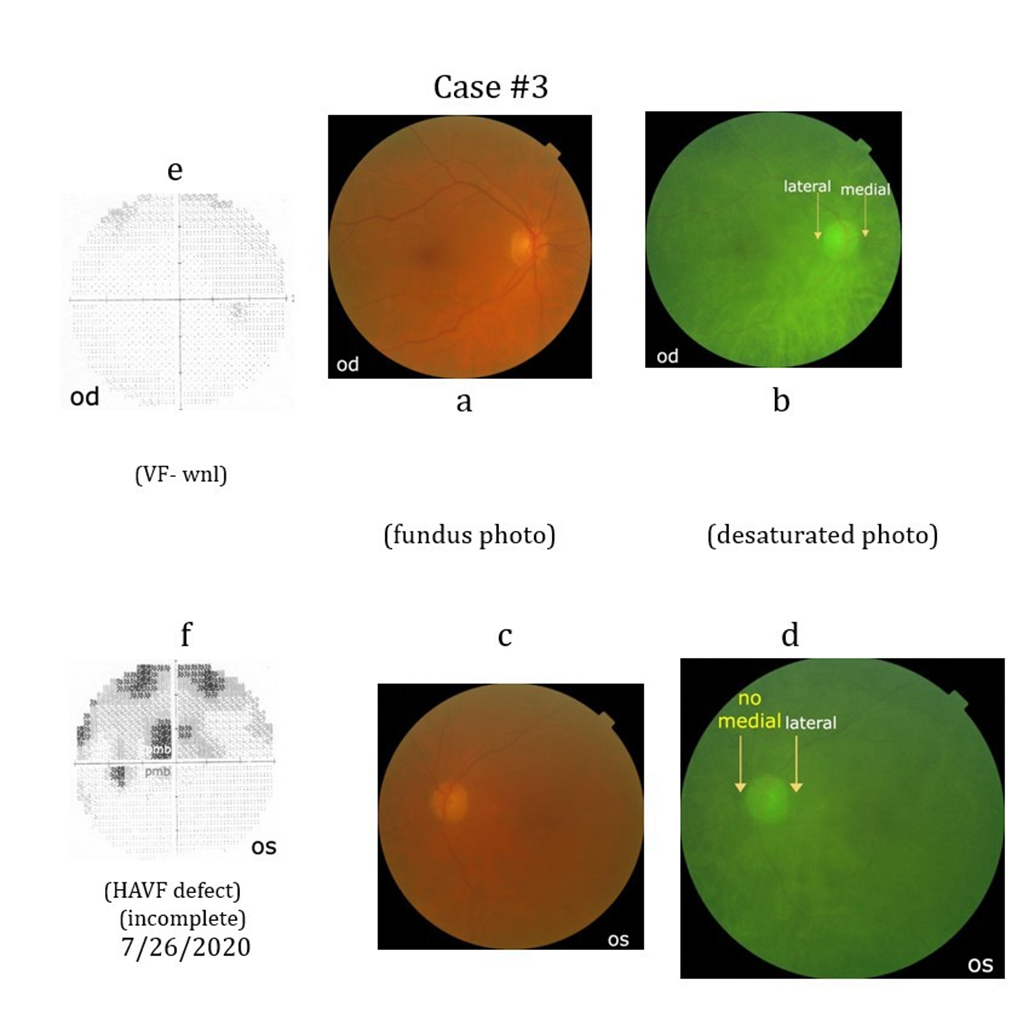
Case #3: A 67-year-old female underwent an angioplasty resulting in multiple emboli involving her left eye. Seven years after the angioplasty she demonstrated a superior HAVF defect with involvement of the superior and inferior PMB (Case# 3f). The visual field in her right eye was within normal limits (Case#3e). Her visual acuity in the left eve varied from 20/80 to 20/200 seeing only the bottom of numbers on the visual acuity chart. She had a cataract greater in the left eye than the right eye. Her left medial peripapillary choroidal vascular pattern is absent (Case# 3d).
