A Drug Utilization Study To Assess The Usage Of The Fixed-Dose Combination (FDC) Of Teneligliptin + Dapagliflozin In Indian Patients With Type2 Diabetes Mellitus (T2DM) At Various Clinics Across India
by Vijay Neglur1, Narasimman VKR2, Chavi Agarwal3, Shivarama Krishna4, Aditya Verma5, Sumit Bhushan6, Sanjay Choudhari6, Rahee Borulkar6, Kiransing Pawar6, Saiprasad Patil6
1Dr. Negalur's Diabetes & Thyroid Center, Mumbai, India
2VKR Diabets Centre, Chennai, India
3Fortis Escorts Hospital, Delhi, India
4CC Shroff Hospital, Hyderabad, India
5Calcutta Heart Research Centre, Kolkata, India
6Glenmark Pharmaceuticals Ltd, Mumbai, India
*Corresponding author: Sumit Bhushan, Glenmark Pharmaceuticals Ltd, Mumbai, India 400 099.
Received Date: 03 March, 2026
Accepted Date: 10 March, 2026
Published Date: 12 March, 2026
Citation: Neglur V, Narasimman VKR, Agarwal C, Krishna S, Verma A, et al. (2026) A Drug Utilization Study To Assess The Usage Of The Fixed-Dose Combination (FDC) Of Teneligliptin + Dapagliflozin In Indian Patients With Type2 Diabetes Mellitus (T2DM) At Various Clinics Across India. Curr Trends Intern Med 10: 252. DOI: https://doi.org/10.29011/2638-003X.100252
Abstract
Fixed dose combination (FDC) of teneligliptin, a dipeptidyl peptidase-4 (DPP 4) inhibitor and dapagliflozin, a sodium-glucose cotransporter-2 (SGLT2) inhibitor has demonstrated benefits for glycaemic control and glycaemic variability among Indian patients with type 2 diabetes mellitus (T2DM). Despite these encouraging findings, evidence on real world prescribing patterns and utilization of this FDC in Indian clinical practice remains limited. This multicentre, retrospective study aimed to evaluate the real-world usage of FDC of teneligliptin and dapagliflozin in Indian patients with T2DM from clinical data collected from outpatient medical records of T2DM patients prescribed with Teneligliptin + Dapagliflozin FDC (TEN/DPA FDC) across 150 sites. The primary outcome was the assessment of treatment and clinical usage patterns of TEN/DPA FDC among the patients. A total of 750 patients with mean age of 55.03±10.74 years, majority (63.20%) males and diabetes duration of 6.79±5.29 years. The most frequent comorbidities were obesity, cardiovascular disease and dyslipidaemia. The patients had a suboptimal glycaemic control and about one-fifth of the patients had experienced a hypoglycaemic event in the last one year. About a quarter (~33%) of patients who were prescribed TEN/DPA FDC were treatment naïve and the over two-thirds (~67%) had switched from other antidiabetics. Among the switchers, sulfonylureas plus metformin (34%), metformin monotherapy (~22%), DPP‑4i monotherapy (10%) & DPP-4i plus metformin (8.7%) were most common background therapies that were switched to TEN/DPA FDC. The findings show that patients prescribed with TEN/DPA FDC represent a high-risk T2DM population and used for early intensification and regimen simplification.
Keywords: Teneligliptin, Dapagliflozin, Drug utilization, Type 2 diabetes Mellitus
Background
Type 2 diabetes mellitus (T2DM) is a progressive metabolic disorder characterized by chronic hyperglycemia, which, if inadequately controlled, leads to microvascular and macrovascular complications including nephropathy, retinopathy, neuropathy, and cardiovascular disease [1,2]. Achieving and maintaining optimum glycemic control is therefore critical to reduce long term morbidity and mortality [3]. However, the progressive decline in β cell function in patients with T2DM necessitates timely intensification of antidiabetic therapy and careful selection of drug combinations tailored to patient needs [4,5].
