A Case Report of Gastric Heterotopia in the Rectum: Clinical Presentation, Diagnosis and Management
by Margherita Granziera1, Catrin Simeth1, Deborah Bonazza2, Fabrizio Zanconati2, Fabio Monica1
1Gastroenterology and Digestive Endoscopy Unit, Academic Hospital Cattinara, Strada di Fiume, 447, 34149 Trieste, Italy
2Pathological Anatomy and Histology, Academic Hospital Cattinara, Strada di Fiume, 447, 34149 Trieste, Italy
*Corresponding author: Margherita Granziera, Gastroenterology and Digestive Endoscopy Unit, Academic Hospital Cattinara, Strada di Fiume, 447, 34149 Trieste, Italy
Received Date: 04 November 2025
Accepted Date: 10 November 2025
Published Date: 12 November 2025
Citation: Granziera M, Simeth C, Bonazza D, Zanconati F, Monica F. (2025). A Case Report of Gastric Heterotopia in the Rectum: Clinical Presentation, Diagnosis and Management. Ann Case Report. 10: 2442. https://doi.org/10.29011/2574-7754.102442
Introduction
Gastric heterotopia (GHT) is defined as the presence of gastric tissue at a non-physiological site. Most cases of gastric heterotopia are observed in the esophagus, duodenum and Meckel’s diverticulum [1, 2], while large bowel lesions are an extremely rare and most frequent location of colonic lesions is the rectum [3].
For upper and midgut locations the incidence ranges from 0.1 to 11%, while for the lower gastro-intestinal tract, such as the rectum and anus, the incidence is even lower [4].
Since the first description by Ewell and Jackson in 1939, only about 50 cases of heterotopic gastric mucosa (HGM) in the rectum have been reported [1]. HGM in the rectum is typically identified endoscopically as a polyp [5], diverticulum [6], erythematous patch [5], or ulcer [7, 8] and is usually found between 5 and 8 cm from the anal verge in the posterolateral wall of this area [9]. Clinical presentation can vary from completely asymptomatic (17.02%) to diarrhea (4.25%), anal tenesmus (10.63%), anal pain (14.85%), abdominal pain (17.02%), or gastrointestinal bleeding (55.32%) [5]. HGM-related complications include ulcers, fistulas, or bowel perforation, and they are more common in the pediatric population [10].
Men are predominantly affected (M:F ratio 20:7), and the median age of diagnosis is around 18 years, although cases have been reported from as early as 6 months of age to as late as 51 years [1].
Here, we report a case of gastric heterotopia in the rectum, highlighting its clinical features, diagnostic challenges, and management, with a discussion based on a comprehensive review of the literature.
Case Report
A 50-year-old man was referred to the gastroenterology department at Cattinara Hospital (Trieste) due to perineal pain and diarrhea associated with episodes of rectal bleeding that had persisted for about two months. The patient’s medical history was negative for pre-existing conditions, and there was no family history of digestive tract diseases.
The patient was initially referred by his general practitioner for proctological evaluation which detected no abnormalities but, taking in account the symptoms, the surgeon recommended further investigations, including laboratory tests and a colonoscopy.
Laboratory tests were within normal limits: creatinine 0.73 mg/ dl, AST/ALT 26/21 U/L, white blood cell count 4510/mcl, haemoglobin 14.8 g/dl, platelet count 196000/mcl, with fecal calprotectin below 5.00 mcg/g.
Colonoscopy was performed exploring terminal ileum, without pathological findings in the colon with normal mucosa and regular haustration in all segments. Rectal retroflexion revealed a sessile hyperemic polyp (0-Is) approximately 12 mm in size near the anal margin, without erosion or ulcer (Figure 1).
The polyp was treated with the injection of a saline solution, methylene blue, and adrenaline (1:20.000) into the submucosal space beneath the lesion, creating a safety cushion. This cushion lifted the lesion, which facilitated the capture and removal en bloc with a diathermic snare. Two TTS clips were placed for prevention of delayed bleeding.
Histological examination revealed the presence of oxyntic and pseudo-pyloric gastric tissue, without signs of dysplasia, consistent with gastric ectopia (Figure 2). Due to complete removal of the lesion and the absence of dysplasia, the patient was advised to return for regional screening with fecal occult blood test after five years. During follow up patient remained asymptomatic, with no further episodes of hematochezia or perineal pain, and regular bowel habits.