Group B:
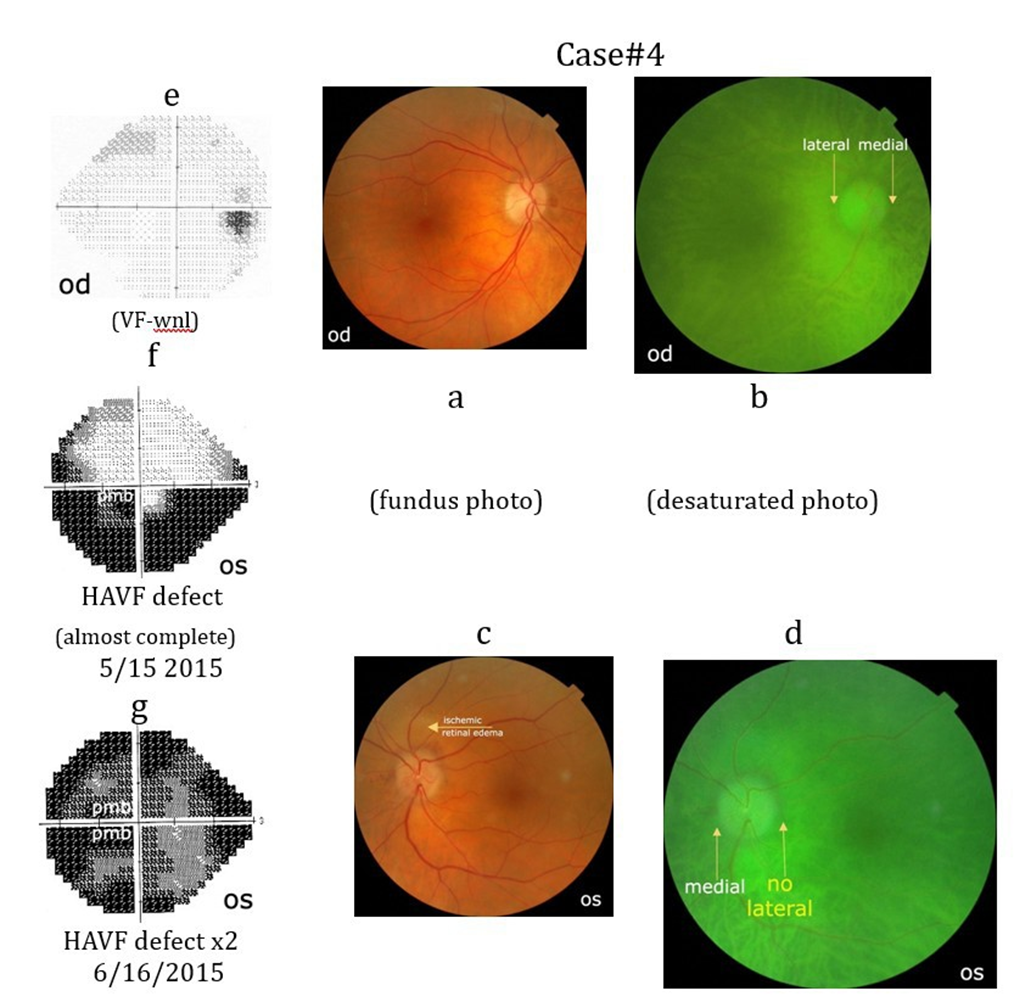
Case# 4: An 84-year-old female presented with an inferior HAVF defect (Case#4f) in her left eye upon awakening. One month later it progressed to a superior HAVF defect (Case#4g). Her visual acuity was initially 20/20 in each eye. She had a hyperemic swollen left ONH and pale retinal edema adjacent to the superior ONH margin. A fluorescein angiogram performed 6 days later demonstrated a patent superior and inferior retinal arterioles with staining of the ONH. She was on no medications and otherwise healthy. Three months later the vision in her left eye was 20/300 with marked optic atrophy. Her left lateral peripapillary vascular pattern is absent (Case#4d).