Among available antidiabetic agents, dipeptidyl peptidase-4 (DPP 4) inhibitors improve glycemic control by enhancing incretin activity, thereby stimulating glucose dependent insulin secretion and suppressing glucagon release, with a favorable safety profile and low risk of hypoglycemia [6,7]. Sodium-glucose cotransporter-2 (SGLT2) inhibitors, act independently of insulin by promoting urinary glucose excretion, and have demonstrated additional benefits in reducing body weight, blood pressure, and cardiovascular and renal risk [8,9]. Combining these two antidiabetic classes offers complementary mechanisms of action, making them an attractive option for patients inadequately controlled on monotherapy or metformin [10]. Substantial evidence demonstrates that combining SGLT2 inhibitors with DPP‑4 inhibitors offers superior glycemic control and weight reduction without additional hypoglycemia risk, while maintaining a favorable safety profile consistent with the known class effects [11, 12]. A recent expert consensus from Indian endocrinologists emphasized the clinical utility of this dual therapy in patients with uncontrolled T2DM, especially those with cardiovascular or renal comorbidities [13].
Teneligliptin, a potent third‑generation DPP‑4 inhibitor, provides sustained 24‑hour enzyme inhibition and stable glycemic control, with dual hepatic and renal clearance allowing use without dose adjustment even in advanced renal disease. Beyond glycemic effects, it improves lipid profile, cardiac function, adiponectin levels, and natriuresis [7]. Its combination with dapagliflozin, an SGLT2 inhibitor, offers complementary mechanisms and favorable safety profile that make this combination a rational therapeutic option in real‑world management of T2DM. The AMPLIFY TIR study, conducted in Indian patients with T2DM, demonstrated significant improvements in 24 hour glucose profiles with this fixed dose combination (FDC), particularly in terms of time in range and glycemic variability [14]. Despite these encouraging findings, evidence on real world prescribing patterns and utilization of this FDC in Indian clinical practice remains limited, underscoring the need for further evaluation. In this study, we aimed to evaluate the real-world usage of FDC of teneligliptin and dapagliflozin in Indian patients with T2DM.
Methods
Study design and data source
This multi-centre, retrospective study collected data from the medical records of outpatients with T2DM across 150 Indian clinics from October 18, 2023 to April 10, 2024. Clinical data was collected using a predesigned structured proforma and entered into an electronic Case Report Form. The study was conducted in accordance with applicable regulations, Good Clinical Practices, and standard operating procedures. All documents, including the protocol and the data collection format, related to this retrospective study were submitted to the Independent Ethics Committee, and the study was conducted per the approved protocol.
Study Population Selection Criteria
Patients who were prescribed Zita D® (Teneligliptin 20 mg + Dapagliflozin 10 mg) FDC (henceforth referred to as TEN/DPA FDC) by treating physician were included in the study. Any patients with incomplete medical records or missing key data were excluded.
Study Outcomes
The demographic and clinical characteristics, including medical history, concomitant medications, and the duration of diabetes, of patients who were prescribed with TEN/DPA FDC were recorded. The primary outcome was the evaluation of treatment and clinical usage patterns of TEN/DPA FDC among the patients.
Data/Statistical Analysis
Descriptive statistics were used to analyse all results. Categorical variables were presented as counts and percentages, and continuous variables were reported as means with standard deviations.
Results
Demographics and Clinical Characteristics
A total of 750 patients were included with a mean±SD age of 55.03±10.74 years, majority (63.20%) were males (Table1). The mean±SD duration of diabetes was 6.79±5.29 years, and mean diastolic and systolic blood pressure (BP) was 87.83±11.36 and 141.06±19.26 mmHg, respectively. About 30% of the patients were current smokers or tobacco users. More than half (55.33%) of the patients had family history of T2DM. Nearly 21% had a history of hypoglycaemic event in the last one year.