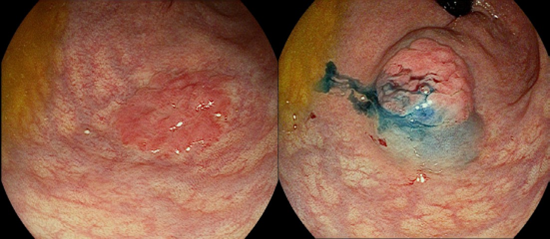
Figure 1: 12 mm sessile polyp in the rectum found on retroflexion, visualized using I-scan imaging, pre (A) and post. (B) saline solution, methylene blue, and adrenaline (1:20.000) injection.
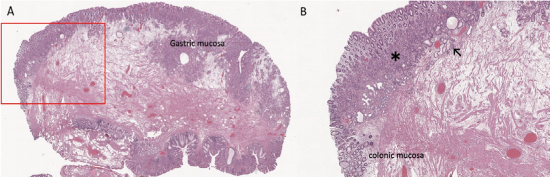
Figure 2: Hematoxylin and Eosin (H&E) Staining - Polypoid Lesion Resection 1x (A) and 2x (B).
Histopathological examination of the resected specimen reveals the presence of ectopic gastric tissue within a loose stromal context, interspersed with dispersed fibers of the muscularis mucosae. At the resection margins, colonic mucosa with preserved architecture is observed, exhibiting crypts that are focally ectatic.
The superficial mucosal portion of the polyp displays gastric-type mucosa, characterized by oxyntic epithelium (*), pyloric glands (↖), and foveolar epithelium. These findings suggest the presence of heterotopic gastric mucosa within the colonic environment.
Discussion
GHT has been reported in nearly every anatomical site of the GI tract, including gallbladder and pancreas [11]. Large bowel lesions are an extremely rare clinical entity; however, the most common location of colonic lesions is the rectum [12]. GHT can occur due to congenital heteroplasia during organogenesis or acquired metaplasia during the repair of damaged epithelium [13].
The mucosal tissue in gastric heteroplasia is composed of fullthickness gastric epithelium and glands, distinguishing it from metaplasia, which usually involves only foveolar-type epithelium mixed with the original tissue [14].
Definitive diagnosis is determined by histopathological findings of gastric mucosa which is characterized by oxyntic gastric tissue in most cases or mixed oxyntic and antral mucosa, or, more rarely, by antral or cardiac mucosa [15]. Moreover, in rare instances Helicobacter pylori organisms are identified in GHT of the rectum [1]. The successful eradication of Helicobacter pylori infection has been reported to lead to resolution of abdominal pain, further confirming the complex nature of this condition [16].
As our understanding of this condition is primarily based on sporadic case reports, there are no established guidelines for optimal treatment and surveillance.
Medical treatment with H2 receptor blockers or proton pump inhibitors can partially or completely resolve symptoms by inhibiting acid production. However, they do not cause the lesion regression and symptoms may recur once the treatment is discontinued [17, 18]. Therefore, definitive treatment remains surgical or endoscopic resection.
Surgical transanal excision [19], endoscopic mucosal resection (EMR) [18] and, in selected cases, endoscopic submucosal dissection (ESD) have been described [15].
It is important to notice that while GHT has the potential to progress to malignancy, the exact rate of malignancy remains unknown. However, cases of colonic adenocarcinoma arising from gastric heterotopia have been documented [20, 21].
This highlights the need for an appropriate management and careful surveillance of GHT cases in the rectum [16].
Conclusion
GHT of the rectum is a rare condition that should be considered in cases of unexplained lower gastrointestinal bleeding. Diagnosis can typically be made through colonoscopy and histological analysis.
When the lesion is located close to the anal verge, it may be missed during examination, emphasizing the importance of thoroughly inspecting the rectal segment just above the anus during colonoscopy in cases of rectal bleeding.
Once GHT is diagnosed, medical treatment may suffice for managing local symptoms. However, endoscopic or surgical resection remains the gold standard for completely removing lesions or managing persistent bleeding.
Given that the understanding of this condition is largely based on sporadic case reports, optimal treatment and surveillance should be tailored to the patient taking in account medical history, comorbidities and risk factors.
References
- Srinivasan R, Loewenstine H, Mayle JE. (1999). “Sessile polypoid gastric heterotopia of rectum: a report of 2 cases and review of the literature” Arch Pathol Lab Med. 123: 222-224.
- Dinarvand P, Vareedayah AA, Phillips NJ, Hachem C, Lai J. (2017). “Gastric heterotopia in rectum: A literature review and its diagnostic pitfall”. SAGE Open Med Case Rep. 5: 2050313X17693968.