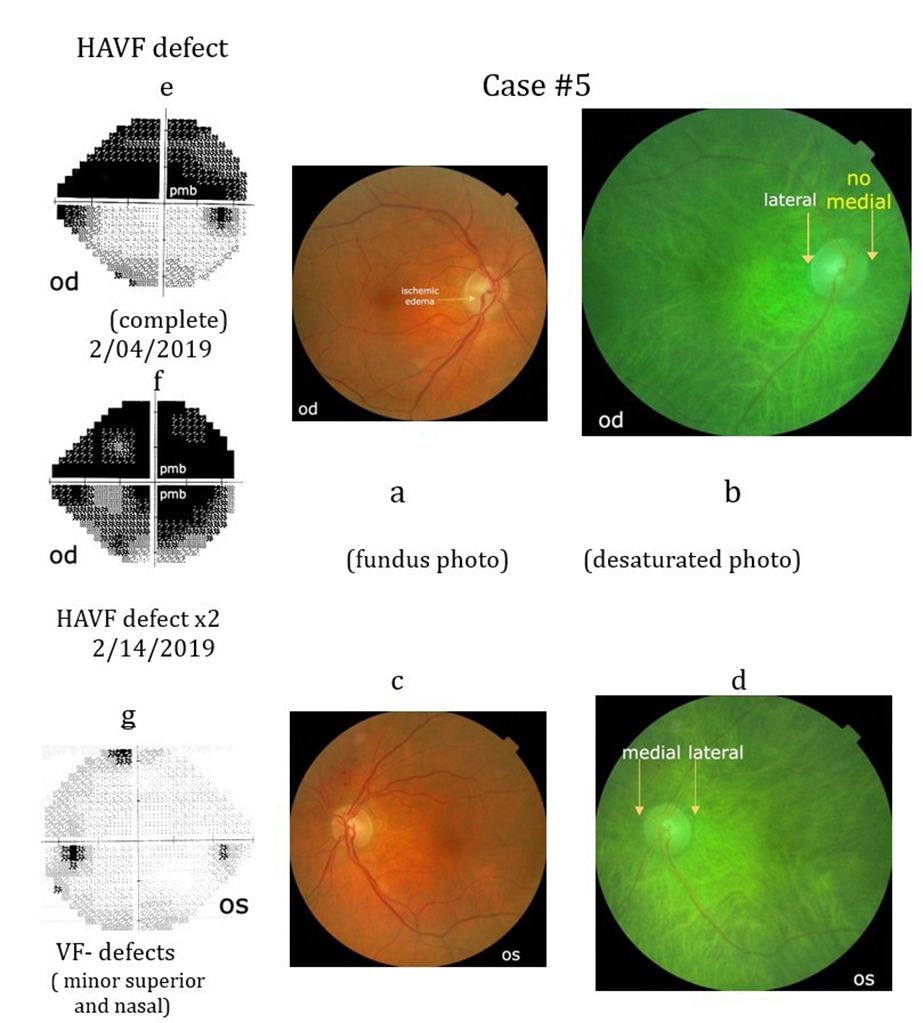
Case #5: An 84-year-old male presented with a superior HAVF (Case #5e) defect that progressed 1 month later to and inferior HAVF defect (Case#5f). Eye exam demonstrated initially a visual acuity of 20/20 in each eye with subtle ischemic swelling of the inferior rim of the right ONH (Case#5a).His initial blood pressure was 125/85, his c-reactive protein < 0.28, erythrocyte sedimentation rate 2mm/ hr and a blood sugar of 345 mg/dl. He had a history of atrial fibrillation, myocardial infarction with cardiac arrest in 2014 and diabetes mellitus type 2 since 2017. CTA of his neck with contrast showed some atherosclerosis of internal carotid arteries with only 20% occlusion on the right side. His right medial peripapillary choroidal vascular pattern is absent (Case#5b).
Group C:
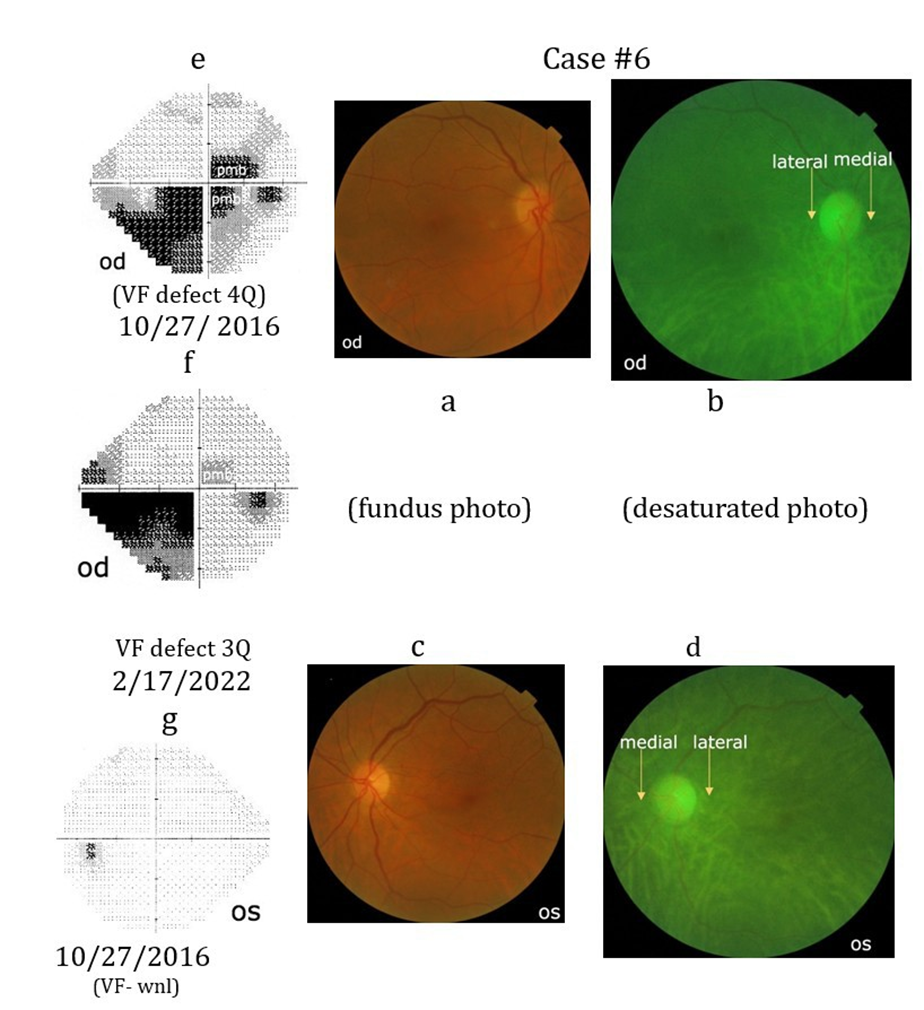
Case #6. A 76-year-old female presented with a visual field defect involving all 4 quadrants and the superior and inferior PMB of her right eye (Case#6e). Two days earlier she developed pain in her right eye during insertion of a carotid stent for a 95% occluded right carotid artery. Her visual acuity in this eye was 20/40. Five years later there was an improvement in her visual field defects, particularly her PMB (Case#6f). Her visual acuity in her right eye improved from 20/50 to 20/25 after cataract extraction. Her visual acuity in her left eye remains 20/20 and her visual field was within normal limits. She had a past history of elevated cholesterol (380-412mg) and an aortic valve repair. In both eyes there is no loss of the peripapillary choroidal vascular pattern (Case#6b, d).
Conclusions
The unique pathogenesis of a HAVF defect begins with the fetal development of a choroidal vascular variation creating two laminar hemispheres and ends with adult laminar ischemia involving one or two of the laminar hemispheres. During the 1st and 2nd trimester of pregnancy a variable number of posterior ciliary arteries (one to five) and of short posterior ciliary arteries (ten to twenty) develop. We believe that two different fetal choroidal vascular variations can also occur in a minority of humans during