|
Characteristic |
Statistics (N=750) |
|
Age, Years |
55.03±10.74 |
|
Gender, n (%) |
|
|
Male |
474 (63.20) |
|
Female |
255 (34.00) |
|
Unknown |
21 (2.80) |
|
Body Weight, Kg (n=722) |
74.25±13.79 |
|
Height, cm (n=637) |
163.05 ± 15.70 |
|
Blood Pressure, mmHg |
|
|
Diastolic (n=728) |
87.83±11.36 |
|
Systolic (n=731) |
141.06±19.26 |
|
Pulse Rate, bpm (n=615) |
82.44±11.55 |
|
Duration of T2DM, Years (n=575) |
6.79±5.29 |
|
Medical History |
|
|
Smoking or tobacco use, n (%) |
|
|
Yes |
228 (30.40) |
|
Duration of smoking or tobacco use, years |
12.99±8.36 |
|
Other Complaints |
|
|
Chest pain, n (%) |
120 (16.00) |
|
Number of episodes |
1.76±1.32 |
|
Shortness of breath, n (%) |
112 (14.93) |
|
Number of episodes |
2.42±3.22 |
|
Family History of T2DM, n (%) |
415 (55.33) |
|
Relevant Medical history, n (%) |
|
|
Limitation of physical activity |
193 (25.73) |
|
Hospitalizations due to any causes in past year |
112 (14.93) |
|
No. of hospitalization due to any cause in past year |
1.70±1.49 |
|
Hypoglycemia in past 1 year |
|
|
Number of patients, n (%) |
157 (20.93) |
|
Number of episodes |
1.34±2.19 |
|
All data are presented as mean±SD, unless otherwise mentioned. bpm, beats per minute; T2DM, type 2 diabetes mellitus. |
|
Table 1: Demographics and Clinical Characteristics of Patients Prescribed with TEN/DPA FDC
Clinical and Laboratory Parameters
The clinical and laboratory characteristics of the study population as depicted in (Table 2) show that patients who were prescribed TEN/DPA FDC had elevated mean fasting glucose (164.36 ± 47.05 mg/dL), post prandial glucose (249.98 ± 73.32 mg/dL), and HbA1c (8.19 ± 1.04%). Renal parameters were outside normal range with a mean serum creatinine of 1.38 ± 1.04 mg/dL and estimated glomerular filtration rate (eGFR) of 70.62 ± 28.69 mL/min/1.73 m2. Patients also had elevated mean total cholesterol (212.56 ± 41.89 mg/dL), LDL-C (124.53 ± 40.75 mg/dL) and triglycerides (194.11 ± 74.61 mg/dL) alongside relatively low HDL-C levels of 52.09 ± 36.27 mg/dL.
|
Parameter |
n |
Values mean±SD |
|
Blood Glucose Profile |
||
|
Fasting Blood Glucose |
639 |
164.36 ± 47.05 |
|
Post-prandial blood Glucose |
628 |
249.98 ± 73.32 |
|
HbA1C (%) |
594 |
8.19 ± 1.04 |
|
Renal Profile |
||
|
Serum creatinine |
175 |
1.38 ± 1.04 |
|
Serum albumin (g/dL) |
101 |
4.53 ± 1.18 |
|
eGFR (mL/min/1.73 m2) |
126 |
70.62 ± 28.69 |
|
Blood Urea Nitrogen |
50 |
26.39 ± 15.02 |
|
Blood/Serum Lipid Profile |
||
|
Total cholesterol |
183 |
212.56 ± 41.89 |
|
LDL cholesterol |
168 |
124.53 ± 40.75 |
|
HDL cholesterol |
163 |
52.09 ± 36.27 |
|
Triglycerides |
153 |
194.11 ± 74.61 |
|
All data are presented as mg/dL, unless otherwise specified. eGFR, estimated glomerular filtration rate; HbA1c, glycated haemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein. |
||
Table 2: Clinical Laboratory Parameters of Patients Prescribed with TEN/DPA FDC
Comorbidities
Among patients who were prescribed TEN/DPA FDC, top three comorbid conditions were obesity (44.27%), cardiovascular disease (42.40%) and dyslipidemia (33.20%) (Figure 1). Other frequent comorbid conditions included CKD (17.47%), and anxiety/depression (16.80%).