- Salem GA, Fazili J, Ali T. (2017). “Gastric heterotopia in the rectum. A rare cause of ectopic gastric tissue” Arab J Gastroenterol. 18: 42-43.
- Terada T. (2011). “Heterotopic gastric mucosa of the gastrointestinal tract: a histopathologic study of 158 cases” Pathol Res Pract. 207: 148-50.
- Galassi L, Guerrazzi G, Romeo BG, Magni M, Tagliabue F, et al. (2021). “Gastric Mucosa Heterotopia in Distal Rectum: a Case Report and Narrative Literature Review”. SN Comprehensive Clinical Medicine. 3: 1469-1476.
- Chen WG, Zhu HT, Yang M, Xu GQ, Chen LH, et al. (2018). “Large heterotopic gastric mucosa and a concomitant diverticulum in the rectum: Clinical experience and endoscopic management”. World J Gastroenterol. 24: 3462-3468.
- Wacrenier A, Fayoux P, Augusto D, Laussel AC, Gosselin B, et al. (2002). “Gastric heterotopia in the nasopharynx”. Int J Pediatr Otorhinolaryngol. 64: 65-67.
- Steele SR, Mullenix PS, Martin MJ, Ormseth E, Weppler E. (2004). “Heterotopic gastric mucosa of the anus: a case report and review of the literature”. Am Surg. 70: 715-719.
- Nakanishi Y, Merenda G. (2015). “Heterotopic fundic and pyloric-type gastric mucosa in the rectum presented as a foveolar hyperplastic polyp”. Case Reports in Clinical Pathology. 2: 1-4.
- Di Nora A, Costanza G, Pizzo F, Oliva CF, Di Mari A, et al. (2022) “Gray matter heterotopia: clinical and neuroimaging report on 22 children”. Acta Neurol Belg. 122: 153-162.
- Jordan FT, Mazzeo RJ, Soiderer MH. (1983). “Heterotopic gastric mucosa of the rectum. A rare cause of rectal bleeding”. Arch Surg. 118: 878-880.
- Salem GA, Fazili J, Ali T. (2017). “Gastric heterotopia in the rectum. A rare cause of ectopic gastric tissue”. Arab J Gastroenterol. 18: 42-43.
- Yu L, Yang Y, Cui L, Peng L, Sun G. (2014). “Heterotopic gastric mucosa of the gastrointestinal tract: prevalence, histological features, and clinical characteristics”. Scand J Gastroenterol. 49: 138-144.
- Ok CY, Akalin A. (2012). “Gastric Heterotopia in the Rectum”. J Med Cases. 3: 113-115.
- Iacopini F, Gotoda T, Elisei W, Rigato P, Montagnese F, et al. (2016). “Heterotopic gastric mucosa in the anus and rectum: first case report of endoscopic submucosal dissection and systematic review”. Gastroenterol Rep (Oxf). 4: 196-205.
- Dye KR, Marshall BJ, Frierson Jr HF, Pambianco DJ, McCallum RW. (1990). “Campylobacter pylori colonizing heterotopic gastric tissue in the rectum”. Am J Clin Pathol. 93: 144-147.
- Sauer CG, Bickston SJ, Borowitz SM. (2010). “Gastric heterotopia of the rectum”. J Pediatr Gastroenterol Nutr. 50: 329-333.
- Soares J, Soares J, Ferreira C, Marques M, Corujeira S, Tavares M, et al. (2017). Endoscopic Mucosectomy in a Child Presenting with Gastric Heterotopia of the Rectum. GE Port J Gastroenterol. 24: 288291.
- Cheli M, Alberti D, Vavassori D, Sonzogni A, Pedretti E, et al. (2007). Heterotopic rectal gastric mucosa: a rare cause of lower gastrointestinal bleeding in children. Case report and review of pediatric literature”. Eur J Pediatr Surg. 17: 50-54.
- Ko H, Park SY, Cha EJ, Sohn JS. (2013). Colonic adenocarcinoma arising from gastric heterotopia: a case study”. Korean J Pathol. 47: 289-292.
- Christensen WN, Sternberg SS. (1987). “Adenocarcinoma of the upper esophagus arising in ectopic gastric mucosa. Two case reports and review of the literature”. Am J Surg Pathol. 11: 397-402.
© by the Authors & Gavin Publishers. This is an Open Access Journal Article Published Under Attribution-Share Alike CC BY-SA: Creative Commons Attribution-Share Alike 4.0 International License. Read More About Open Access Policy.