the second trimester of pregnancy [24]. They result from either markedly depressed or elevated levels of SpO2 in the fetus. The depressed SpO2 levels are associated with cilioretinal artery development from a SPCA or the arterial circle of Zinn Haller to enhance oxygenation of the ONH rim and adjacent retina. The elevated SpO2 levels are associated with the stimulation of sympathetic nerve fibers that block the development of one laminar and prelaminar paraoptic artery from either the medial or the lateral SPCA stalk. We found no involvement of the third peripapillary paraoptic artery during this fetal hypoxic crisis. Research performed in the 1990’s demonstrated that a minority of adult humans develop one laminar and prelaminar paraoptic artery that divides laminar perfusion into 2 hemispheres. A HAVF defect can only occur if laminar perfusion is divided into hemispheres instead of quadrants. Accompanying this visual field defect is loss of the peripapillary choroidal vascular pattern adjacent to the nasal or temporal ONH margin (Case #2d). This loss occurs in the region of the simple laminar WSZ and signifies that a laminar and prelaminar paraoptic arteries are present on the contralateral side of the IOON (Figure #4b, c). Further laminar ischemia leads to a double HAVF defect and ischemic optic neuropathy.
In our cases, adult laminar ischemia occurs from marked paraoptic artery elongating (Case #1), cardiac ischemia (Cases #2, #5), emboli from surgical intervention (Cases #3, #6) and probable sleep apnea (Case #4).
It appears that reflex shunting of laminar perfusion is important (Cases #1, #2, #3) and prevents loss of nerve fiber axons in the uninvolved laminar hemisphere. Shunting of perfusion between the lamina and the prelamina probably occurrs before the development of a double HAVF defect and ischemic optic neuropathy (Cases #4, #5). Shunting of perfusion at the level of the posterior ciliary artery [15] was demonstrated in early ischemic optic neuropathy (Figure #5). Shunting of perfusion with improvement of the superior PMB visual field defect occurs in the left eye (Case #2) and the inferior retinal PMB defect in the right eye (Case#6).