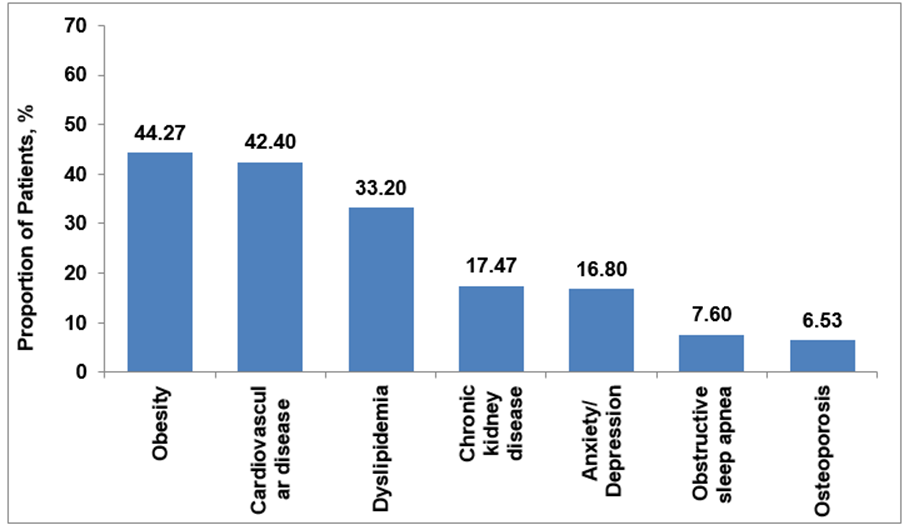
Figure 1: Comorbidities among Patients Prescribed with TEN/DPA FDC (N = 750)
Concomitant Medications
The use of concomitant medications among the study population has been depicted in (Figure 2). Antihypertensives (32.27%) were the most frequent concomitant medication among the study population followed by antidyslipidaemic drugs (19.60%). About 6% of the patients included in the study used varying combinations of anticoagulants, antiplatelets, antidyslipidemics, fibrates, and diuretics.
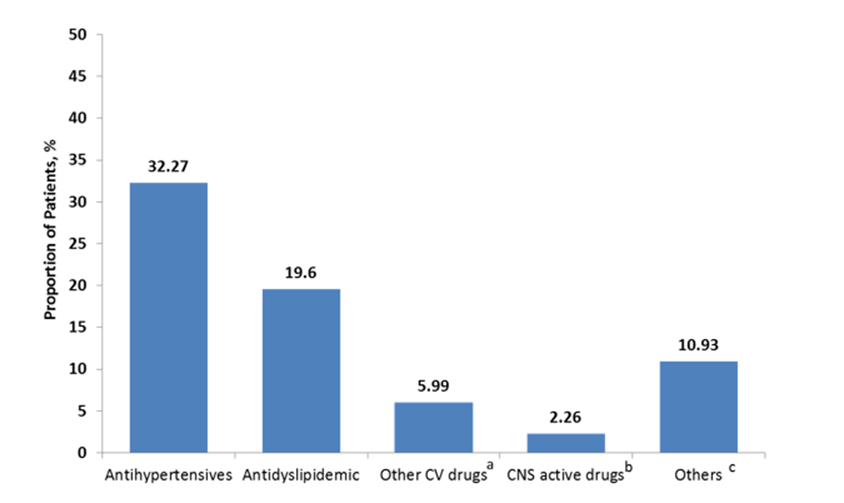
Figure 2: Concomitant Medications of Patients Prescribed with TEN/DPA FDC (N=750)
aincludes anticoagulants, antihyperlipidemic + Diuretics, antihypertensive + antihyperlipidemic, antihypertensive + Diuretics, antilipidemic, antiplatelets, antiplatelets + anticoagulants, diuretics, and fibrates; bincludes antianxiety, anticonvulsants, antidepressant, antipsychotics, and central nervous system agents + Decarboxylase Inhibitors; cincludes analgesics + anti-Inflammatory + anthraquinones + blood vessel dilator + proteoglycan synthesis stimulators, thyroid hormones, and any other medication.
CNS, central nervous system; CV, cardiovascular; FDC fixed dose combination.
Treatment Patterns among Users of TEN/DPA FDC
About a quarter (33.2%; n=249/750) of patients who were prescribed TEN/DPA FDC were treatment naïve and the over two-thirds (66.80%; n=501/750) had switched to TEN/DPA FDC from other antidiabetics. Of those who switched from other antidiabetic therapy, nearly 35% (n=177/501) were previously taking monotherapy, ~55% (n=275/501) were taking dual antidiabetic therapy and almost 10% (n=49/501) were on triple antidiabetic therapy (Figure 3).
The antidiabetic treatment patterns of patients who switched to TEN/DPA FDC have been summarized in (Table 3). Metformin, either alone or in combination, remained the most widely prescribed antidiabetic, in 21.6% (n=108/501) of the patients who switched to TEN/DPA FDC. Among monotherapy regimens, DPP‑4 inhibitors (10%, n=50/501) and sulfonylureas (1.6%, n=8/501) accounted for the next major share after metformin.
Dual‑drug combinations represented the majority of prescriptions and most predominant dual therapy was sulfonylureas (mainly glimepriride) plus metformin (34.1%, n=171/501), followed by DPP-4 inhibitors (sitagliptin, teneligliptin and vildagliptin) plus metformin (8.7%, n=44/501) and combination of DPP-4 inhibitors with SGLT2 inhibitors (dapagliflozin) (3.2%, n=16/501). Other dual combinations including metformin in combination with pioglitazone, voglibose, or SGLT2 inhibitors and combinations of DPP-4 inhibitors were prescribed less frequently (<2.5%).
Triple‑drug therapy accounted for nearly 10% of prescriptions. The most common triple regimen was glimepiride, metformin, and voglibose (4.7%, n=24/501), followed by glimepiride, metformin, and pioglitazone (4.2%, n=21/501).
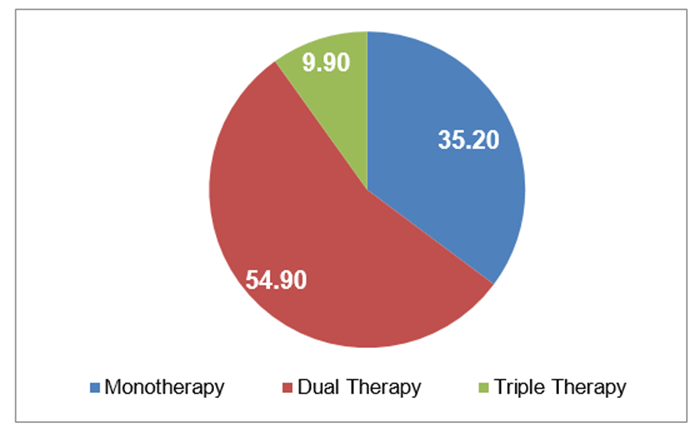
Figure 3: Treatment Patterns among Patients Switching to TEN/DPA FDC.
|
Drug |
Patients, n (%) (N=501) |
|
Monotherapy |
|
|
Metformin |
108 (21.6) |
|
Sulfonylurea Glimepiride Others |
8 (1.6) 3 (0.6) 5 (1.0) |
|
DPP-4 inhibitors Sitagliptin Vildagliptin Teneligliptin Others |
50 (9.9) 16 (3.2) 16 (3.2) 9 (1.8) 9 (1.8) |
|
Insulin |
7 (1.4) |
|
Dual Therapy |
|
|
SU + Metformin Gliclazide + Metformin Glimepiride + Metformin Others |
171 (34.1) 2 (0.4) 164 (32.7) 5 (0.9) |
|
DPP-4 inhibitors + Metformin Sitagliptin + Metformin Teneligliptin + Metformin Vildagliptin + Metformin |
44 (8.8) 16 (3.2) 9 (1.8) 19 (3.8) |
|
SGLT2 inhibitors + Metformin Dapagliflozin + Metformin |
2 (0.4) |
|
Pioglitazone + Metformin |
12 (2.4) |
|
Combination of DPP-4 inhibitors Sitagliptin + Teneligliptin Vildagliptin + Teneligliptin |
21 (4.2) 7 (1.4) 14 (2.8) |
|
DPP-4 inhibitors + SGLT2 inhibitors Dapagliflozin + Vildagliptin Sitagliptin + Dapagliflozin Teneligliptin + Dapagliflozin |
16 (3.3) 9 (1.8) 5 (1.0) 2 (0.4) |
|
Triple Therapy |
|
|
Metformin + SU + Pioglitazone Glibenclamide + Metformin + Pioglitazone Glimepiride + Metformin + Pioglitazone |
23 (4.6) 2 (0.4) 21 (4.2) |
|
Glimepiride + Metformin + Voglibose |
24 (4.8) |
|
Glimepiride + Metformin + Sitagliptin |
2 (0.4) |
|
DPP-4, dipeptidyl peptidase-4; SGLT2, sodium- glucose cotransporter-2; SU, sulfonylurea. |
|
Table 3: Antidiabetic Treatment Patterns among Patients Switched to TEN/DPA FDC
Discussion
This study aimed to characterize the demographic and clinical profile of patients who were prescribed TEN/DPA FDC, and to describe the real-world treatment patterns among outpatients in Indian clinics. The findings demonstrate that the patients who are prescribed TEN/DPA FDC in clinical practice have broad clinical characteristics such as treatment-naive or switching from other therapies, having a diverse range of clinical characteristics, including a notable prevalence of T2DM associated comorbidities and history of substance or nicotine use. These findings offer valuable insights into the profile of patients using this FDC and treatment patterns in Indian in clinical practice.