In older adults a silent fetal vascular variation may be demonstrated indirectly using a fundus photograph and the Pixlr AI powered Editor’s Program as demonstrated in our manuscript.
References
- Olver JM, Spalton DJ, McCartney AC (1994) Quantitative morphology of human retrolaminar optic nerve vasculature. Invest Ophthalmol Vis Sci 35: 3858-3866.
- Onda E, Cioffi GA, Bacon DR, Van Buskirk EM (1995) Microvasculature of the human optic nerve. Am J Ophthalmol 120: 92-102.
- Ohno-Matsui K, Futagami S, Yamashita S, Tokoro T (1998) ZinnHaller arterial ring observed by ICG angiography in high myopia. Br J Ophthalmol 82: 1357-1362.
- Ko MK, Kim DS, Ahn YK (1999) Morphological variations of the peripapillary circle of Zinn-Haller by flat section. Br J Ophthalmol 83: 862-866.
- Burton GJ, Cindrova-Davies T, Yung HW, Jauniaux E (2021) Hypoxia and reproductive health: Oxygen and development of the human placenta. Reproduction 161: 53- 65.
- Filippi L, Pascarella F, Pini A, Cammalleri M, Bagnoli P et al. (2023) Fetal Oxygenation from the 23rd to the 36th Week of Gestation Evaluated through the Umbilical Cord Blood Gas Analysis. Int J Mol Sci 24: 12487.
- Francois J, Neetens A (1954) Vascularization of the optic pathway. I. Lamina cribrosa and optic nerve. Br J Ophthalmol 38: 472-488.
- Hayreh SS (2004) The Weisenfeld Lecture: Posterior Ciliary Artery Circulation in Health and Disease. Invest Ophthalmol Vis Sci 45: 749457.
- Amalric P (1991) Choroidal vascular ischaemia. Eye 5: 519-527.
- Mackenzie PJ, Cioffi GA (2008) Vascular anatomy of the optic nerve head. Can J Ophthalmol 43: 308-312.
- Rager G (1983) Structural analysis of fiber organization during development. Prog Brain Res 1: 3139.
- FitzGibbon T (1997) The human fetal retinal nerve fiber layer and optic nerve head: a DiI and DiA tracing study. Vis Neurosci 14: 433-447.
- Provis JM, van Driel D, Billson FA, P Russell et al. (1985) Human fetal optic nerve: overproduction and elimination of retinal axons during development. J Comp Neurol 239: 92-100.
- Schneider U, Bode F, Schmidt A, Nowack S, Rudolph A et al. (2018) Developmental milestones of the autonomic nervous system revealed via longitudinal monitoring of fetal heart rate variability. PLoS One 13: e0200799.
- Assali NS, Brinkman CR, Woods JR, Dandavino A, Nuwayhid B (1997) Development of neurohumoral control of fetal, neonatal, and adult cardiovascular functions. Am J Obstet Gynecol 129: 748-759.
- Hayreh SS (1990) In vivo choroidal circulation and its watershed zones. Eye Lond 2: 273-289.
- May CA, Rutkowski P (2019) The Horizontal Raphe of the Human Retina and its Watershed Zones. Vision 3: 60.
- Jonas JB, Holbach L, Panda-Jonas S (2013) Peripapillary arterial circle of Zinn-Haller: location and spatial relationships with myopia. PLoS One 8: e78867.
- Jonas JB, Holbach L, Panda-Jonas S (2014) Peripapillary ring: histology and correlations. Acta Ophthalmol 92: 273-279.
- Patel HR, Margo CE (2017) pathology of Ischemic Optic Neuropathy. Arch Pathol Lab Med 141: 162-166.
- Newman NJ, Scherer R, Langenberg P, Kelman S, Feldon S, et al. (2002) The fellow eye in NAION: report from the ischemic optic neuropathy decompression trial follow-up study. Am J Ophthalmol 134: 317-328.
- Scherer RW, Feldon SE, Levin L, Langenberg P, Katz J, et al. (2008) Visual fields at follow-up in the Ischemic Optic Neuropathy Decompression Trial: evaluation of change in pattern defect and severity over time. Ophthalmology 15: 1809-1807.
- Hayreh SS, Podhajsky P (1979) Visual field defects in anterior ischemic optic neuropathy. Doc. Ophthalmol Proc 19: 53-57.
- Watanabe T, Kasahara K, Futagami S, Fang Y, Du R, et al. (2019) Cilioretinal Arteries and Cilioretinal Veins in Eyes with Pathologic Myopia. Sci Rep 9: 2451.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.