The demographics and clinical characteristics of this study population reflect a typical real-world population of patients with T2DM. The baseline profile and cardiometabolic burden (elevated systolic BP and tobacco use) mirror contemporary Indian T2DM cohorts where even newly diagnosed patients frequently meet “high/very high” ASCVD risk thresholds, driven by hypertension and smoking [15]. The mean duration of diabetes of nearly 7 years suggests that a substantial proportion of patients were in the mid-course of disease progression, a stage often associated with treatment intensification and the need for FDC therapy. Notably, more than half of the patients reported a family history of T2DM, underscoring the strong genetic predisposition in this population. Additionally, the occurrence of hypoglycemia in approximately one-fifth of patients within the past year reflects the challenges of glycemic management and the importance of balancing efficacy with safety in antidiabetic therapy. Overall, based on these characteristics the participants represented a population with multiple risk factors, comorbidities, and a clear need for optimized therapeutic strategies.
The clinical and laboratory parameters and the comorbidity profile further emphasized the high-risk profile of patients who were prescribed TEN/DPA FDC and underscores the complexity of T2DM management. Glycemic control was suboptimal, with mean fasting and post-prandial glucose levels and HbA1c exceeding recommended targets. These findings are consistent with large Indian primary care studies, where more than three quarters of patients failed to achieve optimal glycemic control despite therapy intensification [2]. Renal indices revealed early impairment evident from reduced eGFR in a subset of patients, aligning with the CITE study, which reported a high prevalence of CKD among Indian T2DM patients, particularly in those with hypertension and longer disease duration [16]. Dyslipidemia was also evident, with elevated LDL-C and triglycerides, consistent with previous reports among Indian population linking lipid abnormalities to both nephropathy and cardiovascular complications [17]. High prevalence of obesity, cardiovascular disease, and dyslipidemia reflect clustering of cardiometabolic risks that accelerate ASCVD [18]. Presence of Chronic kidney disease (CKD) underlines the burden of renal complications associated with uncontrolled or poorly controlled T2DM, consistent with Indian data on diabetic kidney disease [16]. Therapeutically, this comorbidity pattern support the use of dapagliflozin-based FDCs for their benefits in weight reduction, cardiovascular protection, and renal outcomes, while teneligliptin adds effective glycemic control with low hypoglycemia risk. Notably, considerable rates of anxiety/depression among patients highlighted the psychosocial burden in T2DM patients that emphasizes the need for integrated care addressing both metabolic and psychosocial aspects of diabetes. The prevalence of these comorbidities corroborated the use of concomitant medications including cardiovascular, antihypertensive and antidyslipidemic drugs and suggest polypharmacy among patients with T2DM highlighting the importance of simplifying regimens through rational FDC use, which can improve adherence, reduce pill burden, and optimize outcomes.
About one‑third of patients prescribed the FDC were treatment‑naïve, while two‑thirds had switched from prior therapy, predominantly from dual regimens. This reflects both the role of FDCs in early initiation and their utility in simplifying or intensifying therapy in patients already on multiple agents. The initiation of FDCs in treatment‑naïve patients suggests increasing physician confidence in starting combination therapy earlier in the disease course, consistent with Indian and international guidelines that recommend early intensification to overcome therapeutic inertia. Real‑world Indian surveys, such as the DiSi Survey, have similarly reported that FDCs of DPP‑4 inhibitors and SGLT2 inhibitors are being used both in naïve patients and as step‑up therapy, reflecting their perceived efficacy and tolerability.
Among patients who switched to TEN/DPA FDC, treatment histories revealed a clear pattern of step‑up intensification and regimen simplification. More than one third of the patients were using monotherapy indicating that a substantial proportion of patients were escalated from single‑agent therapy to combination therapy, consistent with guideline recommendations to intensify when glycemic targets are not achieved. More than half of the patients who switched to TEN/DPA FDC were taking a dual therapy, with sulfonylurea + metformin as the most frequent regimen, reflecting traditional prescribing patterns in India. The use of other combinations such as DPP‑4 inhibitors + metformin, and DPP‑4 + SGLT2 inhibitors suggests a gradual shift toward modern agents with better safety and cardiometabolic profiles. Use of triple drug regimen in nearly one tenth of patients reflect attempts at intensification but also highlight the complexity and pill burden that often drive the switch to FDCs.
This distribution mirrors recent Indian prescribing studies, which show that while a combination of sulfonylurea and metformin remains common, there is a growing preference for DPP‑4 and SGLT2 inhibitor–based regimens due to their favorable efficacy, weight neutrality, and lower hypoglycemia risk [19, 20].
This study should be interpreted in light of a few limitations. First, the data are subject to the inherent limitations of medical record abstraction, including potential for missing or incomplete information. Second, the study did not assess the glycemic parameters which limit the assessment of treatment effectiveness, appropriateness and clinical rationale behind prescription patterns in patients with T2DM.
Conclusions
The findings show that patients prescribed with TEN/DPA FDC represent a high-risk T2DM population, marked by poor glycemic control, multiple cardiometabolic comorbidities, and extensive concomitant medication use. The use of FDCs in both treatment-naïve patients and those switching from mono-, dual-, or triple-drug regimens highlights their dual role in early intensification and regimen simplification. These results reinforce the clinical value of TEN/DPA FDC as a rational option in Indian practice, supporting guideline-based, risk-driven strategies that address not only glycemic control but also broader cardiometabolic and psychosocial needs.
Acknowledgements
We would like to extend our thanks to all the institutes and respective investigators and team members for their support. We also appreciate the CRO support by Sclin Soft Technologies Private Limited, India for clinical trial management. We also thank Lifecom Scientia for medical writing support in the study.
Ethical Standards
The work presented in this study was in accordance with the study protocol, the New Drugs and Clinical Trials Rules 2019 issued by the Government of India, the ethical principles that have their origin in the Declaration of Helsinki, International Council for Harmonisation (ICH) Good Clinical Practice (GCP), and all applicable local regulatory requirements. Independent Ethics committee approval was obtained prior to study initiation and data collection.
Conflict Of Interest
Sumit Bhushan, Rahee Borulkar, Kiransing Pawar, Sanjay Choudhari and Saiprasad Patil are employees of Glenmark. All other investigators/authors have no conflicts of interest that are directly relevant to the content of this article.
Funding
This study was funded by Glenmark Pharmaceuticals Limited. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Sponsorship
This study is sponsored by Glenmark Pharmaceuticals Ltd., Mumbai, India.
Data Availability Statement
Data generated during this study are available from the corresponding author on reasonable request.
References
- American Diabetes Association Professional Practice Committee (2024) 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2024. Diabetes Care. 47: S158-S178.
- Borgharkar SS, Das SS (2019) Real-world evidence of glycemic control among patients with type 2 diabetes mellitus in India: the TIGHT study. BMJ Open Diabetes Res Care. 7 :e000654.
- Khunti K, Zaccardi F, Amod A, Aroda VR, Aschner P, et al. 25(20) Glycaemic control is still central in the hierarchy of priorities in type 2 diabetes management. Diabetologia 68: 17–28.
- Makkar BM, Kumar V, Saboo B, et al. (2022) RSSDI Clinical Practice Recommendations for the Management of Type 2 Diabetes Mellitus 2022. International Journal of Diabetes in Developing Countries. 42:S1–S143.
- Davies MJ, Aroda VR, Collins BS, Gabbay RA, Green J, et al. (2022) Management of Hyperglycemia in Type 2 Diabetes, 2022. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 45:2753-2786.
- Tsuchimochi W, Ueno H, Yamashita E, Tsubouchi C, Sakoda H, et al. (2015) Teneligliptin improves glycemic control with the reduction of postprandial insulin requirement in Japanese diabetic patients. Endocr J. 62:13-20.
- Ji L, Li L, Ma J, Li X, Li D, et al. (2021) Efficacy and safety of teneligliptin added to metformin in Chinese patients with type 2 diabetes mellitus inadequately controlled with metformin: A phase 3, randomized, double-blind, placebo-controlled study. Endocrinol Diabetes Metab. 4:e00222.
- Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, et al. (2019) Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 380:347-357.
- Heerspink HJL, Jongs N, Chertow GM, Langkilde AM, McMurray JJV, et al. (2021) Effect of dapagliflozin on the rate of decline in kidney function in patients with chronic kidney disease with and without type 2 diabetes: a prespecified analysis from the DAPA-CKD trial. Lancet Diabetes Endocrinol. 9:743-754.
- Scheen AJ (2017) Pharmacokinetic Characteristics and Clinical Efficacy of an SGLT2 Inhibitor Plus DPP-4 Inhibitor Combination Therapy in Type 2 Diabetes. Clin Pharmacokinet. 56:703-718.
- Li D, Shi W, Wang T, Tang H (2018) SGLT2 inhibitor plus DPP-4 inhibitor as combination therapy for type 2 diabetes: A systematic review and meta-analysis. Diabetes Obes Metab. 20:1972-1976.
- Molina-Vega M, Muñoz-Garach A, Fernández-García JC, Tinahones FJ (2018)The safety of DPP-4 inhibitor and SGLT2 inhibitor combination therapies. Expert Opin Drug Saf. 17:815-824.
- Manjula S, Krishna Kumar M (2024) Expert opinion on the use of SGLT2 inhibitors and its combination with a special focus on dapagliflozin in the management of type 2 diabetes in Indian settings. International Journal of Endocrinology. 6:20-6.
- Barkate HV, Petare AU, Bhushan S, Choudhari SY, Brid M, et al. (2024) 33-PUB: Triple-Arm, Prospective, Multicentre, Randomized, Open-Label, Active-Controlled Study to Assess Effect on FDC of Teneligliptin 20 mg+ Dapagliflozin 10 mg Compared with FDC of Sitagliptin 100 mg+ Dapagliflozin 10 mg and FDC of Linagliptin 5 mg+ Empagliflozin 25 mg on 24-Hour Glucose Profile Assessed by Continuous Glucose Monitoring (CGM) in Indian Patients of Type 2 Diabetes Mellitus In Real-World Setting (AMPLIFY-TIR STUDY)—Interim Report. Diabetes. 73.
- Unnikrishnan AG, Sahay RK, Phadke U, Sharma SK, Shah P, et al. (2022) Cardiovascular risk in newly diagnosed type 2 diabetes patients in India. PLoS One. 17:e0263619.
- Kumar A, Mazumdar A, Bhattacharjee AK, Gupta A, Dasgupta A, et al. (2025) Risk factors associated with Indian type 2 diabetes patients with chronic kidney disease: CITE study, a cross-sectional, real-world, observational study. Renewable Energy. 26: 244.
- Behera S, Lamare AA, Patnaik B, Rattan R, Das S (2020) Study of lipid abnormalities in type 2 diabetes mellitus patients with nephropathy in Eastern India. Journal of Diabetes Mellitus. 10:16.
- Saboo B, Prajapati C, Muralidharan P, Qamra A (2024) DiSi Survey: Use of Generic DPP4i-SGLT2i Fixed-dose Combinations in Indian Clinical Practice. J Assoc Physicians India. 72:22-24.
- Pavithran V, Balachandran P, Saajeeth CI, Hariharan A (2025) Analyzing the shift in prescribing patterns: Sodium Glucose Co-transpoter 2 Inhibitors. Dipeptidyl Peptidase 4 Inhibitors and Traditional Antidiabetic Drugs. Online J Health Allied Scs. 24:5.
- Bhat KA, Singh NP, Maddukuri HR, Routray SN, Sharma S, et al. (2024) ClinicalProfile,comorbidities and therapies in type 2 diabetes patients on sitagliptin-based therapy in Indian Outpatient setting. Cureus. 16:e74820.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.